Abstract
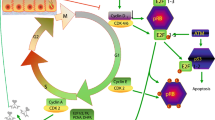
HuR (ELAV11 (embryonic lethal, abnormal vision)-like 1), a ubiquitously expressed member of the ELAV-like RNA-binding protein family, has been shown to regulate the stability and translation of mRNAs that encode factors regulating cellular senescence, thereby impacting on aging. In this review, we discuss the current knowledge of HuR’s role in vascular cell senescence and vascular aging.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wang W. Regulatory RNA-binding proteins in senescence. Ageing Resh Rev, 2012, 11: 485–490
Laurent S. Defining vascular aging and cardiovascular risk. J Hypertens, 2012, 30: S3–8
Inukai S, Slack F. microRNAs and the genetic network in aging. J Mol Biol, 2013, 425: 3601–3608
Yoon JH, Abdelmohsen K, Gorospe M. Posttranscriptional gene regulation by long noncoding RNA. J Mol Biol, 2013, 425: 3723–3730
Peng SS, Chen CY, Xu N, Shyu AB. RNA stabilization by the AU-rich element binding protein, HuR, an ELAV protein. EMBO J, 1998, 17: 3461–3470
Minamino T, Miyauchi H, Yoshida T, Tateno K, Kunieda T, Komuro I. Vascular cell senescence and vascular aging. J Mol Cell Cardiol, 2004, 36: 175–183
Foreman KE, Tang J. Molecular mechanisms of replicative senescence in endothelial cells. Exp Gerontol, 2003, 38: 1251–1257
Watanabe Y, Lee SW, Detmar M, Ajioka I, Dvorak HF. Vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) delays and induces escape from senescence in human dermal microvascular endothelial cells. Oncogene, 1997, 14: 2025–2032
Brewer G. Messenger RNA decay during aging and development. Ageing Res Rev, 2002, 1: 607–625
Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res, 1961, 25: 585–621
Wang W, Caldwell MC, Lin S, Furneaux H, Gorospe M. HuR regulates cyclin A and cyclin B1 mRNA stability during cell proliferation. EMBO J, 2000, 19: 2340–2350
Wang W, Yang X, Cristofalo V, Holbrook N, and Gorospe M. Loss of HuR is linked to reduced expression of proliferative genes during replicative senescence. Mol Cell Biol, 2001, 21: 5889–5898
Chang N, Yi J, Guo G, Liu X, Shang Y, Tong T, Cui Q, Zhan M, Gorospe M, Wang W. HuR uses AUF1 as a cofactor to promote p16INK4 mRNA decay. Mol Cell Biol, 2010, 30: 3875–3886
Zhang X, Liu Z, Yi J, Tang H, Xing J, Yu M, Tong T, Shang Y, Gorospe M, Wang W. NSun2 stabilizes p16INK4 mRNA by methylating the p16 3′UTR. Nat Commun, 2012, 3: 712
Werth D, Grassi G, Konjer N, Dapas B, Farra R, Giansante C, Kandolf R, Guarnieri G, Nordheim A, Heidenreich O. Proliferation of human primary vascular smooth muscle cells depends on serum response factor. Eur J Cell Biol, 2010, 89: 216–224
Li M, Chiu JF, Gagne J, Fukagawa NK. Age-related differences in insulin-like growth factor-1 receptor signaling regulates Akt/ FOXO3a and ERK/Fos pathways in vascular smooth muscle cells. J Cell Physiol, 2008, 217: 377–387
Kullmann M, Göpfert U, Siewe B, Hengst L. ELAV/Hu proteins inhibit p27 translation via an IRES element in the p27 5′UTR. Genes Dev, 2012, 16: 3087–3099
Klöss S, Rodenbach D, Bordel R, Mülsch A. Human-antigen R (HuR) expression in hypertension: downregulation of the mRNA stabilizing protein HuR in genetic hypertension. Hypertension, 2005, 45: 1200–1206
Dean JL, Wait R, Mahtani KR, Sully G, Clark AR, Saklatvala J. The 3′ untranslated region of tumor necrosis factor alpha mRNA is a target of the mRNA-stabilizing factor HuR. Mol Cell Biol, 2001, 21: 721–730
Levy NS, Chung S, Furneaux H, Levy AP. Hypoxic stabilization of vascular endothelial growth factor mRNA by the RNA-binding protein HuR. J Biol Chem, 1998, 273: 6417–6423
Rhee WJ, Ni CW, Zheng Z, Chang K, Jo H, Bao G. HuR regulates the expression of stress-sensitive genes and mediates inflammatory response in human umbilical vein endothelial cells. Proc Natl Acad Sci USA, 2010, 107: 6858–6863
Abdelmohsen K, Pullmann R Jr., Lal A, Kim HH, Galban S, Yang X, Blethrow JD, Walker M, Shubert J, Gillespie DA, Furneaux H, Gorospe M. Phosphorylation of HuR by Chk2 regulates SIRT1 expression. Mol Cell, 2007, 25: 543–557
Nabors LB, Gillespie GY, Harkins L, King PH. HuR, a RNA stability factor, is expressed in malignant brain tumors and binds to adenine- and uridine-rich elements within the 3′ untranslated regions of cytokine and angiogenic factor mRNAs. Cancer Res, 2001, 61: 2154–2161
Chen HZ, Wan YZ, Liu DP. Cross-talk between SIRT1 and p66Shc in vascular diseases. Trends Cardiovasc Med, 2013, 23: 237–241
Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B, Howitz KT, Gorospe M, de Cabo R, Sinclair DA. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science, 2004, 305: 390–392
Rodier F, Campisi J. Four faces of cellular senescence. J Cell Biol, 2011, 192: 547–556
Wu WH, Wang SH, Kuan II, Kao YS, Wu PJ, Liang CJ, Chien HF, Kao CH, Huang CJ, Chen YL. Sesamin attenuates intercellular cell adhesion molecule-1 expression in vitro in TNF-alpha-treated human aortic endothelial cells and in vivo in apolipoprotein-E-deficient mice. Mol Nutr Food Res, 2010, 54: 1340–1350
Doller A, Pfeilschifter J, Eberhardt W. Signalling pathways regulating nucleo-cytoplasmic shuttling of the mRNA-binding protein HuR. Cell Signal, 2008, 20: 2165–2173
Doller A, Schlepckow K, Schwalbe H, Pfeilschifter J, Eberhardt W. Tandem phosphorylation of serines 221 and 318 by protein kinase Cdelta coordinates mRNA binding and nucleocytoplasmic shuttling of HuR. Mol Cell Biol, 2010, 30: 1397–1410
Amadio M, Bucolo C, Leggio GM, Drago F, Govoni S, Pascale A. The PKCbeta/HuR/VEGF pathway in diabetic retinopathy. Biochem Pharmacol, 2010, 80: 1230–1237
Kim MY, Hur J, Jeong S. Emerging roles of RNA and RNA-binding protein network in cancer cells. BMB Rep, 2009, 42: 125–130
Lal A, Mazan-Mamczarz K, Kawai T, Yang X, Martindale JL, Gorospe M. Concurrent versus individual binding of HuR and AUF1 to common labile target mRNAs. EMBO J, 2004, 23: 3092–3102
Marasa BS, Srikantan S, Martindale JL, Kim MM, Lee EK, Gorospe M, Abdelmohsen K. microRNA profiling in human diploid fibroblasts uncovers miR-519 role in replicative senescence. Aging (Albany NY), 2010, 2: 333–343
Abdelmohsen K, Srikantan S, Kuwano Y, Gorospe M. miR-519 reduces cell proliferation by lowering RNA-binding protein HuR levels. Proc Natl Acad Sci USA, 2008, 105: 20297–20302
Yi J, Chang N, Liu X, Guo G, Xue L, Tong T, Gorospe M, Wang W. Reduced nuclear export of HuR mRNA by HuR is linked to the loss of HuR in replicative senescence. Nucleic Acids Res, 2010, 38: 1547–1558
Cheng HS, Sivachandran N, Lau A, Boudreau E, Zhao JL, Baltimore D, Delgado-Olguin P, Cybulsky MI, Fish JE. microRNA-146 represses endothelial activation by inhibiting pro-inflammatory pathways. EMBO Mol Med, 2013, 5: 949–966
Xu F, Zhang X, Lei Y, Liu X, Liu Z, Tong T, Wang W. Loss of repression of HuR translation by miR-16 may be responsible for the elevation of HuR in human breast carcinoma. J Cell Biochem, 2010, 111: 727–734
Guo X, Wu Y, Hartley RS. microRNA-125a represses cell growth by targeting HuR in breast cancer. RNA Biol, 2009, 6: 575–583
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wang, W. HuR and post-transcriptional regulation in vascular aging. Sci. China Life Sci. 57, 863–866 (2014). https://doi.org/10.1007/s11427-014-4706-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4706-2