Abstract
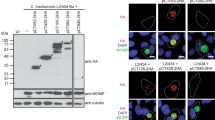
The inclusion membrane proteins play potentially important roles in chlamydial biology and pathogenesis. Here we localized and characterized the hypothetical protein CT440 in Chlamydia trachomatis-infected cells. The open reading frame (ORF) encoding the CT440 protein from the C. trachomatis serovar D genome was cloned into the prokaryotic expression vector pGEX-6p and expressed as a glutathione-S-transferase (GST) fusion protein in E. coli XL1-Blue. The CT440 fusion protein was used to immunize mice to raise antigen-specific antibody. After verification by Western blot and immunofluorescence assay (IFA), the specific antibody was used to localize the endogenous CT440 protein and to detect its expression pattern in Chlamydia-infected cells. Cytosolic expression of CT440 in HeLa cells was also carried out to evaluate the effect of the CT440 protein on the subsequent chlamydial infection. The results showed that the hypothetical protein CT440 was localized in the C. trachomatis inclusion membrane, and was detectable 12 h after chlamydial infection. Expression of CT440 in the cytoplasm did not inhibit the subsequent chlamydial infection. In summary, we have identified a new inclusion membrane protein that may be an important candidate for understanding C. trachomatis pathogenesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mabey D C, Solomon A W, Foster A. Trachoma. Lancet, 2003, 362: 223–229
Brunham R C, Rey-Ladino J. Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nat Rev Immunol, 2005, 5: 149–161
Belland R, Ojcius D M, Byrne G I. Chlamydia. Nat Rev Microbiol, 2004, 2: 530–531
Kilmarx P H, Mock P A, Levine W C. Effect of Chlamydia trachomatis coinfection on HIV shedding in genital tract secretions. Sex Transm Dis, 2001, 28: 347–348
Mbizvo E M, Msuya S E, Stray-Pedersen B, et al. HIV seroprevalence and its associations with the other reproductive tract infections in asymptomatic women in Harare, Zimbabwe. Int J STD AIDS, 2001, 12: 524–531
Smith J S, Muñoz N, Herrero R, et al. Evidence for Chlamydia trachomatis as a human papillomavirus cofactor in the etiology of invasive cervical cancer in Brazil and the Philippines. J Infect Dis, 2002, 185: 324–331
Wallin K L, Wiklund F, Luostarinen T, et al. A population-based prospective study of Chlamydia trachomatis infection and cervical carcinoma. Int J Cancer, 2002, 101: 371–374
Hackstadt T. The diverse habitats of obligate intracellular parasites. Curr Opin Microbiol, 1998, 1: 82–87
Hackstadt T, Fischer E R, Scidmore M A, et al. Origins and functions of the chlamydial inclusion. Trends Microbiol, 1997, 5: 288–293
Abdelrahman Y M, Belland R J. The chlamydial developmental cycle. FEMS Microbiol Rev, 2005, 29: 949–959
Carabeo R A, Mead D J, Hackstadt T. Golgi-dependent transport of cholesterol to the Chlamydia trachomatis inclusion. Proc Natl Acad Sci USA, 2003, 100: 6771–6776
Su H, McClarty G, Dong F, et al. Activation of Raf/MEK/ERK/cPLA2 signaling pathway is essential for chlamydial acquisition of host glycerophospholipids. J Biol Chem, 2004, 279: 9409–9416
Zhong G. Chlamydia trachomatis secretion of proteases for manipulating host signaling pathways. Front Microbiol, 2011, 2: 14
Li Z, Chen D, Zhong Y, et al. The chlamydial plasmid-encoded protein pgp3 is secreted into the cytosol of Chlamydia-infected cells. Infect Immun, 2008, 76: 3415–3428
Lei L, Qi M, Budrys N, et al. Localization of Chlamydia trachomatis hypothetical protein CT311 in host cell cytoplasm. Microb Pathog, 2011, 51: 101–109
Qi M, Gong S, Lei L, et al. A Chlamydia trachomatis OmcB C-terminal fragment is released into the host cell cytoplasm and is immunogenic in humans. Infect Immun, 2011, 79: 2193–2203
Li Z, Wu Y, Huang Q, et al. Localization and characterization of hypothetical protein CT358 in the Chlamydia trachomatis-infected cells. Prog Biochem Biophys, 2009, 36: 549–554
Li Z, Zhong Y, Lei L, et al. Antibodies from women urogenitally infected with C. trachomatis predominantly recognized the plasmid protein pgp3 in a conformation-dependent manner. BMC Microbiol, 2008, 8: 90
Xiao Y, Zhong Y, Su H, et al. NF-kappa B activation is not required for Chlamydia trachomatis inhibition of host epithelial cell apoptosis. J Immunol, 2005, 174: 1701–1708
Li Z, Chen C, Chen D, et al. Characterization of fifty putative inclusion membrane proteins encoded in the Chlamydia trachomatis genome. Infect Immun, 2008, 76: 2746–2757
Bannantine J P, Griffiths R S, Viratyosin W, et al. A secondary structure motif predictive of protein localization to the chlamydial inclusion membrane. Cell Microbiol, 2000, 2: 35–47
Rockey D D, Scidmore M A, Bannantine J P, et al. Proteins in the chlamydial inclusion membrane. Microbes Infect, 2002, 4: 333–340
Fling S P, Sutherland R A, Steele L N, et al. CD8+ T cells recognize an inclusion membrane-associated protein from the vacuolar pathogen Chlamydia trachomatis. Proc Natl Acad Sci USA, 2001, 98: 1160–1165
Toh H, Miura K, Shirai M, et al. In silico inference of inclusion membrane protein family in obligate intracellular parasites chlamydiae. DNA Res, 2003, 10: 9–17
Alzhanov D, Barnes J, Hruby D E, et al. Chlamydial development is blocked in host cells transfected with Chlamydophila caviae incA. BMC Microbiol, 2004, 4: 24
Delevoye C, Nilges M, Dautry-Varsat A, et al. Conservation of the biochemical properties of IncA from Chlamydia trachomatis and Chlamydia caviae: oligomerization of IncA mediates interaction between facing membranes. J Biol Chem, 2004, 279: 46896–46906
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Li, Z., Huang, Q., Su, S. et al. Localization and characterization of the hypothetical protein CT440 in Chlamydia trachomatis-infected cells. Sci. China Life Sci. 54, 1048–1054 (2011). https://doi.org/10.1007/s11427-011-4243-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4243-1