Abstract
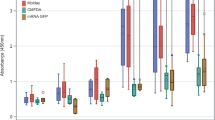
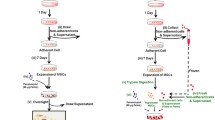
Recently, transplantation of allogeneic and autologous cells has been used for regenerative medicine. A critical issue is monitoring migration and homing of transplanted cells, as well as engraftment efficiency and functional capability in vivo. Monitoring of superparamagnetic iron oxide (SPIO) particles by magnetic resonance imaging (MRI) has been used in animal models and clinical settings to track labeled cells. A major limitation of MRI is that the signals do not show biological characteristics of transplanted cells in vivo. Bone marrow mesenchymal stem cells (MSCs) have been extensively investigated for their various therapeutic properties, and exhibit the potential to differentiate into cells of diverse lineages. In this study, cynomolgus monkey MSCs (cMSCs) were labeled with Molday ION Rhodamine-B™ (MIRB), a new SPIO agent, to investigate and characterize the biophysical and MRI properties of labeled cMSCs in vitro and in vivo. The results indicate that MIRB is biocompatible and useful for cMSCs labeling and cell tracking by multimodality imaging. Our method is helpful for detection of transplanted stem cells in vivo, which is required for understanding mechanisms of cell therapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Caplan A I. Review: mesenchymal stem cells: cell-based reconstructive therapy in orthopedics. Tissue Eng, 2005, 11: 1198–1211
Dyson S C, Barker R A. Cell-based therapies for Parkinson’s disease. Expert Rev Neurother, 2011, 11: 831–844
Raikwar S P, Zavazava N. Spontaneous in vivo differentiation of embryonic stem cell-derived pancreatic endoderm-like cells corrects hyperglycemia in diabetic mice. Transplantation, 2011, 91: 11–20
Bussolati B, Camussi G. Adult stem cells and renal repair. J Nephrol, 2006, 19: 706–709
Daldrup-Link H E, Rudelius M, Piontek G, et al. Migration of iron oxide-labeled human hematopoietic progenitor cells in a mouse model: in vivo monitoring with 1.5-T MR imaging equipment. Radiology, 2005, 234: 197–205
Budde M D, Frank J A. Magnetic tagging of therapeutic cells for MRI. J Null Med, 2009, 50: 171–174
Adler E D, Bystrup A, Briley-Saebo K C, et al. In vivo detection of embryonic stem cell-derived cardiovascular progenitor cells using Cy3-labeled GadofluorineM in murine myocardium. JACC Cardiovasc Imag, 2009, 2: 1114–1122
Guzman R, Uchida N, Bliss TM, et al. Long-term monitoring of transplanted human neural stem cells in developmental and pathological contexts with MRI. Proc Natl Acad Sci USA, 2007, 104: 10211–10216
Higuchi T, Anton M, Dumler K, et al. Combined reporter gene PET and iron oxide MRI for monitoring survival and localization of transplanted cells in the rat heart. J Nucl Med, 2009, 50: 1088–1094
Chang N K, Jeong Y Y, Park J S, et al. Tracking of neural stem cells in rats with intracerebral hemorrhage by the use of 3T MRI. Korean J Radiol, 2008, 9: 196–204
Onda T, Honmou O, Harada K, et al. Therapeutic benefits by human mesenchymal stem cells (hMSCs) and Ang-1 gene-modified hMSCs after cerebral ischemia. J Cereb Blood Flow Metab, 2008, 28: 329–340
Cai J, Zhang X, Wang X, et al. In vivo MR imaging of magnetically labeled mesenchymal stem cells transplanted into rat liver through hepatic arterial injection. Contrast Media Mol Imag, 2008, 3: 61–66
Kim D, Chun B G, Kim Y K, et al. In vivo tracking of human mesenchymal stem cells in experimental stroke. Cell Transplant, 2008, 16: 1007–1012
Lu C W, Hung Y, Hsiao J K, et al. Bifunctional magnetic silica nanoparticles for highly efficient human stem cell labeling. Nano Lett, 2007, 7: 149–154
Mulder W J, Koole R, Brandwijk R J, et al. Quantum dots with a paramagnetic coating as a bimodal molecular imaging probe. Nano Lett, 2006, 6: 1–6
Sung C K, Hong K A, Lin S, et al. Dual-modal nanoprobes for imaging of mesenchymal stem cell transplant by MRI and fluorescence imaging. Korean J Radiol, 2009, 10: 613–622
Addicott B, Willman M, Rodriguez J, et al. Mesenchymal stem cell labeling and in vitro MR characterization at 1.5 T of new SPIO contrast agent: Molday ION Rhodamine-B™. Contrast Media Mol Imaging, 2011, 6: 7–18
Emborg M E, Ebert A D, Moirano J, et al. GDNF-secreting human neural progenitor cells increase tyrosine hydroxylase and VMAT2 expression in MPTP-treated cynomolgus monkeys. Cell Transplant, 2008, 17: 383–395
Kim S H, Lee W J, Lim H K, et al. SPIO-enhanced MRI findings of well-differentiated hepatocellular carcinomas: correlation with MDCT findings. Korean J Radiol, 2009, 10: 112–120
Satija N K, Singh V K, Verma Y K, et al. Mesenchymal stem cell-based therapy: a new paradigm in regenerative medicine. J Cell Mol Med, 2009, 13: 4385–402
Wagner J, Kean T, Young R, et al. Optimizing mesenchymal stem cell-based therapeutics. Curr Opin Biotechnol, 2009, 20: 531–536
Summer R, Fine A. Mesenchymal progenitor cell research: limitations and recommendations. Proc Am Thorac Soc, 2008, 5: 707–710
Lee J W, Fang X, Krasnodembskaya A, et al. Concise review: mesenchymal stem cells for acute lung injury: Role of paracrine soluble factors. Stem Cells, 2011, 29: 913–919
Bussolati B, Tetta C, Camussi G. Contribution of stem cells to kidney repair. Am J Nephrol, 2008, 28: 813–822
Fattahi H, Laurent S, Liu F, et al. Magnetoliposomes as multimodal contrast agents for molecular imaging and cancer nanotheragnostics. Nanomedicine (Lond), 2011, 6: 529–544
Fan C, Gao W, Chen Z, et al. Tumor selectivity of stealth multifunctionalized superparamagnetic iron oxide nanoparticles. Int J Pharm, 2011, 404: 180–190
Bulte J W. In vivo MRI cell tracking: clinical studies. AJR Am J Roentgenol, 2009, 193: 314–325
Kedziorek D A, Kraitchman D L. Superparamagnetic iron oxide labeling of stem cells for MRI tracking and delivery in cardiovascular disease. Methods Mol Biol, 2010, 660: 171–183
Wagner J, Kean T, Young R, et al. Optimizing mesenchymal stem cell-based therapeutics. Curr Opin Biotechnol, 2009, 20: 531–536
Karussis D, Karageorgiou C, Vaknin-Dembinsky A, et al. Safety and immunological effects of mesenchymal stem cell transplantation in patients with multiple sclerosis and amyotrophic lateral sclerosis. Arch Neurol, 2010, 67: 1187–1194
Addicott B, Willman M, Rodriguez J, et al. Mesenchymal stem cell labeling and in vitro MR characterization at 1.5 T of new SPIO contrast agent: Molday ION Rhodamine-B™. Contrast Media Mol Imaging, 2011, 6: 7–18
Frank J A, Miller B R, Arbab A S, et al. Clinically applicable labeling of mammalian and stem cells by combining superparamagnetic iron oxides and transfection agents. Radiology, 2003, 228: 480–487
Song M, Moon W K, Kim Y, et al. Labeling efficacy of superparamagnetic iron oxide nanoparticles to human neural stem cells: comparison of ferumoxides, monocrystalline iron oxide, cross-linked iron oxide (CLIO)-NH2 and tat-CLIO. Korean J Radiol, 2007, 8: 365–371
Hill J M, Dick A J, Raman V K, et al. Serial cardiac magnetic resonance imaging of injected mesenchymal stem cells. Circulation, 2003, 108: 1009–1014
Chang J S, Chang K L, Hwang D F, et al. In vitro cytotoxicitiy of silica nanoparticles at high concentrations strongly depends on the metabolic activity type of the cell line. Environ Sci Technol, 2007, 41: 2064–2068
Huang H, Xie Q, Kang M, et al. Labeling transplanted mice islet with polyvinylpyrrolidone coated superparamagnetic iron oxide nanoparticles for in vivo detection by magnetic resonance imaging. Nanotechnology, 2009, 20: 365101
So P W, Kalber T, Hunt D, et al. Efficient and rapid labeling of transplanted cell populations with superparamagnetic iron oxide nanoparticles using cell surface chemical biotinylation for in vivo monitoring by MRI. Cell Transplant, 2010, 19: 419–429
Mainenti P P, Mancini M, Mainolfi C, et al. Detection of colo-rectal liver metastases: prospective comparison of contrast enhanced US, multidetector CT, PET/CT, and 1.5 Tesla MR with extracellular and reticulo-endothelial cell specific contrast agents. Abdom Imaging, 2010, 35: 511–521
Yoon T J, Yu K N, Kim E, et al. Specific targeting, cell sorting, and bioimaging with smart magnetic silica core-shell nanomaterials. Small, 2006, 2: 209–215
Lu C W, Hung Y, Hsiao J K, et al. Bifunctional magnetic silica nanoparticles for highly efficient human stem cell labeling. Nano Lett, 2007, 7: 149–154
Veiseh O, Sun C, Gunn J, et al. Optical and MRI multifunctional nanoprobe for targeting gliomas. Nano Lett, 2005, 5: 1003–1008
Kim J S, Yoon T J, Kim H K, et al. Sentinel lymph node mapping of the stomach using fluorescent magnetic nanoparticles in rabbits. Korean J Gastroenterol, 2008, 51: 19–24
Weissleder R. A clearer vision for in vivo imaging. Nat Biotechnol, 2001, 19: 316–317
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ren, Z., Wang, J., Zou, C. et al. Labeling of cynomolgus monkey bone marrow-derived mesenchymal stem cells for cell tracking by multimodality imaging. Sci. China Life Sci. 54, 981–987 (2011). https://doi.org/10.1007/s11427-011-4239-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4239-x