Abstract
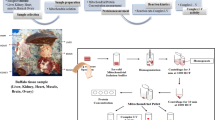
Mitochondrion plays the key functions in mammalian cells. It is believed that mitochondrion exerts the common biologic functions in many tissues, but also performs some specific functions correspondent with tissues where it is localized. To identify the tissue-specific mitochondrial proteins, we carried out a systematic survey towards mitochondrial proteins in the tissues of C57BL/6J mouse, such as liver, kidney and heart. The mitochondrial proteins were separated by 2DE and identified by MALDI-TOF/TOF MS. Total of 87 unique proteins were identified as the tissue-specific ones, and some representatives were further verified through ICPL quantification and Western blot. Because these issue-specific proteins are coded from nuclear genes, real-time PCR was employed to examine the mRNA status of six typical genes found in the tissues.With combining of the expression data and the co-localization images obtained from confocal microscope, we came to the conclusion that the tissue-specifically mitochondrial proteins were widely distributed among the mouse tissues. Our investigation, therefore, indeed provides a solid base to further explore the biological significance of the mitochondrial proteins with tissue-orientation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hockenbery D M, Oltvai Z N, Yin X M, et al. Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell, 1993, 75:241–251, 1:CAS:528:DyaK3sXms1Omtr8%3D, 10.1016/0092-8674(93)80066-N, 7503812
Kluck R M, Bossy-Wetzel E, Green D R, et al. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science, 1997, 275:1132–1136, 1:CAS:528:DyaK2sXhtlKjtro%3D, 10.1126/science.275.5303.1132, 9027315
Terkeltaub R, Johnson K, Murphy A, et al. Invited review: The mitochondrion in osteoarthritis. Mitochondrion, 2002, 1:301–319, 1:CAS:528:DC%2BD38XhvF2mtLo%3D, 10.1016/S1567-7249(01)00037-X, 16120285
Wallace D C. Mitochondrial diseases in man and mouse. Science, 1999, 283:1482–1488, 1:CAS:528:DyaK1MXhs1ygurk%3D, 10.1126/science.283.5407.1482, 10066162
Andersson S G, Zomorodipour A, Andersson J O, et al. The genome sequence of Rickettsia prowazekii and the origin of mitochondria. Nature, 1998, 396:133–140, 1:CAS:528:DyaK1cXnslamtL0%3D, 10.1038/24094, 9823893
Thorburn D R. Diverse powerhouses. Nat Genet, 2004, 36:13–14, 1:CAS:528:DC%2BD3sXhtVWhurbK, 10.1038/ng0104-13, 14702034
Shuman S K. A physician’s guide to coordinating oral health and primary care. Geriatrics, 1990, 45:47–51, 54, 57, 1:STN:280:DyaK3czjslemug%3D%3D, 2199337
Fawcett D W, Doxsey S, Buscher G. Salivary gland of the tick vector (R. appendiculatus) of East Coast fever. I. Ultrastructure of the type III acinus. Tissue Cell, 1981, 13:209–230, 1:STN:280:DyaL38%2FosFektg%3D%3D, 10.1016/0040-8166(81)90002-1, 7198303
Fawcett D W, Doxsey S, Buscher G. Salivary gland of the tick vector (R. appendiculatus) of East Coast fever. II. Cellular basis for fluid secretion in the type III acinus. Tissue Cell, 1981, 13:231–253, 1:STN:280:DyaL38%2FosFektw%3D%3D, 10.1016/0040-8166(81)90003-3, 7198304
Vayssiere J L, Cordeau L, Larcher J C, et al. Tissue-specific mitochondrial proteins. Biochimie, 1989, 71:787–791, 1:CAS:528:DyaL1MXlvFWqtb8%3D, 10.1016/0300-9084(89)90041-2, 2505859
Watmough N J, Bindoff L A, Birch-Machin M A, et al. Impaired mitochondrial beta-oxidation in a patient with an abnormality of the respiratory chain. Studies in skeletal muscle mitochondria. J Clin Invest, 1990, 85:177–184, 1:STN:280:DyaK3c7htVCktw%3D%3D, 10.1172/JCI114409, 2153151
Reichert A S, Neupert W. Mitochondriomics or what makes us breathe. Trends Genet, 2004, 20:555–562, 1:CAS:528:DC%2BD2cXotlajsL0%3D, 10.1016/j.tig.2004.08.012, 15475115
Mootha V K, Bunkenborg J, Olsen J V, et al. Integrated analysis of protein composition, tissue diversity, and gene regulation in mouse mitochondria. Cell, 2003, 115:629–640, 1:CAS:528:DC%2BD3sXps1OhtLk%3D, 10.1016/S0092-8674(03)00926-7, 14651853
Forner F, Foster L J, Campanaro S, et al. Quantitative proteomic comparison of rat mitochondria from muscle, heart, and liver. Mol Cell Proteomics, 2006, 5:608–619, 1:CAS:528:DC%2BD28XjsVCmtb0%3D, 16415296
Kislinger T, Cox B, Kannan A, et al. Global survey of organ and organelle protein expression in mouse: combined proteomic and transcriptomic profiling. Cell, 2006, 125:173–186, 1:CAS:528:DC%2BD28Xjsl2jsb0%3D, 10.1016/j.cell.2006.01.044, 16615898
Payne R M, Haas R C, Strauss A W. Structural characterization and tissue-specific expression of the mRNAs encoding isoenzymes from two rat mitochondrial creatine kinase genes. Biochim Biophys Acta, 1991, 1089:352–361, 1:CAS:528:DyaK38Xhs1OmsLk%3D, 1859839
Payne R M, Strauss A W. Expression of the mitochondrial creatine kinase genes. Mol Cell Biochem, 1994, 133/134:235–243, 10.1007/BF01267957
Baum C L, Kansal S, Davidson N O. Regulation of sterol carrier protein-2 gene expression in rat liver and small intestine. J Lipid Res, 1993, 34:729–739, 1:CAS:528:DyaK3sXksVKjsb4%3D, 8509712
Chesterton C J. Distribution of cholesterol precursors and other lipids among rat liver intracellular structures. J Biol Chem, 1968, 243:1147–1151, 1:CAS:528:DyaF1cXnsFSmsg%3D%3D, 5646164
Reinhart M P, Billheimer J T, Faust J R, et al. Subcellular localization of the enzymes of cholesterol biosynthesis and metabolism in rat liver. J Biol Chem, 1987, 262:9649–9655, 1:CAS:528:DyaL2sXlslejsbg%3D, 3597431
Keller G A, Scallen T J, Clarke D, et al. Subcellular localization of sterol carrier protein-2 in rat hepatocytes: its primary localization to peroxisomes. J Cell Biol, 1989, 108:1353–1361, 1:CAS:528:DyaL1MXhslSltrs%3D, 10.1083/jcb.108.4.1353, 2925789
Moncecchi D, Pastuszyn A, Scallen T J. cDNA sequence and bacterial expression of mouse liver sterol carrier protein-2. J Biol Chem, 1991, 266:9885–9892, 1:CAS:528:DyaK38XjsFehtg%3D%3D, 1709640
Gisler S M, Stagljar I, Traebert M, et al. Interaction of the type IIa Na/Pi cotransporter with PDZ proteins. J Biol Chem, 2001, 276:9206–9213, 1:CAS:528:DC%2BD3MXisVCmsr8%3D, 10.1074/jbc.M008745200, 11099500
Reczek D, Berryman M, Bretscher A. Identification of EBP50: A PDZ-containing phosphoprotein that associates with members of the ezrin-radixin-moesin family. J Cell Biol, 1997, 139:169–179, 1:CAS:528:DyaK2sXmsFaqsLc%3D, 10.1083/jcb.139.1.169, 9314537
Wang S, Yue H, Derin R B, et al. Accessory protein facilitated CFTR-CFTR interaction, a molecular mechanism to potentiate the chloride channel activity. Cell, 2000, 103:169–179, 1:CAS:528:DC%2BD3cXnt1yqurw%3D, 10.1016/S0092-8674(00)00096-9, 11051556
Yun C H, Oh S, Zizak M, et al. cAMP-mediated inhibition of the epithelial brush border Na+/H+ exchanger, NHE3, requires an associated regulatory protein. Proc Natl Acad Sci USA, 1997, 94:3010–3015, 1:CAS:528:DyaK2sXisVKgtr0%3D, 10.1073/pnas.94.7.3010, 9096337
Jiang X S, Dai J, Sheng Q H, et al. A comparative proteomic strategy for subcellular proteome research: ICAT approach coupled with bioinformatics prediction to ascertain rat liver mitochondrial proteins and indication of mitochondrial localization for catalase. Mol Cell Proteomics, 2005, 4:12–34, 1:CAS:528:DC%2BD2MXhtleitrs%3D, 15507458
Eaton S, Bursby T, Middleton B, et al. The mitochondrial trifunctional protein: Centre of a beta-oxidation metabolon? Biochem Soc Trans, 2000, 28:177–182, 1:CAS:528:DC%2BD3cXhsVKqtLk%3D, 10816122
Uchida Y, Izai K, Orii T, et al. Novel fatty acid beta-oxidation enzymes in rat liver mitochondria. II. Purification and properties of enoyl-coenzyme A (CoA) hydratase/3-hydroxyacyl-CoA dehydrogenase/3-ketoacyl-CoA thiolase trifunctional protein. J Biol Chem, 1992, 267:1034–1041, 1:CAS:528:DyaK38XnvFSqtw%3D%3D, 1730633
Bateman K S, Brownie E R, Wolodko W T, et al. Structure of the mammalian CoA transferase from pig heart. Biochemistry, 2002, 41:14455–14462, 1:CAS:528:DC%2BD38Xos1ejtb4%3D, 10.1021/bi020568f, 12463743
Rangarajan E S, Li Y, Ajamian E, et al. Crystallographic trapping of the glutamyl-CoA thioester intermediate of family I CoA transferases. J Biol Chem, 2005, 280:42919–42928, 1:CAS:528:DC%2BD2MXhtlCmsb7N, 10.1074/jbc.M510522200, 16253988
Hersh L B, Jencks W P. Isolation of an enzyme-coenzyme A intermediate from succinyl coenzyme A-acetoacetate coenzyme A transferase. J Biol Chem, 1967, 242:339–340, 1:CAS:528:DyaF2sXmt1WjtQ%3D%3D, 6016620
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wang, Y., Sun, H., Ru, Y. et al. Proteomic survey towards the tissue-specific proteins of mouse mitochondria. Sci. China Life Sci. 54, 3–15 (2011). https://doi.org/10.1007/s11427-010-4107-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4107-0