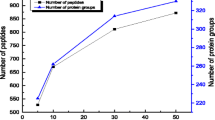
Abstract
In proteomics, attention has focused on various immobilized enzyme reactors (IMERs) for the realization of high throughput digestion. In this report, a novel organic-inorganic hybrid monolith based IMER was prepared in a 100 μm i.d. capillary with 3-glycidoxypropyltrimethoxysilane (GLYMO) as the monomer and tetraethoxysilane (TEOS) as the crosslinker. Trypsin immobilization was achieved via the reaction between vicinal diol groups, which were obtained from hydrolysis of epoxy groups, and the amino groups of trypsin. Bovine serum albumin was digested thoroughly by this IMER in 47 s. After micro-reverse phase liquid chromatography-tandem mass spectrometry (μRPLC-MS/MS) analysis and database searching, beyond 35% sequence coverage was obtained, and the result was comparable to that of 12 h in solution digestion. The present IMER has potential for high throughput digestion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aaron A K, Michael J M. Effects of modified digestion schemes on the identification of proteins from complex mixtures. J Proteome Res, 2006, 5: 695–700, 10.1021/pr050315j
Hervey, Strader M B, Hurst G B. Comparison of digestion protocols for microgram quantities of enriched protein samples. J Proteome Res, 2007, 6: 3054–3061, 1:CAS:528:DC%2BD2sXns1Ohtbs%3D, 10.1021/pr070159b, 17616116
Byrne R E, Scanu A M. Soluble and immobilized trypsin as structural probes of human plasma high-density lipoproteins: Enzyme properties and kinetics of proteolysis. Biochemistry, 1983, 22: 2894–2903, 1:CAS:528:DyaL3sXitFais78%3D, 10.1021/bi00281a018, 6871169
Li Y, Yan B, Deng C H, et al. Efficient on-chip proteolysis system based on functionalized magnetic silica microspheres. Proteomics, 2007, 7: 2330–2339, 1:CAS:528:DC%2BD2sXos1GhtLw%3D, 10.1002/pmic.200700112, 17570518
Li Y, Xu X Q, Deng C H, et al. Immobilization of trypsin on superparamagnetic nanoparticles for rapid and effective proteolysis. J Proteome Res, 2007, 6: 3849–3855, 1:CAS:528:DC%2BD2sXosVOqt7k%3D, 10.1021/pr070132s, 17676785
Li Y, Yan B, Deng C H, et al. On-plate digestion of proteins using novel trypsin-immobilized magnetic nanospheres for MALDI-TOF-MS analysis. Proteomics, 2007, 7: 3661–3671, 1:CAS:528:DC%2BD2sXhtlShsbrP, 10.1002/pmic.200700464, 17853514
Cooper G W, Chen J Z, Li Y, et al. Membrane-based nanoscale proteolytic reactor enabling protein digestion, peptide separation, and protein identification using mass spectrometry. Anal Chem, 2003, 75: 1067–1074, 1:CAS:528:DC%2BD3sXlsFemuw%3D%3D, 10.1021/ac025768b, 12641224
Slysz G W, Lewis D F, Schriemer D C. Detection and identification of sub-nanogram levels of protein in a nanoLC-trypsin-MS system. J Proteome Res, 2006, 5: 1959–1966, 1:CAS:528:DC%2BD28XmvF2mt78%3D, 10.1021/pr060142d, 16889418
Calleri E, Temporini C, Perani E, et al. Trypsin-based monolithic bioreactor coupled on-line with LC/MS/MS system for protein digestion and variant identification in standard solutions and serum samples. J Proteome Res, 2005, 4: 481–490, 1:CAS:528:DC%2BD2MXhsl2qtro%3D, 10.1021/pr049796h, 15822925
Sakai-Kato K, Kato M, Toyooka T. On-line trypsin-encapsulated enzyme reactor by the sol-gel method integrated into capillary electrophoresis. Anal Chem, 2002, 74: 2943–2949, 1:CAS:528:DC%2BD38XjvVajtLc%3D, 10.1021/ac0200421, 12141651
Slysz G W, Schriemer D C. Blending protein separation and peptide analysis through real-time proteolytic digestion. Anal Chem, 2005, 77: 1572–1579, 1:CAS:528:DC%2BD2MXps1Kltg%3D%3D, 10.1021/ac048698c, 15762559
Duan J C, Sun L L, Liang Z, et al. Rapid protein digestion and identification using monolithic enzymatic microreactor coupled with nano-liquid chromatography-electrospray ionization mass spectrometry. J Chromatogr A, 2006, 1106: 165–174, 1:CAS:528:DC%2BD28XovVWjtQ%3D%3D, 10.1016/j.chroma.2005.11.102, 16364346
Ma J F, Qiao X Q, Deng Q L, et al. Organic-inorganic hybrid silica monolith based immobilized trypsin reactor with high enzymatic activity. Anal Chem, 2008, 80: 2949–2956, 1:CAS:528:DC%2BD1cXjtVCqsbg%3D, 10.1021/ac702343a, 18333626
Han M J, Lee S Y. The Escherichia coli proteome: Past, present, and future prospects. Microbiol Mol Biol R, 2006, 7: 362–439, 10.1128/MMBR.00036-05
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wu, S., Ma, J., Yang, K. et al. A novel organic-inorganic hybrid monolith for trypsin immobilization. Sci. China Life Sci. 54, 54–59 (2011). https://doi.org/10.1007/s11427-010-4108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4108-z