Abstract
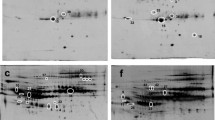
The efficacy of interferon (IFN) is limited in about 1/3 of patients with chronic hepatitis B (CHB). We used two-dimensional electrophoresis (2-DE)-based proteomic strategies to identify potential serum markers predicting hepatitis B e antigen (HBeAg) seroconversion in these patients during IFN therapy. Two groups of patients were enrolled: training and validation. In the training group, 2-DE experiments and subsequent identification of altered levels of proteins showed that α-2-HS-glycoprotein, leucine-rich α-2-glycoprotein, and haptoglobin were significantly upregulated as compared with baseline levels in the HBeAg seroconversion group, whereas apolipoprotein C-III precursor, leucine-rich α-2-glycoprotein, and α-albumin were downregulated in the non-seroconversion group. For patients with HBeAg seroconversion in the training group, Western blot analyses showed that α-2-HS-glycoprotein levels in 75% of patients were significantly upregulated at the end of the treatment as compared with baseline levels. Subsequent experiments in the validation group showed that α-2-HS-glycoprotein levels were significantly increased at week 4 in 83.33% of patients in the HBeAg seroconversion group. Dynamic changes in the serum level of α-2-HS-glycoprotein may be a potential early marker for predicting HBeAg seroconversion during IFN treatment for CHB.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lok A S, McMahon B J. Chronic hepatitis B. Hepatology, 2007, 45: 507–539, 1:CAS:528:DC%2BD2sXisVKns7o%3D, 10.1002/hep.21513, 17256718
Liaw Y F, Tai D I, Chu C M, et al. The development of cirrhosis in patients with chronic type B hepatitis: A prospective study. Hepatology, 1988, 8: 493–496, 1:STN:280:DyaL1c3is1CmtQ%3D%3D, 10.1002/hep.1840080310, 3371868
De Jongh F E, Janssen H L, De Man R A, et al. Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology, 1992, 103: 1630–1635, 1426884
Fattovich G, Giustina G, Schalm S W, et al. Occurrence of hepatocellular carcinoma and decompensation in Western European patients with cirrhosis type B. Hepatology, 1995, 21: 77–82, 1:STN:280:DyaK2M7gsVOrsw%3D%3D, 7806171
Samuel C E. Antiviral actions of interferons. Clin Microbiol Rev, 2001, 14: 778–809, 1:CAS:528:DC%2BD3MXot1Wms7o%3D, 10.1128/CMR.14.4.778-809.2001, 11585785
Levv D E, Garcia-Sastre A. The virus battles: IFN induction of the antiviral state and mechanisms of viral evasion. Cytokine Growth Factor Rev, 2001, 12: 143–156, 10.1016/S1359-6101(00)00027-7
Biron C A. Interferons-a and -b as imune regulators: A new look. Immunity, 2001, 14: 661–664, 1:CAS:528:DC%2BD3MXkvFSltr0%3D, 10.1016/S1074-7613(01)00154-6, 11420036
Grander D, Sangfelt O, Erickson S. How does interferon exert its cell growth inhibitory effect? Eur J Haematol, 1997, 59: 129–135, 1:CAS:528:DyaK2sXmtlCktrc%3D, 10.1111/j.1600-0609.1997.tb00965.x, 9310119
Wong D K, Cheung A M, O’Rourke K, et al. Effect of alphainterferon in patients with hepatitis B e antigen-positive chronic hepatitis B: A meta-analysis. Ann Intern Med, 1993, 119: 312–323, 1:STN:280:DyaK3szhvFWqsQ%3D%3D, 8328741
Janssen H L, van Zonneveld M, Senturk H, et al. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: A randomised trial. Lancet, 2005, 365: 123–129, 1:CAS:528:DC%2BD2MXhvF2gsQ%3D%3D, 10.1016/S0140-6736(05)17701-0, 15639293
Perrillo R P, Schiff E R, Davis G L, et al. A randomized, controlled trial of interferon alfa-2b alone and after prednisone withdrawal for the treatment of chronic hepatitis B. The Hepatitis Interventional Therapy Group. N Engl J Med, 1990, 323: 295–301, 1:STN:280:DyaK3czgvF2huw%3D%3D, 10.1056/NEJM199008023230503, 2195346
Lok A S, Wu P C, Lai C L, et al. A controlled trial of interferon with or without prednisone priming for chronic hepatitis B. Gastroenterology, 1992, 102: 2091–2097, 1:STN:280:DyaK383msleitg%3D%3D, 1587429
Feng J T, Shang S, Beretta L. Proteomics for the early detection and treatment of hepatocellular carcinoma. Oncogene, 2006, 25: 3810–3817, 1:CAS:528:DC%2BD28XmtFCntLw%3D, 10.1038/sj.onc.1209551, 16799622
Poon T C, Hui A Y, Chan H L, et al. Prediction of liver fibrosis and cirrhosis in chronic hepatitis B infection by serum proteomic fingerprinting: A pilot study. Clin Chem, 2005, 51: 328–335, 1:CAS:528:DC%2BD2MXhtVGhu7g%3D, 10.1373/clinchem.2004.041764, 15590748
Low T Y, Leow C K, Salto-Tellez M, et al. A proteomic analysis of thioacetamide-induced hepatotoxicity and cirrhosis in rat livers. Proteomics, 2004, 4: 3960–3974, 1:CAS:528:DC%2BD2cXhtFarsb%2FN, 10.1002/pmic.200400852, 15526343
He Q Y, Lau G K, Zhou Y, et al. Serum biomarkers of hepatitis B virus infected liver inflammation: A proteomic study. Proteomics, 2003, 3: 666–674, 1:CAS:528:DC%2BD3sXjvFGisLg%3D, 10.1002/pmic.200300394, 12748946
Parent R, Beretta L. Proteomics in the study of liver pathology. J Hepatol, 2005, 43: 177–183, 1:CAS:528:DC%2BD2MXlt1GhsL4%3D, 10.1016/j.jhep.2005.04.001, 15894398
Lv S, Wei L, Wang J H, et al. Identification of novel molecular candidates for acute liver failure in plasma of BALB/c murine model. J Proteome Res, 2007, 6: 2746–2752, 1:CAS:528:DC%2BD2sXmsVKmtrY%3D, 10.1021/pr0701759, 17569552
Li C, Tan Y X, Zhou H, et al. Proteomic analysis of hepatitis B virus-associated hepatocellular carcinoma: Identification of potential tumor markers. Proteomics, 2005, 5: 1125–1139, 10.1002/pmic.200401141, 15759316
Kim W, Oe L S, Kim J S, et al. Comparison of proteome between hepatitis B virus- and hepatitis C virus-associated hepatocellular carcinoma. Clin Cancer Res, 2003, 9: 5493–5500, 1:CAS:528:DC%2BD3sXpsVejtLo%3D, 14654528
Hoofnagle J H, Schafritz D A, Popper H. Chronic type B hepatitis and the “healthy” HBsAg carrier state. Hepatology, 1987, 7: 758–763, 1:STN:280:DyaL2s3nsl2rtQ%3D%3D, 10.1002/hep.1840070424, 3301618
Hoofnagle J H. Chronic type B hepatitis. Gastroenterology, 1983, 84: 422–424, 1:STN:280:DyaL3s%2FotVegug%3D%3D, 6848416
Chu C M, Karayiannis P, Fowler M J, et al. Natural history of chronic hepatitis B virus infection in Taiwan: Studies of hepatitis B virus DNA in serum. Hepatology, 1985, 5: 431–434, 1:STN:280:DyaL2M3gsFOrtg%3D%3D, 10.1002/hep.1840050315, 3997072
Srinivas P R, Wagner A S, Reddy L V, et al. Serum α-2-HSglycoprotein is an inhibitor of the human insulin receptor at the tyrosine kinase level. Mol Endocrinol, 1993, 7: 1445–1455, 1:CAS:528:DyaK2cXhvVGqtbg%3D, 10.1210/me.7.11.1445, 7906861
Kalabay L, Chavin K, Lebreton J P, et al. Human recombinant α-2-HS glycoprotein is produced in insect cells as a full-length inhibitor of the insulin receptor tyrosine kinase. Horm Metab Res, 1998, 30: 1–6, 1:CAS:528:DyaK1cXnvF2iug%3D%3D, 10.1055/s-2007-978822, 9503031
Demetriou M, Binkert C, Sukhu B, et al. Fetuin/alpha2-HS glycoprotein is a transforming growth factor-β type II receptor mimic and cytokine antagonist. J Biol Chem, 1996, 271: 12755–12761, 1:CAS:528:DyaK28XjtlGjtL4%3D, 10.1074/jbc.271.22.12755, 8662721
Ohnishi T, Nakamura O, Arakaki N, et al. Effect of phosphorylated rat fetuin on the growth of hepatocytes in primary cultures in the presence of human hepatocyte-growth factor. Evidence that phosphorylated fetuin is a natural modulator of hepatocyte-growth factor. Eur J Biochem, 1997, 243: 753–761, 1:CAS:528:DyaK2sXht1Cjtrk%3D, 10.1111/j.1432-1033.1997.00753.x, 9057842
Kalabay L, Jakab L, Prohászka Z, et al. Human fetuin/alpha-2-HS-glycoprotein level as a novel indicator of liver cell function and short-term mortality in patients with liver cirrhosis and liver cancer. Eur J Gastroenterol Hepatol, 2002, 14: 389–394, 10.1097/00042737-200204000-00009, 11943951
Kalabay L, Gráf L, Vörös K, et al. Human serum fetuin A/alpha2HSglycoprotein level is associated with long-term survival in patients with alcoholic liver cirrhosis, comparison with the Child-Pugh and MELD scores. BMC Gastroenterol, 2007, 7: 15, 10.1186/1471-230X-7-15, 17394649
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Ma, H., Wang, J., Guo, F. et al. α-2-HS-glycoprotein is a potential marker predicting hepatitis B e antigen seroconversion in patients with chronic hepatitis B during treatment with pegylated interferon alfa-2b. Sci. China Life Sci. 54, 39–47 (2011). https://doi.org/10.1007/s11427-010-4111-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4111-4