Abstract
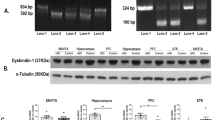
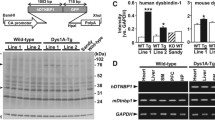
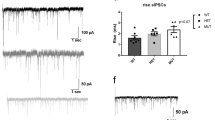
The Drosophila homolog of schizophrenia susceptibility gene dysbindin (Ddysb) affects a range of behaviors through regulation of multiple neurotransmitter signals, including dopamine activity. To gain insights into mechanisms underlying Ddysb-dependent regulation of dopamine signal, we investigated interaction between Ddysb and Ebony, the Drosophila β-alanyl-monoamine synthase involved in dopamine recycling. We found that Ddysb was capable of regulating expression of Ebony in a bi-directional manner and its subcellular distribution. Such regulation is confined to glial cells. The expression level of ebony and its accumulation in glial soma depend positively on Ddysb activity, whereas its distribution in glial processes is bound to be reduced in response to any alterations of Ddysb from the normal control level, either an increase or decrease. An optimal binding ratio between Dysb and Ebony might contribute to such non-linear effects. Thus, Ddysb-dependent regulation of Ebony could be one of the mechanisms that mediate dopamine signal.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Straub RE, Jiang Y, MacLean CJ, Ma Y, Webb BT, Myakishev MV, Harris-Kerr C, Wormley, B, Sadek H, Kadambi B, Cesare AJ, Gibberman A, Wang X, O’Neill FA, Walsh D, Kendler KS, Genetic variation in the 6p22.3 gene DTNBP1, the human ortholog of the mouse dysbindin gene, is associated with schizophrenia. Am J Hum Genet, 2002, 71: 337–348
Williams NM, O’Donovan MC, Owen MJ. Is the dysbindin gene (DTNBP1) a susceptibility gene for schizophrenia? Schizophr Bull, 2005, 31: 800–805
Talbot K, Eidem WL, Tinsley CL, Benson MA, Thompson EW, Smith RJ, Hahn CG, Siegel SJ, Trojanowski JQ, Gur RE, Blake DJ, Arnold SE. Dysbindin-1 is reduced in intrinsic, glutamatergic terminals of the hippocampal formation in schizophrenia. J Clin Invest, 2004, 113: 1353–1363
Tang J, LeGros RP, Louneva N, Yeh L, Cohen JW, Hahn CG, Blake DJ, Arnold SE, Talbot K. Dysbindin-1 in dorsolateral prefrontal cortex of schizophrenia cases is reduced in an isoform-specific manner unrelated to dysbindin-1 mRNA expression. Hum Mol Genet, 2009, 18: 3851–3863
Benson MA, Newey SE, Martin-Rendon E, Hawkes R, Blake DJ. Dysbindin, a novel coiled-coil-containing protein that interacts with the dystrobrevins in muscle and brain. J Biol Chem, 2001, 276: 24232–24241
Veroni C, Grasso M, Macchia G, Ramoni C, Ceccarini M, Petrucci TC, Macioce P. Beta-dystrobrevin, a kinesin-binding receptor, interacts with the extracellular matrix components pancortins. J Neurosci Res, 2007, 85: 2631–2639
Li W, Zhang Q, Oiso N, Novak EK, Gautam R, O’Brien EP, Tinsley CL, Blake DJ, Spritz RA, Copeland NG, Jenkins NA, Amato D, Roe BA, Starcevic M, Dell’Angelica EC, Elliott RW, Mishra V, Kingsmore SF, Paylor RE, Swank RT. Hermansky-Pudlak syndrome type 7 (HPS-7) results from mutant dysbindin, a member of the biogenesis of lysosome-related organelles complex 1 (BLOC-1). Nat Genet, 2003, 35: 84–89
Starcevic M, Dell’Angelica EC. Identification of snapin and three novel proteins (BLOS1, BLOS2, and BLOS3/reduced pigmentation) as subunits of biogenesis of lysosome-related organelles complex-1 (BLOC-1). J Biol Chem, 2004, 279: 28393–28401
Lee HH, Nemecek D, Schindler C, Smith WJ, Ghirlando R, Steven AC, Bonifacino JS, Hurley JH. Assembly and architecture of biogenesis of lysosome-related organelles complex-1 (BLOC-1). J Biol Chem, 2012, 287: 5882–5890
Iizuka Y, Sei Y, Weinberger DR, Straub RE. Evidence that the BLOC-1 protein dysbindin modulates dopamine D2 receptor internalization and signaling but not D1 internalization. J Neurosci, 2007, 27: 12390–12395
Hattori S, Murotani T, Matsuzaki S, Ishizuka T, Kumamoto N, Takeda M, Tohyama M, Yamatodani A, Kunugi H, Hashimoto R. Behavioral abnormalities and dopamine reductions in sdy mutant mice with a deletion in Dtnbp1, a susceptibility gene for schizophrenia. Biochem Biophys Res Commun, 2008, 373: 298–302
Shao L, Shuai Y, Wang J, Feng S, Lu B, Li Z, Zhao Y, Wang L, Zhong Y. Schizophrenia susceptibility gene dysbindin regulates glutamatergic and dopaminergic functions via distinctive mechanisms in Drosophila. Proc Natl Acad Sci USA, 2011, 108: 18831–18836
Richardt A, Kemme T, Wagner S, Schwarzer D, Marahiel MA, Hovemann BT. Ebony, a novel nonribosomal peptide synthetase for beta-alanine conjugation with biogenic amines in Drosophila. J Biol Chem, 2003, 278: 41160–41166
Jacobs ME. Influence of beta-alanine on ultrastructure, tanning, and melanization of Drosophila melanogaster cuticles. Biochem Genet, 1980, 18: 65–76
Jacobs ME. Influence of beta-alanine on mating and territorialism in Drosophila melanogaster. Behav Genet, 1978, 8: 487–502
Liu T, Dartevelle L, Yuan C, Wei H, Wang Y, Ferveur JF, Guo A. Increased dopamine level enhances male-male courtship in Drosophila. J Neurosci, 2008, 28: 5539–5546
Newby LM, Jackson FR. Drosophila ebony mutants have altered circadian activity rhythms but normal eclosion rhythms. J Neurogenet, 1991, 7: 85–101
Suh J, Jackson FR. Drosophila ebony activity is required in glia for the circadian regulation of locomotor activity. Neuron, 2007, 55: 435–447
Biffo S, Grillo M, Margolis FL. Cellular localization of carnosine-like and anserine-like immunoreactivities in rodent and avian central nervous system. Neuroscience, 1990, 35: 637–651
Tsuneyoshi Y, Tomonaga S, Asechi M, Morishita K, Denbow DM, Furuse M. Central administration of dipeptides, beta-alanyl-BCAAs, induces hyperactivity in chicks. BMC Neurosci, 2007, 8: 37
Wittkopp PJ, True JR, Carroll SB. Reciprocal functions of the Drosophila yellow and ebony proteins in the development and evolution of pigment patterns. Development, 2002, 129: 1849–1858
Wang L, Chiang HC, Wu W, Liang B, Xie Z, Yao X, Ma W, Du S, Zhong Y. Epidermal growth factor receptor is a preferred target for treating amyloid-beta-induced memory loss. Proc Natl Acad Sci USA, 2012, 109: 16743–16748
Chiang AS, Liu YC, Chiu SL, Hu SH, Huang CY, Hsieh CH. Three-dimensional mapping of brain neuropils in the cockroach, Diploptera punctata. J Comp Neurol, 2001, 440: 1–11
Schuldiner O, Berdnik D, Levy JM, Wu JS, Luginbuhl D, Gontang AC, Luo L. piggyBac-based mosaic screen identifies a postmitotic function for cohesin in regulating developmental axon pruning. Dev Cell, 2008, 14: 227–238
Gokhale A, Larimore J, Werner E, So L, Moreno-De-Luca A, Lese-Martin C, Lupashin VV, Smith Y, Faundez V. Quantitative proteomic and genetic analyses of the schizophrenia susceptibility factor dysbindin identify novel roles of the biogenesis of lysosome-related organelles complex 1. J Neurosci, 2012, 32: 3697–3711
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at link.springer.com
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Lu, B., Shao, L., Feng, S. et al. The β-alanyl-monoamine synthase ebony is regulated by schizophrenia susceptibility gene dysbindin in Drosophila. Sci. China Life Sci. 57, 46–51 (2014). https://doi.org/10.1007/s11427-013-4595-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4595-9