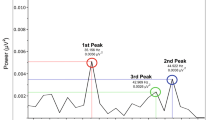
Abstract
Baclofen is a selective gamma-aminobutyric acid (GABA) type B agonist that may have important medicinal uses, such as in analgesics and drug addiction treatment. In addition, evidence is accumulating that suggests GABAergic-mediated neurotransmission is altered during aging. This study investigated whether baclofen administration (5 mg kg−1) induces differential effects on cortical electrical activity with age. Electroencephalograms (EEGs) were recorded from young (3–4 months) and aged (15–17 months) rats, and both the absolute and relative powers in five frequency bands (delta: 2–4 Hz; theta: 4–8 Hz; alpha: 8–12 Hz; beta: 12–20 Hz; gamma: 20–100 Hz) were analyzed. Before administration of baclofen, we found that the EEG relative power in the beta band was higher in the aged than that in the young rats. After administration of baclofen, there was a slower increase in the relative power in the delta band in the aged than that in the young rats. Moreover, there was no significant difference between the two age groups in absolute power in any frequency band. These findings indicate that baclofen treatment appears to differentially modify cortical EEG activity as a function of age. Our data further elucidate the relationship between GABAB receptor-mediated neurotransmission and aging.
Article PDF
Similar content being viewed by others
References
Leventhal A G, Wang Y, Pu M, et al. GABA and its agonists improved visual cortical function in senescent monkeys. Science, 2003, 300: 812–815 12730605, 10.1126/science.1082874, 1:CAS:528:DC%2BD3sXjtlSktL0%3D
Gutierrez A, Khan Z U, Morris S J, et al. Age-related decrease of GABAA receptor subunits and glutamic acid decarboxylase in the rat inferior colliculus. J Neurosci, 1994, 14: 7469–7477 7996188, 1:CAS:528:DyaK2MXis1emuro%3D
Turgeon S M, Albin R L. GABAB binding sites in early adult and aging rat brain. Neurobiol Aging, 1994, 15: 705–711 7891825, 10.1016/0197-4580(94)90052-3, 1:CAS:528:DyaK2MXis1Crurs%3D
Mhatre M C, Ticku M K. Aging related alterations in GABAA receptor subunit mRNA levels in Fischer rats. Brain Res Mol Brain Res, 1992, 14: 71–78 1323020, 10.1016/0169-328X(92)90012-Z, 1:CAS:528:DyaK38XkvVeqtr0%3D
Milbrandt J C, Albin R L, Caspary D M. Age-related decrease in GABAB receptor binding in the Fischer 344 rat inferior colliculus. Neurobiol Aging, 1994, 15: 699–703 7891824, 10.1016/0197-4580(94)90051-5, 1:CAS:528:DyaK2MXis1Cruro%3D
Fu Y, Guo L, Zhang J, et al. Differential effects of ageing on the EEG during pentobarbital and ketamine anaesthesia. Eur J Anaesthesiol, 2008, 25:826–833 18570701, 10.1017/S0265021508004687, 1:CAS:528:DC%2BD1cXht1CjsLjP
Him A, Johnston A R, Yau J L, et al. Tonic activity and GABA responsiveness of medial vestibular nucleus neurons in aged rats. Neuroreport, 2001, 12: 3965–3968 11742220, 10.1097/00001756-200112210-00022, 1:CAS:528:DC%2BD38Xjsl2ltQ%3D%3D
Brioni J D. Role of GABA during the multiple consolidation of memory. Drug Develop Res, 1993, 28: 3–27 10.1002/ddr.430280103, 1:CAS:528:DyaK3sXhtFKrt7s%3D
Chu D C, Penney J B, Young A B. Quantitative autoradiography of hippocampal GABAB and GABAA receptor changes in Alzheimer’s disease. Neurosci Lett, 1987, 82: 246–252 2827074, 10.1016/0304-3940(87)90264-3, 1:CAS:528:DyaL1cXltlOltg%3D%3D
Chu D C, Penney J B, Young A B. Cortical GABAB and GABAA receptors in Alzheimer’s disease: a quantitative autoradiographic study. Neurology, 1987, 37: 1454–1459 2819782, 1:STN:280:DyaL2szhsFynsA%3D%3D
Colombo G, Agabio R, Carai M A, et al. Ability of baclofen in reducing alcohol intake and withdrawal severity: I—Preclinical evidence. Alcohol Clin Exp Res, 2000, 24: 58–66 10656194, 1:CAS:528:DC%2BD3cXhtVWgt78%3D
Ebenezer I S. The effect of intracerebroventricular administration of baclofen on food intake in rats. Neuroreport, 1990, 1: 73–76 1966600, 10.1097/00001756-199009000-00020, 1:CAS:528:DyaK38Xlt1Cgs7Y%3D
Ebenezer I S, Pringle A K. The effect of systemic administration of baclofen on food intake in rats. Neuropharmacology, 1992, 31: 39–42 1311808, 10.1016/0028-3908(92)90158-L, 1:CAS:528:DyaK38XpvF2jtw%3D%3D
Braida D, Ottonello F, Sala M. Eptastigmine restores the aged rat’s normal cortical spectral power pattern. Pharmacol Res, 2000, 42: 495–500 11023715, 10.1006/phrs.2000.0732, 1:CAS:528:DC%2BD3cXntFWqtro%3D
Sebban C, Tesolin B, Coulomb B, et al. Comparative effects of almitrine and raubasine, singly and in combination, on electroencephalographic activity in young and old rats. Exp Gerontol, 1989, 24: 11–24 2707311, 10.1016/0531-5565(89)90031-4, 1:CAS:528:DyaL1MXovVGiug%3D%3D
Schultz A, Grouven U, Zander I, et al. Age-related effects in the EEG during propofol anaesthesia. Acta Anaesthesiol Scand, 2004, 48: 27–34 14674970, 10.1111/j.1399-6576.2004.00258.x, 1:CAS:528:DC%2BD2cXhsVGns7Y%3D
Paxinos, G. and Watson, C. The rat brain in stereotaxic coordinates. San Diego (CA): Academic Press. 2005
Liu N, Liu Y, Fan Y, et al. EEG activities in the orbitofrontal cortex and dorsolateral prefrontal cortex during the development of morphine dependence, tolerance and withdrawal in rhesus monkeys. Brain Res, 2005, 1053: 137–145 16054602, 10.1016/j.brainres.2005.06.037, 1:CAS:528:DC%2BD2MXnslyisrg%3D
Sun N, Li Y, Tian S, et al. Dynamic changes in orbitofrontal neuronal activity in rats during opiate administration and withdrawal. Neuroscience, 2006, 138: 77–82 16377092, 10.1016/j.neuroscience.2005.10.034, 1:CAS:528:DC%2BD28XhsF2nsLo%3D
Kishitake M, Yamanouchi K. Facilitatory effects of WAY-100635, a 5-HT1A receptor antagonist, on lordosis in female rats. Neurosci Lett, 2004, 371: 147–151 15519746, 10.1016/j.neulet.2004.08.058, 1:CAS:528:DC%2BD2cXptFOrsr0%3D
Clarke A R, Barry R J, McCarthy R, et al. Age and sex effects in the EEG: differences in two subtypes of attention-deficit/hyperactivity disorder. Clin Neurophysiol, 2001, 112: 815–826 11336897, 10.1016/S1388-2457(01)00487-4, 1:STN:280:DC%2BD3MzntFSktw%3D%3D
Gasser T, Verleger R, Bacher P, et al. Development of the EEG of school-age children and adolescents. I. Analysis of band power. Electroencephalogr Clin Neurophysiol, 1988, 69: 91–99 2446839, 10.1016/0013-4694(88)90204-0, 1:STN:280:DyaL1c%2Fot1artw%3D%3D
Benninger C, Matthis P, Scheffner D. EEG development of healthy boys and girls. Results of a longitudinal study. Electroencephalogr Clin Neurophysiol, 1984, 57: 1–12 6198138, 10.1016/0013-4694(84)90002-6, 1:STN:280:DyaL2c7gvVWktQ%3D%3D
John E R, Ahn H, Prichep L, et al. Developmental equations for the electroencephalogram. Science, 1980, 210: 1255–1258 7434026, 10.1126/science.7434026, 1:STN:280:DyaL3M%2Fls1ChtA%3D%3D
Duffy F H, McAnulty G B, Albert M S. The pattern of age-related differences in electrophysiological activity of healthy males and females. Neurobiol Aging, 1993, 14: 73–84 8450936, 10.1016/0197-4580(93)90025-7, 1:STN:280:DyaK3s7ps1Oltg%3D%3D
Knott V J, Harr A. Assessing the topographic EEG changes associated with aging and acute/long-term effects of smoking. Neuropsychobiology, 1996, 33: 210–222 8840345, 10.1159/000119279, 1:STN:280:DyaK28vjslOgtA%3D%3D
Pfurtscheller G, Stancak A Jr, Neuper C. Event-related synchronization (ERS) in the alpha band—an electrophysiological correlate of cortical idling: a review. Int J Psychophysiol, 1996, 24: 39–46 8978434, 10.1016/S0167-8760(96)00066-9, 1:STN:280:DyaK2s7jtlSktw%3D%3D
Babiloni C, Frisoni G, Steriade M, et al. Frontal white matter volume and delta EEG sources negatively correlate in awake subjects with mild cognitive impairment and Alzheimer’s disease. Clin Neurophysiol, 2006, 117: 1113–1129 16564740, 10.1016/j.clinph.2006.01.020
Penolazzi B, Spironelli C, Angrilli A. Delta EEG activity as a marker of dysfunctional linguistic processing in developmental dyslexia. Psychophysiology, 2008, 45: 1025–1033 18803600, 10.1111/j.1469-8986.2008.00709.x
Spironelli C, Angrilli A. EEG delta band as a marker of brain damage in aphasic patients after recovery of language. Neuropsychologia, 2009, 47: 988–994 19027029, 10.1016/j.neuropsychologia.2008.10.019
Dossi R C, Nunez A, Steriade M. Electrophysiology of a slow (0.5–4 Hz) intrinsic oscillation of cat thalamocortical neurones in vivo. J Physiol, 1992, 447: 215–234 1593448, 1:STN:280:DyaK383nslersg%3D%3D
Steriade M, Dossi R C, Nunez A. Network modulation of a slow intrinsic oscillation of cat thalamocortical neurons implicated in sleep delta waves: cortically induced synchronization and brainstem cholinergic suppression. J Neurosci, 1991, 11: 3200–3217 1941080, 1:STN:280:DyaK38%2FksVOlug%3D%3D
Billard J M, Lamour Y, Dutar P. Decreased monosynaptic GABAB-mediated inhibitory postsynaptic potentials in hippocampal CA1 pyramidal cells in the aged rat: pharmacological characterization and possible mechanisms. J Neurophysiol, 1995, 74: 539–546 7472361, 1:CAS:528:DyaK2MXosVChtLo%3D
Caspary D M, Raza A, Lawhorn Armour B A, et al. Immunocytochemical and neurochemical evidence for age-related loss of GABA in the inferior colliculus: implications for neural presbycusis. J Neurosci, 1990, 10: 2363–2372 1973948, 1:CAS:528:DyaK3cXlsVGktLo%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Fu, Y., Cui, J. & Ma, Y. Differential effects of aging on EEG after baclofen administration. Sci. China Life Sci. 54, 459–465 (2011). https://doi.org/10.1007/s11427-011-4170-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4170-1