Abstract
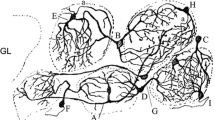
In recent years the evolution of olfactory bulb periglomerular cells, as well as the function of periglomerular cells in olfactory encoding, has attracted increasing attention. Studies of neural information encoding based on the analysis of simulation and modeling have given rise to electrophysiological models of periglomerular cells, which have an important role in the understanding of the biology of these cells. In this review we provide a brief introduction to the anatomy of the olfactory system and the cell types in the olfactory bulb. We elaborate on the latest progress in the study of the heterogeneity of periglomerular cells based on different classification criteria, such as molecular markers, structure, ion channels and action potentials. Then, we discuss the several existing electrophysiological models of periglomerular cells, and we highlight the problems and defects of these models. Finally, considering our present work, we propose a future direction for electrophysiological investigations of periglomerular cells and for the modeling of periglomerular cells and olfactory information encoding.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Laurent G. A systems perspective on early olfactory coding. Science, 1999, 286: 723–728
Puopolo M, Belluzzi O. Inhibitory synapses among interneurons in the glomerular layer of rat and frog olfactory bulbs. J Neurophysiol, 1998, 80: 344–349
Davison A P, Feng J F, Brown D. Spike synchronization in a biophysically-detailed model of the olfactory bulb. Neurocomputing, 2001, 38: 515–521
Laurent G. Olfactory network dynamics and the coding of multidimensional signals. Nat Rev Neurosci, 2002, 3: 884–895
Bazhenov M, Stopfer M, Rabinovich M, et al. Model of transient oscillatory synchronization in the locust antennal lobe. Neuron, 2001, 30: 553–567
Brody C D, Hopfield J J. Simple networks for spike-timing-based computation, with application to olfactory processing. Neuron, 2003, 37: 843–852
Linster C, Cleland T A. How spike synchronization among olfactory neurons can contribute to sensory discrimination. J Comput Neurosci, 2001, 10: 187–193
Cleland T A, Sethupathy P. Non-topographical contrast enhancement in the olfactory bulb. BMC Neurosci, 2006, 7: 7
Cleland T A, Linster C. Computation in the olfactory system. Chem Sens, 2005, 30: 801–813
Davison A P. Mathematical modelling of information processing in the olfactory bulb. Dissertation for Doctoral Degree. Cambridge: University of Cambridge, 2001
Kosaka K, Toida K, Aika Y, et al. How simple is the organization of the olfactory glomerulus? The heterogeneity of so-called periglomerular cells. Neurosci Res, 1998, 30: 101–110
Shipley M T, Ennis M. Functional organization of olfactory system. J Neurobiol, 1996, 30: 123–176
Engl N. Taste and smell in disease. J Med, 1983, 309: 1062–1063
Lledo P M, Didier A, Carleton A, et al. A dendrodendritic reciprocal synapse provides a recurrent excitatory connection in the olfactory bulb. Proc Natl Acad Sci USA, 2001, 98: 6441–6446
Martinez D P, Freeman W J. Periglomerular cell-action on mitral cells in olfactory-bulb shown by current source density analysis. Brain Res, 1984, 308: 223–233
Sakurai Y. Population coding by cell assemblies—What it really is in the brain. Neurosci Res, 1996, 26: 1–16
Averbeck B B, Lee D. Coding and transmission of information by neural ensembles. Trends Neurosci, 2004, 27: 225–230
Johnson D H, Gruner C M, Baggerly K, et al. Information-theoretic analysis of neural coding. J Comput Neurosci, 2001, 10: 47–69
Strong S P, Koberle R, van Steveninck R R D, et al. Entropy and information in neural spike trains. Phys Rev Lett, 1998, 80: 197–200
Pinching A J, Powell T P. The neuropil of the periglomerular region of the olfactory bulb. J Cell Sci, 1971, 9: 379–409
Ribak C E, Vaughn J E, Saito K, et al. Glutamate decarboxylase localization in neurons of the olfactory bulb. Brain Res, 1977, 126: 1–18
Toida K, Kosaka K, Heizmann C W, et al. Chemically defined neuron groups and their subpopulations in the glomerular layer of the rat main olfactory bulb: III. Structural features of calbindin D28K-immunoreactive neurons. J Comp Neurol, 1998, 392: 179–198
Toida K, Kosaka K, Aika Y, et al. Chemically defined neuron groups and their subpopulations in the glomerular layer of the rat main olfactory bulb—IV. Intraglomerular synapses of tyrosine hydroxylase-immunoreactive neurons. Neuroscience, 2000, 101: 11–17
Alonso J R, Brinon J G, Martinez-Guijarro F J, et al. Coexpression of neurocalcin with other calcium-binding proteins in the rat main olfactory bulb. J Comp Neurol, 1999, 407: 404–414
Kosaka K, Kosaka T. Synaptic organization of the glomerulus in the main olfactory bulb: compartments of the glomerulus and heterogeneity of the periglomerular cells. Anat Sci Int, 2005, 80: 80–90
Puopolo M, Belluzzi O. Functional heterogeneity of periglomerular cells in the rat olfactory bulb. Europ J Neurosci, 1998, 10: 1073–1083
Puopolo M, Bean B P, Raviola E. Spontaneous activity of isolated dopaminergic periglomerular cells of the main olfactory bulb. J Neurophysiol, 2005, 94: 3618–3627
Pignatelli A, Kobayashi K, Okano H, et al. Functional properties of dopaminergic neurones in the mouse olfactory bulb. J Physiol, 2005, 564: 501–514
McQuiston A R, Katz L C. Electrophysiology of interneurons in the glomerular layer of the rat olfactory bulb. J Neurophysiol, 2001, 86: 1899–1907
Bardoni R, Magherini P C, Belluzzi O. Sodium current in periglomerular cells of frog olfactory bulb in vitro. Brain Res, 1995, 703: 19–25
Puopolo M, Belluzzi O. Sodium current in periglomerular cells of rat olfactory bulb in vitro. Neuroreport, 1996, 7: 1846–1850
Bufler J, Zufall F, Franke C, et al. Patch-clamp recordings of spiking and nonspiking interneurons from rabbit olfactory-bulb slices—membrane-properties and ionic currents. J Comp Physiol—Sens Neural Behav Physiol, 1992, 170: 145–152
Arruda D, Publio R, Roque A C. Reduced compartmental model of the periglomerular cell of the mammalian olfactory bulb. In: The 18th Annual Computational Neuroscience Meeting, Berlin, Germany, 2009
Davison A P, Feng J F, Brown D. A reduced compartmental model of the mitral cell for use in network models of the olfactory bulb. Brain Res Bull, 2000, 51: 393–399
Cadetti L, Belluzzi O. Hyperpolarisation-activated current in glomerular cells of the rat olfactory bulb. Neuroreport, 2001, 12: 3117–3120
Arruda D. The periglomerular cell of the olfactory bulb and its role in the odor processing: a computational model. Dissertation for Master’s Degree. Ribeirao Preto: University of Sao Paulo, 2010
Ye X, Liang B, Ying N, et al. The function of periglomerular cells on olfactory coding in a detailed electrophysiological model of vertebrate olfactory bulb. In: IEEE 5th International Conference on the Bio-Inspired Computing: Theories and Applications, Changsha, China, 2010
Migliore M, Cook E P, Jaffe D B, et al. Computer simulations of morphologically reconstructed CA3 hippocampal neurons. J Neurophysiol, 1995, 73: 1157–1168
Destexhe A, Contreras D, Steriade M, et al. In vivo, in vitro, and computational analysis of dendritic calcium currents in thalamic reticular neurons. J Neurosci, 1996, 16: 169–185
Destexhe A, Neubig M, Ulrich D, et al. Dendritic low-threshold calcium currents in thalamic relay cells. J Neurosci, 1998, 18: 3574–3588
Midtgaard J, Pinato G. Dendritic sodium spikelets and low-threshold calcium spikes in turtle olfactory bulb granule cells. J Neurophysiol, 2005, 93: 1285–1294
Pinato G, Midtgaard J. Regulation of granule cell excitability by a low-threshold calcium spike in turtle olfactory bulb. J Neurophysiol, 2003, 90: 3341–3351
Branco T, Clark B A, Hausser M. Dendritic discrimination of temporal input sequences in cortical neurons. Science, 2010, 329: 1671–1675
Destexhe A, Contreras D. Neuronal computations with stochastic network states. Science, 2006, 314: 85–90
Jones I L, Livi P, Lewandowska M K, et al. The potential of microelectrode arrays and microelectronics for biomedical research and diagnostics. Anal Bioanal Chem, 2011, 399: 2313–2329
Ling S, Gao T, Liu J, et al. The fabrication of an olfactory receptor neuron chip based on planar multi-electrode array and its odor-response analysis. Biosens Bioelectron, 2010, 26: 1124–1128
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Ying, N., Tian, J., Yu, S. et al. Progress in defining heterogeneity and modeling periglomerular cells in the olfactory bulb. Sci. China Life Sci. 55, 567–575 (2012). https://doi.org/10.1007/s11427-012-4346-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4346-3