Abstract
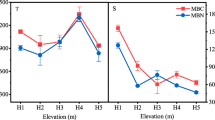
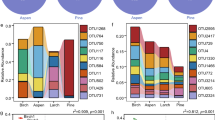
This study investigated the influence of broadleaf and conifer vegetation on soil microbial communities in a distinct vertical distribution belt in Northeast China. Soil samples were taken at 0–5, 5–10 and 10–20 cm depths from four vegetation types at different altitudes, which were characterized by poplar (Populus davidiana) (1250–1300 m), poplar (P. davidiana) mixed with birch (Betula platyphylla) (1370–1550 m), birch (B. platyphylla) (1550–1720 m), and larch (Larix principis-rupprechtii) (1840–1890 m). Microbial biomass and community structure were determined using the fumigation-extraction method and phospholipid fatty acid (PLFA) analysis, and soil fungal community level physiological profiles (CLPP) were characterized using Biolog FF Microplates. It was found that soil properties, especially soil organic carbon and water content, contributed significantly to the variations in soil microbes. With increasing soil depth, the soil microbial biomass, fungal biomass, and fungal catabolic ability diminished; however, the ratio of fungi to bacteria increased. The fungal ratio was higher under larch forests compared to that under poplar, birch, and their mixed forests, although the soil microbial biomass was lower. The direct contribution of vegetation types to the soil microbial community variation was 12%. If the indirect contribution through soil organic carbon was included, variations in the vegetation type had substantial influences on soil microbial composition and diversity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bardgett R D, Wardle D A. Aboveground-belowground Linkages: Biotic Interactions, Ecosystem Processes, and Global Change. New York: Oxford University Press, 2010
Zak D R, Holmes W E, White D C, et al. Plant diversity, soil microbial communities, and ecosystem function: are there any links? Ecology, 2003, 84: 2042–2050
Wagg C, Husband B C, Green D S, et al. Soil microbial communities from an elevational cline differ in their effect on conifer seedling growth. Plant Soil, 2011, 340: 491–504
Broughton L C, Gross K L. Patterns of diversity in plant and soil microbial communities along a productivity gradient in a Michigan old-field. Oecologia, 2000, 125: 420–427
Maly S, Korthals G W, Van Dijk C, et al. Effect of vegetation manipulation of abandoned arable land on soil microbial properties. Biol Fert Soils, 2000, 31: 121–127
Carney K M, Matson P A. Plant communities, soil microorganisms, and soil carbon cycling: does altering the world belowground matter to ecosystem functioning? Ecosystems, 2005, 8: 928–940
Eviner V T. Plant traits that influence ecosystem processes vary independently among species. Ecology, 2004, 85: 2215–2229
De Deyn G B, Cornelissen J H C, Bardgett R D. Plant functional traits and soil carbon sequestration in contrasting biomes. Ecol Lett, 2008, 11: 516–531
Hobbie S E, Ogdahl M, Chorover J, et al. Tree species effects on soil organic matter dynamics: the role of soil cation composition. Ecosystems, 2007, 10: 999–1018
Saetre P, Baath E. Spatial variation and patterns of soil microbial community structure in a mixed spruce-birch stand. Soil Biol Biochem, 2000, 32: 909–917
Tscherko D, Hammesfahr U, Zeltner G, et al. Plant succession and rhizosphere microbial communities in a recently deglaciated alpine terrain. Basic Appl Ecol, 2005, 6: 367–383
Merila P, Malmivaara-Lamsa M, Spetz P, et al. Soil organic matter quality as a link between microbial community structure and vegetation composition along a successional gradient in a boreal forest. Appl Soil Ecol, 2010, 46: 259–267
Aneja M K, Sharma S, Fleischmann F, et al. Microbial colonization of beech and spruce litter —influence of decomposition site and plant litter species on the diversity of microbial community. Microbial Ecol, 2006, 52: 127–135
Kazakou E, Vile D, Shipley B, et al. Co-variations in litter decomposition, leaf traits and plant growth in species from a Mediterranean old-field succession. Funct Ecol, 2006, 20: 21–30
Zak D R, Tilman D, Parmenter R R, et al. Plant-production and soil-microorganisms in late-successional ecosystems—a continental-scale study. Ecology, 1994, 75: 2333–2347
Mitchell R J, Hester A J, Campbell C D, et al. Explaining the variation in the soil microbial community: do vegetation composition and soil chemistry explain the same or different parts of the microbial variation? Plant Soil, 2012, 351: 355–362
Wardle D A, Yeates G W, Nicholson K S, et al. Response of soil microbial biomass dynamics, activity and plant litter decomposition to agricultural intensification over a seven-year period. Soil Biol Biochem, 1999, 31: 1707–1720
Bååth E, Anderson T H. Comparison of soil fungal/bacterial ratios in a pH gradient using physiological and PLFA-based techniques. Soil Biol Biochem, 2003, 35: 955–963
Fierer N, Strickland M S, Liptzin D, et al. Global patterns in belowground communities. Ecol Lett, 2009, 12: 1238–1249
Staddon W J, Trevors J T, Duchesne L C, et al. Soil microbial diversity and community structure across a climatic gradient in western Canada. Biodivers Conserv, 1998, 7: 1081–1092
Carletti P, Vendramin E, Pizzeghello D, et al. Soil humic compounds and microbial communities in six spruce forests as function of parent material, slope aspect and stand age. Plant Soil, 2009, 315: 47–65
Rogers B F, Tate R L. Temporal analysis of the soil microbial community along a toposequence in Pineland soils. Soil Biol Biochem, 2001, 33: 1389–1401
White C, Tardif J C, Adkins A, et al. Functional diversity of microbial communities in the mixed boreal plain forest of central Canada. Soil Biol Biochem, 2005, 37: 1359–1372
Merila P, Strommer R, Fritze H. Soil microbial activity and community structure along a primary succession transect on the land-uplift coast in western Finland. Soil Biol Biochem, 2002, 34: 1647–1654
Hogberg M N, Baath E, Nordgren A, et al. Contrasting effects of nitrogen availability on plant carbon supply to mycorrhizal fungi and saprotrophs—a hypothesis based on field observations in boreal forest. New Phytol, 2003, 160: 225–238
Frostegård A, Bååth E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol and Fert Soils, 1996, 22: 59–65
Ulery A L, Graham R C, Chadwick O A, et al. Decade-scale changes of soil carbon, nitrogen and exchangeable cations under chaparral and pine. Geoderma, 1995, 65: 121–134
Taylor D L, Herriott I C, Stone K E, et al. Structure and resilience of fungal communities in Alaskan boreal forest soils. Can J Forest Res, 2010, 40: 1288–1301
Vance E D, Brookes P C, Jenkinson D S. An extraction method for measuring soil microbial biomass C. Soil Biol Biochem, 1987, 19: 703–707
Wu J, Joergensen R G, Pommerening B, et al. Measurement of soil microbial biomass C by fumigation extraction—an automated procedure. Soil Biol Biochem, 1990, 22: 1167–1169
Brookes P C, Kragt J F, Powlson D S, et al. Chloroform fumigation and the release of soil-nitrogen—the effects of fumigation time and temperature. Soil Biol Biochem, 1985, 17: 831–835
Bligh E G, Dyer W J. Orange-red flesh in cod and haddock. J Fish Res Board Canada, 1959, 16: 449–452
Frostegård J, Haegerstrand A, Gidlund M, et al. Biologically modified LDL increases the adhesive properties of endothelial-cells. Atherosclerosis, 1991, 90: 119–126
Frostegård A, Bååth E, Tunlid A. Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty-acid analysis. Soil Biol Biochem, 1993, 25: 723–730
Frostegård A, Tunlid A, Bååth E. Phospholipid fatty-acid composition, biomass, and activity of microbial communities from 2 soil types experimentally exposed to different heavy-metals. Appl Environ Microb, 1993, 59: 3605–3617
Bååth E. The use of neutral lipid fatty acids to indicate the physiological conditions of soil fungi. Microbial Ecol, 2003, 45: 373–383
Singh M P. Application of Biolog Ff MicroPlate for substrate utilization and metabolite profiling of closely related fungi. J Microbiol Meth, 2009, 77: 102–108
Dixon P. Vegan, a package of R functions for community ecology. J Veg Sci, 2003, 14: 927–930
Borcard D, Legendre P, Drapeau P. Partialling out the spatial component of ecological variation. Ecology, 1992, 73: 1045–1055
Okland R H. Partitioning the variation in a plot-by-species data matrix that is related to n sets of explanatory variables. J Veg Sci, 2003, 14: 693–700
Manly B F J. Randomization, Bootstrap and Monte Carlo Methods in Biology. London: Chapman & Hall/CRC, 2006
Frostegård A, Tunlid A, Bååth E. Microbial biomass measured as total lipid phosphate in soils of different organic content. J Microbiol Meth, 1991, 14: 151–163
Saetre P. Spatial patterns of ground vegetation, soil microbial biomass and activity in a mixed spruce-birch stand. Ecography, 1999, 22: 183–192
Fontaine S, Mariotti A, Abbadie L. The priming effect of organic matter: a question of microbial competition? Soil Biol Biochem, 2003, 35: 837–843
Kramer C, Gleixner G. Soil organic matter in soil depth profiles: distinct carbon preferences of microbial groups during carbon transformation. Soil Biol Biochem, 2008, 40: 425–433
Joergensen R G, Wichern F. Quantitative assessment of the fungal contribution to microbial tissue in soil. Soil Biol Biochem, 2008, 40: 2977–2991
Bauhus J, Pare D, Cote L. Effects of tree species, stand age and soil type on soil microbial biomass and its activity in a southern boreal forest. Soil Biol Biochem, 1998, 30: 1077–1089
Ponge J F. Succession of fungi and fauna during decomposition of needles in a small area of Scots pine litter. Plant Soil, 1991, 138: 99–113
Quideau S A, Chadwick O A, Graham R C, et al. Base cation biogeochemistry and weathering under oak and pine: a controlled long-term experiment. Biogeochemistry, 1996, 35: 377–398
Wardle D A, Nilsson M C, Zackrisson O, et al. Determinants of litter mixing effects in a Swedish boreal forest. Soil Biol Biochem, 2003, 35: 827–835
Myers R T, Zak D R, White D C, et al. Landscape-level patterns of microbial community composition and substrate use in upland forest ecosystems. Soil Sci Soc Am J, 2001, 65: 359–367
Rey A, Pegoraro E, Jarvis P G. Carbon mineralization rates at different soil depths across a network of European forest sites (forcast). Eur J Soil Sci, 2008, 59: 1049–1062
Jiang Y M, Chen C R, Liu Y Q, et al. Soil soluble organic carbon and nitrogen pools under mono- and mixed species forest ecosystems in subtropical china. J Soil Sediment, 2010, 10: 1071–1081
Bailey V L, Smith J L, Bolton H. Fungal-to-bacterial ratios in soils investigated for enhanced C sequestration. Soil Biol Biochem, 2002, 34: 997–1007
Ingwersen J, Poll C, Streck T, et al. Micro-scale modelling of carbon turnover driven by microbial succession at a biogeochemical interface. Soil Biol Biochem, 2008, 40: 864–878
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Wang, M., Qu, L., Ma, K. et al. Soil microbial properties under different vegetation types on Mountain Han. Sci. China Life Sci. 56, 561–570 (2013). https://doi.org/10.1007/s11427-013-4486-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4486-0