Abstract
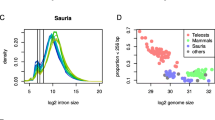
All eukaryotic genomes have genes with introns in variable sizes. As far as spliceosomal introns are concerned, there are at least three basic parameters to stratify introns across diverse eukaryotic taxa: size, number, and sequence context. The number parameter is highly variable in lower eukaryotes, especially among protozoan and fungal species, which ranges from less than 4% to 78% of the genes. Over greater evolutionary time scales, the number parameter undoubtedly increases as observed in higher plants and higher vertebrates, reaching greater than 12.5 exons per gene in average among mammalian genomes. The size parameter is more complex, where multiple modes appear at work. Aside from intronless genes, there are three other types of intron-containing genes: half-sized, minimal, and size-expandable introns. The half-sized introns have only been found in a limited number of genomes among protozoan and fungal lineages and the other two types are prevalent in all animal and plant genomes. Among the size-expandable introns, the sizes of plant introns are expansion-limited in that the large introns exceeding 1000 bp are fewer in numbers and transposon-free as compared to the large introns among animals, where the larger introns are filled with transposable elements and appear expansion-flexible, reaching several kilobasepairs (kbp) and even thousands of kbp in size. Most of the intron parameters can be studied as signatures of the specific splicing machineries of different eukaryotic lineages and are highly relevant to the regulation of gene expression and functionality. In particular, the transcription-splicing-export coupling of eukaryotic intron dispensing leads to a working hypothesis that all intron parameters are evolved to be efficient and function-related in processing and routing the spliced transcripts.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berget S M, Moore C, Sharp P A. Spliced segments at the 5′ terminus of adenovirus 2 late mRNA. Proc Natl Acad Sci USA, 1977, 74: 3171–3175
Chow L T, Gelinas R E, Broker T R, et al. An amazing sequence arrangement at the 5′ ends of adenovirus 2 messenger RNA. Cell, 1977, 12: 1–8
Yu J, Wong G K S, Wang J, et al. Shotgun sequencing. In: Encyclopedia of Molecular Cell Biology and Molecular Medicine. 2nd ed. Wiley-VCH, 2005, 13: 71–114
Zhang Z, Wong G K S, Yu J. Protein coding. In: eLS. Chichester: John Wiley & Sons Ltd., 2013
Hong X, Scofield D G, Lynch M. Intron size, abundance, and distribution within untranslated regions of genes. Mol Biol Evol, 2006, 23: 2392–2404
Zhu J, He F, Wang D, et al. A novel role for minimal introns: routing mRNAs to the cytosol. PLoS ONE, 2010, 5: e10144
Wang J, Li S T, Zhang Y, et al. Vertebrate gene predictions and the problem of large genes. Nat Rev Genet, 2003, 4: 741–749
Yu J, Yang Z, Kibukawa M, et al. Minimal introns are not “junk”. Genome Res, 2002, 12: 1185–1189
Wong G K S, Passey D, Yu J. Most of the human genome is transcribed. Genome Res, 2001, 11: 1975–1977
Wong G K S, Passey D, Huang Y, et al. Is “junk” DNA mostly intron DNA? Genome Res, 2000, 10: 1672–1678
Wang D, Yu J. Both size and GC-content of minimal introns are selected in human population. PLoS ONE, 2011, 6: e17945
Wong G K S, Wang J, Passey D, et al. Codon-usage gradients in Gramineae genomes. Genome Res, 2002, 12: 851–856
Jamison S F, Crow A, Garcia-Blanco M A. The spliceosome assembly pathway in mammalian extracts. Mol Cell Bio, 1992, 12: 4279–4287
Pessa H K, Will C L, Meng X, et al. Minor spliceosome components are predominantly localized in the nucleus. Proc Nat Acad Sci USA, 2008, 105: 8655–8660
Shionyu M, Yamaguchi A, Shinoda K, et al. AS-ALPS: a database for analyzing the effects of alternative splicing on protein structure, interaction and network in human and mouse. Nucleic Acids Res, 2009, 37: D305–D309
Oliveros J C. VENNY. An interactive tool for comparing lists with Venn Diagrams. 2007. http://bioinfogp.cnb.csic.es/tools/venny/index.html
Wendel J F, Cronn R C, Alvarez I, et al. Intron size and genome size in plants. Mol Biol Evol, 2002, 19: 2346–2352
Hawkins J S, Kim H, Nason J D, et al. Differential lineage-specific amplification of transposable elements is responsible for genome size variation in Gossypium. Genome Res, 2006, 16: 1252–1261
Reddy A S. Alternative splicing of pre-messenger RNAs in plants in the genomic era. Ann Rev Plant Biol, 2007, 58: 267–294
Naeem H, Kalyna S, Marquez Y, et al. Alternative splicing in plants—coming of age. Trend Plant Sci, 2012, 17: 616–623
Nilsen T W. Trans-splicing of nematode premessenger RNA. Ann Rev Micro, 1993, 47: 413–440
Zemann A, Bekke A, Kiefmann M, et al. Evolution of small nucleolar RNAs in nematodes. Nucleic Acids Res, 2006, 34: 2676–2685
Kurek R, Reugels A M, Lammermann U, et al. Molecular aspects of intron evolution in dynein encoding mega-genes on the heterochromatic Y chromosome of Drosophila sp. Genetica, 2000, 109: 113–123
Reugels A M, Kurek R, Lammermann U, et al. Mega-introns in the dynein gene DhDhc7(Y) on the heterochromatic Y chromosome give rise to the giant threads loops in primary spermatocytes of Drosophila hydei. Genetics, 2000, 154: 759–769
Lopez A J. Alternative splicing of pre-mRNA: developmental consequences and mechanisms of regulation. Ann Rev Genet, 1998, 32: 279–305
Hatton A R, Subramaniam V, Lopez A J. Generation of alternative ultrabithorax isoforms and stepwise removal of large intron by resplicing at exon exon junctions. Mol Cell, 1998, 2: 787–796
Burnette J M, Miyamoto-Sato E, Schaub M A, et al. Subdivision of large introns in Drosophila by recursive splicing at nonexonic elements. Genetics, 2005, 170: 661–674
Yu J. Life on two tracks. Genomic Prot Bioinf, 2012, 10: 123–126
Khodor Y L, Menet J S, Tolan M, et al. Cotranscriptional splicing efficiency differs dramatically between Drosophila and mouse. RNA, 2012, 18: 2174–2186
Yu J, Hu S, Wang J, et al. A draft sequence assembly of the rice (Oryza sativa ssp. indica) genome. Science, 2002, 296: 79–93
Yu J, Wang J, Lin W, et al. The genomes of Oryza sativa: a history of duplications rice genomes. PLoS Biol, 2005, 3: e38
Wang J, Zhang J, Li R, et al. Evolutionary transients in the rice transcriptome. Genomic Prot Bioinf, 2010, 8: 223–228
Hong X, Scofield D G, Lynch M. Intron size, abundance, and distribution within untranslated regions of genes. Mol Biol Evol, 2006, 23: 2392–2404
Havlioglu N, Wang J, Fushimi K, et al. An intronic signal for alternative splicing in the human genome. PLoS ONE, 2007, 2: e1246
Cremer T, Cremer M, Dietzel S, et al. Chromosome territories—a functional nuclear landscape. Curr Opin Cell Biol, 2006, 18: 307–316
Darnell J E Jr. Reflections on the history of pre-mRNA processing and highlights of current knowledge: a unified picture. RNA, 2013, 19: 443–460
Caudron-Herger M, Rippe K. Nuclear architecture by RNA. Curr Opin Genet Dev, 2012, 22: 179–187
Yu J. Challenges to the common dogma. Genomic Prot Bioinf, 2012, 10: 55–57
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
JiaYan, W., JingFa, X., LingPing, W. et al. Systematic analysis of intron size and abundance parameters in diverse lineages. Sci. China Life Sci. 56, 968–974 (2013). https://doi.org/10.1007/s11427-013-4540-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4540-y