Abstract
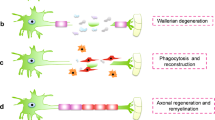
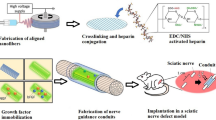
Peripheral nerve functional recovery after nerve injury generally requires multiple growth factors by synergistic effect. However, the optical combination of multiple synergistic growth factors for axonal regeneration has been scarcely considered up to now. Meanwhile, the use of growth factors in promoting nerve regeneration was limited by its short biological half-life in vivo, its vulnerability to structure disruption or hydrolyzation, leading to loss of bioactivity. Herein, a novel polymeric nanoparticle delivery system composed of heparin and ɛ-poly-L-lysine (PL) was prepared for control release of nerve growth factor (NGF) and basic fibroblast growth factor (bFGF). The nanoparticles were synthesized by polyelectrolyte complexation in aqueous solution at room temperature, followed by cross-linking with biological genipin. The obtained nanoparticles had a spherical shape, with a mean diameter of about 246 nm, and high growth factors encapsulation efficiency as well as good stability. NGF and bFGF were encapsulated in the nanoparticles and showed a continuous and slow release behavior in vitro. The bioactivities of the released growth factors were evaluated, and exhibited the synergistic effect. The controlled release of the dual synergistic growth factors would improve the treatment of peripheral nerve injury to mimic the natural cellular microenvironments.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Konofaos P, Halen JPV. Nerve repair by means of tubulization: past, present, future. J Reconstruct Microsurg, 2013, 29: 149–163
Wang XD, Hu W, Cao Y, Yao J, Wu J, Gu XS. Dog sciatic nerve regeneration across a 30-mm defect bridged by a chitosan/PGA artificial nerve graft. Brain, 2005, 128: 1897–1910
Evans GR. Challenges to nerve regeneration. Semin Surg Oncol, 2000, 19: 312–318
Huang W, Begum R, Barber T, Ibba V, Tee NC, Hussain M, Arastoo M, Yang Q, Robson LG, Lesage S, Gheysens T, Skaer NJ, Knight DP, Priestley JV. Regenerative potential of silk conduits in repair of peripheral nerve injury in adult rats. Biomaterials, 2012, 33: 59–71
Lee BK, Ju YM, Cho JG, Jackson JD, Lee SJ, Atala A, Yoo JJ. End-to-side neurorrhaphy using an electrospun PCL/collagen nerve conduit for complex peripheral motor nerve regeneration. Biomaterials, 2012, 33: 9027–9036
Khaing ZZ, Schmidt CE. Advances in natural biomaterials for nerve tissue repair. Neurosci Lett, 2012, 519: 103–114
Belkas JS, Shoichet MS, Midha R. Axonal guidance channels in peripheral nerve regeneration. Oper Tech Orthop, 2004, 14: 190–198
Pabari A, Yang SY, Seifalian AM, Mosahebi A. Modern surgical management of peripheral nerve gap. J Plastic Reconstruct Aesthet Surg, 2010, 63: 1941–1948
Daly W, Yao L, Zeugolis D, Windebank A, Pandit A. A biomaterials approach to peripheral nerve regeneration: bridging the peripheral nerve gap and enhancing functional recovery. J Royal Soc Interface, 2012, 9: 202–221
Ho PR, Coan GM, Cheng ET, Niell C, Tarn DM, Zhou H, Sierra D, Terris DJ. Repair with collagen tubules linked with brain-derived neurotrophic factor and ciliary neurotrophic factor in a rat sciatic nerve injury model. Arch Otolaryngol Head Neck Surg, 1998, 124: 761–766
Aebischer P, Salessiotis AN, Winn SR. Basic fibroblast growth factor released from synthetic guidance channels facilitates peripheral nerve regeneration across long nerve gaps. J Neurosci Res, 1989, 23: 282–289
Gu X, Ding F, Yang Y, Liu J. Construction of tissue engineered nerve grafts and their application in peripheral nerve regeneration. Prog Neurobiol, 2011, 93: 204–230
Lundborg G. A 25-year perspective of peripheral nerve surgery: evolving neuroscientific concepts and clinical significance. J Hand Surg Am, 2000, 25: 391–414
Flight MH. Neurotrophic factors: ride back to the nucleus. Nat Rev Neurosci, 2011, 12: 550–551
Davis JB, Stroobant P. Platelet-derived growth factors and fibroblast growth factors are mitogens for rat Schwann cells. J Cell Biol, 1990, 110: 1353–1360
Torigoe K, Hashimoto K, Lundborg G. A role of migratory Schwann cells in a conditioning effect of peripheral nerve regeneration. Exp Neurol, 1999, 160: 99–108
Madduri S, Gander B. Growth factor delivery systems and repair strategies for damaged peripheral nerves. J Contr Release, 2012, 161: 274–282
Madduri S, Papaloizos M, Gander B. Synergistic effect of GDNF and NGF on axonal branching and elongation in vitro. Neurosci Res, 2009, 65: 88–97
Zeng W, Huang J, Xiao W, Rong M, Yuan Z, Luo Z. Ionically cross-linked chitosan microspheres for controlled release of bioactive nerve growth factor. Int J Pharm, 2011, 421: 283–290
Dyondi D, Webster TJ, Banerjee R. A nanoparticulate injectable hydrogel as a tissue engineering scaffold for multiple growth factor for multiple growth factor delivery for bone regeneration. Int J Nanomed, 2013, 8: 47–59
Zhang Y, Chan HF, Leong KW. Advanced materials and processing for drug delivery: the past and the future. Adv Drug Deliv Rev, 2013, 65: 104–120
Liu JM, Wang X, Marin-Muller C, Wang H, Lin PH, Yao Q, Chen C. Current advances in research and clinical application of PLGA-based nanotechnology. Expert Rev Mol Diagn, 2009, 9: 325–341
Shukla SC, Singh A, Pandey AK, Mishra A. Review on production and medical applications of epsilon-polylysine. Biochem Eng J, 2012, 65: 70–81
Lin YH, Chang CH, Wu YS, Hsu YM, Chiou SF, Chen YJ. Development of pH-responsive chitosan/heparin nanoparticles for stomach-specific anti-Helicobacter pylori therapy. Biomaterials, 2009, 30: 3332–3342
Chu HH, Johnson NR, Mason NS, Wang YD. A [polycation:heparin] complex releases growth factors with enhanced bioactivity. J Contr Release, 2011, 150: 157–163
Goncalves NP, Oliveira H, Pego AP, Saraiva MJ. A novel nanoparticle delivery system for in vivo targeting of the sciatic nerve: impact on regeneration. Nanomedicine, 2012, 7: 1167–1180
Chau CH, Shum DK, Chan YS, So KF. Heparan sulphates upregulate regeneration of transected sciatic nerves of adult guinea-pigs. Eur J Neurosci, 1999, 11: 1914–1926
Yang YM, Zhao WJ, He JH, Zhao YH, Ding F, Gu XS. Nerve conduits based on immobilization of nerve growth factor onto modified chitosan by using genipin as a crosslinking agent. Eur J Pharm Biopharm, 2011, 79: 519–525
Mi FL, Tan YC, Liang HF, Sung HW. In vivo biocompatibility and degradability of a novel injectable-chitosan-based implant. Biomaterials, 2002, 23: 181–191
Zern BJ, Chu HH, Wang YD. Control growth factor release using a self-assembled [polycation:heparin] complex. PLoS ONE, 2010, 5: e11017
Stanwick JC, Baumann MD, Schoichet MS. Enhanced neurotrophin-3 bioactivity and release from a nanoparticle-loaded composite hydrogel. 3 bioactivity and release from a nanoparticle-loaded composite hydrogel. J Contr Release, 2012, 160: 666–675
Sakiyama-Elbert SE, Hubbell JA. Controlled release of nerve growth factor from a heparin-containing fibrin-based cell ingrowth matrix. J Contr Release, 2000, 69: 149–158
Freeman I, Kedem A, Cohen S. The effect of sulfation of alginate hydrogels on the specific binding and controlled release of heparin-binding proteins. Biomaterials, 2008, 29: 3260–3268
Chung J, Kubota H, Ozaki Y, Uda S, Kuroda S. Timing-dependent actions of NGF required for cell differentitation. PLoS ONE, 2010, 5: e9011
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Zhang, L., Zhou, Y., Li, G. et al. Nanoparticle mediated controlled delivery of dual growth factors. Sci. China Life Sci. 57, 256–262 (2014). https://doi.org/10.1007/s11427-014-4606-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4606-5