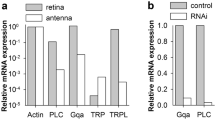
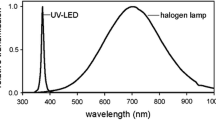
Abstract
The Drosophila visual transduction is the fastest known G protein-coupled signaling cascade and has been served as a model for understanding the molecular mechanisms of other G protein-coupled signaling cascades. Numbers of components in visual transduction machinery have been identified. Based on the functional characterization of these genes, a model for Drosophila phototransduction has been outlined, including rhodopsin activation, phosphoinoside signaling, and the opening of TRP and TRPL channels. Recently, the characterization of mutants, showing slow termination, revealed the physiological significance and the mechanism of rapid termination of light response.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pak W L, Grossfield J, White N V. Nonphototactic mutants in a study of vision of Drosophila. Nature, 1969, 222: 351–354
Ranganathan R, Harris G L, Stevens C F, et al. A Drosophila mutant defective in extracellular calcium-dependent photoreceptor deactivation and rapid desensitization. Nature, 1991, 354: 230–232
Bloomquist B T, Shortridge R D, Schneuwly S, et al. Isolation of a putative phospholipase C gene of Drosophila, norpA, and its role in phototransduction. Cell, 1988, 54: 723–733
Montell C, Rubin G M. Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction. Neuron, 1989, 2:1313–1323
Phillips A M, Bull A, Kelly L E. Identification of a Drosophila gene encoding a calmodulin-binding protein with homology to the trp phototransduction gene. Neuron, 1992. 8: 631–642
Hardie R C, Minke B. The trp gene is essential for a light-activated Ca2+ channel in Drosophila photoreceptors. Neuron, 1992, 8:643–651
Niemeyer B A, Suzuki E, Scott K, et al. The Drosophila light-activated conductance is composed of the two channels TRP and TRPL. Cell, 1996, 85:651–659
Fu Y, Yau K W. Phototransduction in mouse rods and cones. Pflugers Arch, 2007, 454: 805–819
Berson D M. Phototransduction in ganglion-cell photoreceptors. Pflugers Arch, 2007, 454: 849–855
Qiu X, Kumbalasiri T, Carlson S M, et al. Induction of photosensitivity by heterologous expression of melanopsin. Nature, 2005, 433: 745–749
Provencio I, Jiang G, De Grip W J, et al. Melanopsin: An opsin in melanophores, brain, and eye. Proc Natl Acad Sci USA, 1998, 95: 340–345
Provencio I, Rodriguez I R, Jiang G, et al. A novel human opsin in the inner retina. J Neurosci, 2000, 20: 600–605
Ruby N F, Brennan T J, Xie X, et al. Role of melanopsin in circadian responses to light. Science, 2002, 298: 2211–2213
Panda S, Sato T K, Castrucci A M, et al. Melanopsin (Opn4) requirement for normal light-induced circadian phase shifting. Science, 2002, 298: 2213–2216
Hattar S, Liao H W, Takao M, et al. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science, 2002, 295: 1065–1070
Hannibal J, Hindersson P, Knudsen S M, et al. The photopigment melanopsin is exclusively present in pituitary adenylate cyclase-activating polypeptide-containing retinal ganglion cells of the retinohypothalamic tract. J Neurosci, 2002, 22: RC191.
Panda S, Nayak S K, Campo B, et al. Illumination of the melanopsin signaling pathway. Science, 2005, 307: 600–604
Isoldi M C, Rollag M D, Castrucci A M, et al. Rhabdomeric phototransduction initiated by the vertebrate photopigment melanopsin. Proc Natl Acad Sci USA, 2005, 102: 1217–1221
Ready D F, Hanson T E, Benzer S. Development of the Drosophila retina, a neurocrystalline lattice. Dev Biol, 1976, 53: 217–240
O’Tousa J E, Baehr W, Martin R L, et al. The Drosophila ninaE gene encodes an opsin. Cell, 1985, 40: 839–850
Zuker C S. Cowman A F, Rubin G M. Isolation and structure of a rhodopsin gene from D. melanogaster. Cell, 1985, 40: 851–858
Fryxell K J, Meyerowitz E M. An opsin gene that is expressed only in the R7 photoreceptor cell of Drosophila. EMBO J, 1987, 6: 443–451
Zuker C S, Montell C, Jones K, et al. A rhodopsin gene expressed in photoreceptor cell R7 of the Drosophila eye: homologies with other signal-transducing molecules. J Neurosci, 1987, 7: 1550–1557
Montell C, Jones K, Zuker C, et al. A second opsin gene expressed in the ultraviolet-sensitive R7 photoreceptor cells of Drosophila melanogaster. J Neurosci, 1987. 7: 1558–1566
Fortini M E, Rubin G M. Analysis of cis-acting requirements of the Rh3 and Rh4 genes reveals a bipartite organization to rhodopsin promoters in Drosophila melanogaster. Genes Dev, 1990, 4: 444–463
Ting C Y, Lee C H. Visual circuit development in Drosophila. Curr Opin Neurobiol, 2007, 17: 65–72
Choe K M, Clandinin T R. Thinking about visual behavior; learning about photoreceptor function. Curr Top Dev Biol, 2005, 69: 187–213
Hotta Y, Benzer S. Genetic dissection of the Drosophila nervous system by means of mosaics. Proc Natl Acad Sci USA, 1970, 67: 1156–1163
Pak W L, Grossfield J, Arnold K S. Mutants of the visual pathway of Drosophila melanogaster. Nature, 1970, 227: 518–520
Harris WA, Stark WS. Hereditary retinal degeneration in Drosophila melanogaster. A mutant defect associated with the phototransduction process. J Gen Physiol, 1977, 69: 261–291
Steele F, O’Tousa J E. Rhodopsin activation causes retinal degeneration in Drosophila rdgC mutant. Neuron, 1990, 4: 883–890
Hotta Y, Benzer S. Abnormal electroretinograms in visual mutants of Drosophila. Nature, 1969, 222: 354–356
Cosens D J, Manning A. Abnormal electroretinogram from a Drosophila mutant. Nature, 1969, 224: 285–287
Hengstenberg R, Gotz K G. Effect of facet-separating pigments on the perception of light and contrast in eye mutants of Drosophila. Kybernetik, 1967, 3: 276–285
Pak WL, Leung H T. Genetic approaches to visual transduction in Drosophila melanogaster. Recep Chann, 2003, 9: 149–167
Han J, Gong P, Reddig K, Mitra M, Guo P, Li HS. et al. The fly CAMTA transcription factor potentiates deactivation of rhodopsin, a G protein-coupled light receptor. Cell, 2006. 127: 847–858
Dolph P, Nair A, Raghu P. Electroretinogram recordings of Drosophila. Cold Spring Harb Protoc, 2011: pdb.prot5549. doi: 10.1101/pdb.prot5549..
Dolph P J, Ranganathan R, Colley N J, et al. Arrestin function in inactivation of G protein-coupled receptor rhodopsin in vivo. Science, 1993, 260: 1910–1916
Xu H, Lee S J, Suzuki E, et al. A lysosomal tetraspanin associated with retinal degeneration identified via a genome-wide screen. Embo J, 2004, 23: 811–822
Garcia-Murillas I, Pettitt T, Macdonald E, et al. lazaro encodes a lipid phosphate phosphohydrolase that regulates phosphatidylinositol turnover during Drosophila phototransduction. Neuron, 2006, 49: 533–546
Kwon Y, Montell C. Dependence on the Lazaro phosphatidic acid phosphatase for the maximum light response. Curr Biol, 2006, 16: 723–729
Goldsmith T H, Marks B C, Bernard G D. Separation and identification of geometric isomers of 3-hydroxyretinoids and occurrence in the eyes of insects. Vision Res, 1986, 26: 1763–1769
Kiselev A, Subramaniam S. Activation and regeneration of rhodopsin in the insect visual cycle. Science, 1994, 266: 1369–1373
Emeis D, Kühn H, Reichert J, et al. Complex formation between metarhodopsin II and GTP-binding protein in bovine photoreceptor membranes leads to a shift of the photoproduct equilibrium. FEBS Lett, 1982, 143: 29–34
Kibelbek J, Mitchell D C, Beach J M, et al. Functional equivalence of metarhodopsin II and the Gt-activating form of photolyzed bovine rhodopsin. Biochemistry, 1991, 30: 6761–6768
Palczewski K, Saari J C. Activation and inactivation steps in the visual transduction pathway. Curr Opin Neurobiol, 1997, 7: 500–504
Yau K W, Baylor D A. Cyclic GMP-activated conductance of retinal photoreceptor cells. Annu Rev Neurosci, 1989, 12: 289–327
Devary O, Heichal O, Blumenfeld A, et al. Coupling of photoexcited rhodopsin to inositol phospholipid hydrolysis in fly photoreceptors. Proc Natl Acad Sci USA, 1987, 84: 6939–6943
Lee Y J, Shah S, Suzuki E, et al. The Drosophila dgq gene encodes a G alpha protein that mediates phototransduction. Neuron, 1994, 13: 1143–1157
Scott K, Becker A, Sun Y, et al. Gq alpha protein function in vivo: genetic dissection of its role in photoreceptor cell physiology. Neuron, 1995, 15: 919–927
Han J, Reddig K, Li H S. Prolonged G(q) activity triggers fly rhodopsin endocytosis and degradation, and reduces photoreceptor sensitivity. Embo J, 2007, 26: 4966–4973
Satoh A K, Ready D F. Arrestin1 mediates light-dependent rhodopsin endocytosis and cell survival. Curr Biol, 2005, 15: 1722–1733
Ranganathan R, Stevens C F. Arrestin binding determines the rate of inactivation of the G protein-coupled receptor rhodopsin in vivo. Cell, 1995, 81: 841–848
Toyoshima S, Matsumoto N, Wang P, et al. Purification and partial amino acid sequences of phosphoinositide-specific phospholipase C of Drosophila eye. J Biol Chem, 1990, 265: 14842–14848
Smith D P, Shieh B H, Zuker C S. Isolation and structure of an arrestin gene from Drosophila. Proc Natl Acad Sci USA, 1990, 87: 1003–1007
Hyde D R, Mecklenburg K L, Pollock J A, et al. Twenty Drosophila visual system cDNA clones: one is a homolog of human arrestin. Proc Natl Acad Sci USA, 1990, 87: 1008–1012
Scott K, Zuker C. Lights out: deactivation of the phototransduction cascade. Trends Biochem Sci, 1997, 22: 350–354
Vinós J, Jalink K, Hardy R W, et al. A G protein-coupled receptor phosphatase required for rhodopsin function. Science, 1997, 277: 687–690
Montell C. PLC fills a GAP in G-protein-coupled signalling. Nat Cell Biol, 2000, 2: E82–83
Cook B, Bar-Yaacov M, Cohen Ben-Ami H, et al. Phospholipase C and termination of G-protein-mediated signalling in vivo. Nat Cell Biol, 2000, 2: 296–301
Huber A, Sander P, Bähner M, et al. The TRP Ca2+ channel assembled in a signaling complex by the PDZ domain protein INAD is phosphorylated through the interaction with protein kinase C (ePKC). FEBS Lett, 1998, 425: 317–322
Smith D P, Ranganathan R, Hardy R W, et al. Photoreceptor deactivation and retinal degeneration mediated by a photoreceptor-specific protein kinase C. Science, 1991, 254: 1478–1484
Schaeffer E, Smith D, Mardon G, et al. Isolation and characterization of two new Drosophila protein kinase C genes, including one specifically expressed in photoreceptor cells. Cell, 1989, 57: 403–412
Huber A, Sander P, Paulsen R. Phosphorylation of the InaD gene product, a photoreceptor membrane protein required for recovery of visual excitation. J Biol Chem, 1996, 271: 11710–11717
Chevesich J, Kreuz A J, Montell C. Requirement for the PDZ domain protein, INAD, for localization of the trp store-operated channel to a signaling complex. Neuron, 1997, 18: 95–105
Warr C G, Kelly L E. Identification and characterization of two distinct calmodulin-binding sites in the Trpl ion-channel protein of Drosophila melanogaster. Biochem J, 1996, 314 ( Pt 2): 497–503
Scott K, Sun Y, Beckingham K, et al. Calmodulin regulation of Drosophila light-activated channels and receptor function mediates termination of the light response in vivo. Cell, 1997. 91: 375–383
Ranganathan R, Harris G L, Stevens C F, et al. A Drosophila mutant defective in extracellular calcium-dependent photoreceptor deactivation and rapid desensitization. Nature, 1991, 354: 230–232
Hardie R C, Minke B. Calcium-dependent inactivation of light-sensitive channels in Drosophila photoreceptors. J Gen Physiol, 1994, 103: 409–427
Hardie R C. whole-cell recordings of the light induced current in dissociated Drosophila photoreceptors: Evidence for feedback by calcium permeating the light-sensitive channels. Proc Royal Soc London. Ser B: Biol Sci, 1991, 245: 203–210
Wang T, Xu H, Oberwinkler J, et al. Light activation, adaptation, and cell survival functions of the Na+/Ca2+ exchanger CalX. Neuron, 2005, 45: 367–378
Merrill, C.E., et al., Visual arrestins in olfactory pathways of Drosophila and the malaria vector mosquito Anopheles gambiae. Proc Natl Acad Sci USA, 2002, 99: 1633–1638
Lee S J, Xu H, Kang L W, et al. Light adaptation through phosphoinositide-regulated translocation of Drosophila visual arrestin. Neuron, 2003, 39: 121–132
Lee S J, Montell C. Suppression of constant-light-induced blindness but not retinal degeneration by inhibition of the rhodopsin degradation pathway. Curr Biol, 2004, 14: 2076–2085
Wang T, Xu H, Oberwinkler J, et al. Light activation, adaptation, and cell survival functions of the Na+/Ca2+exchanger CalX. Neuron, 2005. 45(3): p. 367–78
Scott K, Sun Y, Beckingham K, et al. Calmodulin regulation of Drosophila light-activated channels and receptor function mediates termination of the light response in vivo. Cell, 1997, 91: 375–383
Wu L, Niemeyer B, Colley N, et al. Regulation of PLC-mediated signalling in vivo by CDP-diacylglycerol synthase. Nature, 1995, 373: 216–222
Rosenbaum E E, Hardie R C, Colley N J. Calnexin is essential for rhodopsin maturation, Ca2+ regulation, and photoreceptor cell survival. Neuron, 2006, 49: 229–241
Yuan Q, Lin F, Zheng X, et al. Serotonin modulates circadian entrainment in Drosophila. Neuron, 2005, 47: 115–127
Hardie R C, Martin F, Cochrane G W, et al. Molecular basis of amplification in Drosophila phototransduction: roles for G protein, phospholipase C, and diacylglycerol kinase. Neuron, 2002, 36: 689–701
Dolph P J, Man-Son-Hing H, Yarfitz S, et al. An eye-specific G beta subunit essential for termination of the phototransduction cascade. Nature, 1994, 370: 59–61
Elia N, Frechter S, Gedi Y, et al. Excess of Gbetae over Gqalphae in vivo prevents dark, spontaneous activity of Drosophila photoreceptors. J Cell Biol, 2005, 171:517–526
Schillo S, Belusic G, Hartmann K, et al. Targeted mutagenesis of the farnesylation site of Drosophila Ggammae disrupts membrane association of the G protein betagamma complex and affects the light sensitivity of the visual system. J Biol Chem, 2004, 279: 36309–36316
Lee SJ, Xu H, Montell C. Rhodopsin kinase activity modulates the amplitude of the visual response in Drosophila. Proc Natl Acad Sci USA, 2004, 101: 11874–11879
Smith D P, Ranganathan R, Hardy R W, et al. Photoreceptor deactivation and retinal degeneration mediated by a photoreceptor-specific protein kinase C. Science, 1991, 254: 1478–1484
Shieh B H, Niemeyer B. A novel protein encoded by the InaD gene regulates recovery of visual transduction in Drosophila. Neuron, 1995, 14: 201–210
Scott K, Zuker C S. Assembly of the Drosophila phototransduction cascade into a signalling complex shapes elementary responses. Nature, 1998, 395: 805–808
Tsunoda S, Sierralta J, Sun Y, et al. A multivalent PDZ-domain protein assembles signalling complexes in a G-protein-coupled cascade. Nature, 1997, 388: 243–249
Mishra P, Socolich M, Wall M A, et al. Dynamic scaffolding in a G protein-coupled signaling system. Cell, 2007, 131: 80–92
Li C, Geng C, Leung H T, et al. INAF, a protein required for transient receptor potential Ca(2+) channel function. Proc Natl Acad Sci USA, 1999, 96: 13474–13479
Stamnes M A, Shieh B H, Chuman L, et al. The cyclophilin homolog ninaA is a tissue-specific integral membrane protein required for the proper synthesis of a subset of Drosophila rhodopsins. Cell, 1991, 65: 219–227
Gu G, Yang J, Mitchell K A, et al. Drosophila ninaB and ninaD act outside of retina to produce rhodopsin chromophore. J Biol Chem, 2004, 279: 18608–18613
Wes P D, Xu X Z, Li H S, et al. Termination of phototransduction requires binding of the NINAC myosin III and the PDZ protein INAD. Nat Neurosci, 1999, 2: 447–453
Porter J A, Montell C. Distinct roles of the Drosophila ninaC kinase and myosin domains revealed by systematic mutagenesis. J Cell Biol, 1993, 122: 601–612
Hofstee C A, Henderson S, Hardie R C, et al. Differential effects of ninaC proteins (p132 and p174) on light-activated currents and pupil mechanism in Drosophila photoreceptors. Vis Neurosci, 1996, 13: 897–906
Wang T, Montell C. Rhodopsin formation in Drosophila is dependent on the PINTA retinoid-binding protein. J Neurosci, 2005, 25: 5187–5194
Scavarda N J, O’Tousa J, Pak W L. Drosophila locus with gene-dosage effects on rhodopsin. Proc Natl Acad Sci USA, 1983, 80: 4441–4445
Iakhine R, Chorna-Ornan I, Zars T, et al. Novel dominant rhodopsin mutation triggers two mechanisms of retinal degeneration and photoreceptor desensitization. J Neurosci, 2004, 24: 2516–2526
Bentrop J, Schwab K, Pak W L, et al. Site-directed mutagenesis of highly conserved amino acids in the first cytoplasmic loop of Drosophila Rh1 opsin blocks rhodopsin synthesis in the nascent state. EMBO J, 1997, 16: 1600–1609
Sarfare S, Ahmad S T, Joyce M V, et al. The Drosophila ninaG oxidoreductase acts in visual pigment chromophore production. J Biol Chem, 2005, 280: 11895–11901
Kim S, Chen D M, Zavarella K,. et al. Substitution of a non-retinal phospholipase C in Drosophila phototransduction. Insect Mol Biol, 2003, 12: 147–153
LaLonde M M, Janssens H, Rosenbaum E, et al. Regulation of phototransduction responsiveness and retinal degeneration by a phospholipase D-generated signaling lipid. J Cell Biol, 2005, 169: 471–479
Matsumoto H, Kurien B T, Takagi Y, et al. Phosrestin I undergoes the earliest light-induced phosphorylation by a calcium/calmodulin-dependent protein kinase in Drosophila photoreceptors. Neuron, 1994, 12: 997–1010
O’Tousa J E, Leonard D S, Pak W L. Morphological defects in oraJK84 photoreceptors caused by mutation in R1-6 opsin gene of Drosophila. J Neurogenet, 1989, 6: 41–52
Rajaram S, Scott R L, Nash H A. Retrograde signaling from the brain to the retina modulates the termination of the light response in Drosophila. Proc Natl Acad Sci USA, 2005, 102: 17840–17845
Wang T, Montell C. A phosphoinositide synthase required for a sustained light response. J Neurosci, 2006, 26: 12816–12825
Zelhof A C, Koundakjian E, Scully A L, et al. Mutation of the photoreceptor specific homeodomain gene Pph13 results in defects in phototransduction and rhabdomere morphogenesis. Development, 2003, 130: 4383–4392
Johnson M A, Frayer K L, Stark W S. Characteristics of RDGA: Mutants with retinal degeneration in Drosophila. J Insect Physiol, 1982, 28: 233–242
Milligan S C, Alb J G Jr, Elagina R B, et al. The phosphatidylinositol transfer protein domain of Drosophila retinal degeneration B protein is essential for photoreceptor cell survival and recovery from light stimulation. J Cell Biol, 1997, 139: 351–363
Xiong W C, Okano H, Patel N H, et al. repo encodes a glial-specific homeo domain protein required in the Drosophila nervous system. Genes Dev, 1994, 8: 981–994
Venkatachalam K, Wasserman D, Wang X, et al. Dependence on a retinophilin/myosin complex for stability of PKC and INAD and termination of phototransduction. J Neurosci, 2010, 30: 11337–11345
Wang T, Jiao Y, Montell C. Dissection of the pathway required for generation of vitamin A and for Drosophila phototransduction. J Cell Biol, 2007, 177: 305–316
Huang F D, Matthies H J, Speese S D, et al. Rolling blackout, a newly identified PIP2-DAG pathway lipase required for Drosophila phototransduction. Nat Neurosci, 2004,7: 1070–1078
Benzer S. Behavioral mutants of Drosophila isolated by countercurrent distribution. Proc Natl Acad Sci USA, 1967, 58: 1112–1119
Lindsley D L, Zimm G G. The Genome of Drosophila melanogaster. viii ed. 1992. 1133
Leung H T, Geng C, Pak W L. Phenotypes of trpl mutants and interactions between the transient receptor potential (TRP) and TRP-like channels in Drosophila. J Neurosci, 2000, 20: 6797–6803
Popescu D C, Ham A J, Shieh B H. Scaffolding protein INAD regulates deactivation of vision by promoting phosphorylation of transient receptor potential by eye protein kinase C in Drosophila. J Neurosci, 2006, 26: 8570–8577
Leung H T, Tseng-Crank J, Kim E, et al. DAG lipase activity is necessary for TRP channel regulation in Drosophila photoreceptors. Neuron, 2008, 58: 884–896
Yoon J, Ben-Ami H C, Hong Y S, et al. Novel mechanism of massive photoreceptor degeneration caused by mutations in the trp gene of Drosophila. J Neurosci, 2000, 20: 649–659
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Tian, Y., Hu, W., Tong, H. et al. Phototransduction in Drosophila. Sci. China Life Sci. 55, 27–34 (2012). https://doi.org/10.1007/s11427-012-4272-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4272-4