Abstract
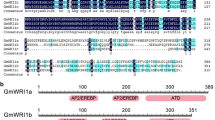
As the main structural protein of oil body, OLEOSIN is highly expressed only during seed development. OLEOSIN promoter is a very useful tool for seed-specific gene engineering and seed bioreactor designing. The B3 domain transcription factor leafy cotyledon2 (LEC2) plays an important role in regulating seed development and seed-specific gene expression. Here, we first report how seed-specific B3 domain transcription factor leafy cotyledon2 (LEC2) efficiently activates OLEOSIN expression. The central promoter region of OLEOSIN, responsible for seed specificity and LEC2 activation, was determined by 5′-deletion analysis. Binding experiments in yeast cells and electrophoretic mobility shift assays showed that LEC2 specifically bound to two conserved RY elements in this region. In transient expression assays, mutation in either RY element dramatically reduced LEC2 activation of OLEOSIN promoter activity, while double mutation abolished it. Analysis of the distribution of RY elements in seed-specific genes activated by LEC2 also supported the idea that genes containing neighboring RY elements responded strongly to LEC2 activation. Therefore, we conclude that two neighboring RY elements are essential for efficient LEC2 activation of OLEOSIN expression. These findings will help us better utilize seed-specific promoter activity.
Similar content being viewed by others
References
Huang A H C. OLEOSINs and oil bodies in seeds and other organs. Plant Physiol, 1996, 110: 1055–1061 8934621, 10.1104/pp.110.4.1055, 1:CAS:528:DyaK28XitlWjsbs%3D
Jolivet P, Roux E, Andrea S, et al. Protein composition of oil bodies in Arabidopsis thaliana ecotype WS. Plant Physiol Biochem, 2004, 42: 501–509 15246063, 10.1016/j.plaphy.2004.04.006, 1:CAS:528:DC%2BD2cXlsFyku7o%3D
Nykiforuk C L, Boothe J G, Murray E W, et al. Transgenic expression and recovery of biologically active recombinant human insulin from Arabidopsis thaliana seeds. Plant Biotechnol J, 2006, 4: 77–85 17177787, 10.1111/j.1467-7652.2005.00159.x, 1:CAS:528:DC%2BD28XisFWltLc%3D
Hatzopoulos P, Franz G, Choy L, et al. Interaction of nuclear factors with upstream sequences of a lipid body membrane protein gene from carrot. Plant Cell, 1990, 2: 457–467 2152171, 10.1105/tpc.2.5.457, 1:CAS:528:DyaK3MXhvVekuw%3D%3D
Keddie J S, Hubner G, Slocombe S P, et al. Cloning and characterisation of an oleosin from Brassica napus. Plant Mol Biol, 1992, 19: 443–453 1377966, 10.1007/BF00023392, 1:CAS:528:DyaK3sXit1Ogs7s%3D
Keddie J S, Tsiantis M, Piffanelli P, et al. A seed-specific Brassica napus oleosin promoter interacts with a G-box-specific protein and may be bi-directional. Plant Mol Biol, 1994, 24: 327–340 8111035, 10.1007/BF00020171, 1:CAS:528:DyaK2cXis1aqs7k%3D
Plant A L, Van Rooijen G J H, Anderson C P, et al. Regulation of an Arabidopsis oleosin promoter in transgenic Brassica napus. Plant Mol Biol, 1994, 25: 193–205 8018869, 10.1007/BF00023237, 1:CAS:528:DyaK2cXltVWitbk%3D
Rowley D L, Herman E M. The upstream domain of soybean oleosins contains regulatory elements similar to those of legume storage proteins. Biochim Biophys Acta, 1997, 1345: 1–4 9084495, 1:CAS:528:DyaK2sXht1Khs70%3D
Harada J J. Role of Arabidopsis leafy cotyledon genes in seed development. J Plant Physiol, 2001, 158: 405–409 10.1078/0176-1617-00351, 1:CAS:528:DC%2BD3MXjs1yjtL4%3D
Kirik V, Kölle K, Balzer H J, et al. Two new oleosin isoforms with altered expression patterns in seeds of the Arabidopsis mutant fus3. Plant Mol Biol, 1996, 31: 413–417 8756606, 10.1007/BF00021803, 1:CAS:528:DyaK28XltFGlu74%3D
To A, Valon C, Savino G, et al. A network of local and redundant gene regulation governs Arabidopsis seed maturation. Plant Cell, 2006, 18: 1642–1651 16731585, 10.1105/tpc.105.039925, 1:CAS:528:DC%2BD28XmvV2qt7k%3D
Crowe A J, Abenes M, Plant A, et al. The seed-specific transactivator, ABI3, induces oleosin expression. Plant Sci, 2000, 151: 171–181 10808073, 10.1016/S0168-9452(99)00214-9, 1:CAS:528:DC%2BD3cXhtF2rt7s%3D
Mendoza M S, Dubreucq B, Miquel M, et al. Leafy cotyledon2 activation is sufficient to trigger the accumulation of oil and seed-specific mRNAs in Arabidopsis leaves. FEBS Lett, 2005, 579: 4666–4670 10.1016/j.febslet.2005.07.037
Braybrook S A, Stone S L, Park S, et al. Genes directly regulated by leafy cotyledon2 provide insight into the control of embryo maturation and somatic embryogenesis. Proc Natl Acad Sci USA, 2006, 103: 3468–3473 16492731, 10.1073/pnas.0511331103, 1:CAS:528:DC%2BD28XksF2ku7k%3D
Clough S J, Bent A F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J, 1998, 16: 735–743 10069079, 10.1046/j.1365-313x.1998.00343.x, 1:STN:280:DyaK1M7mvVagsQ%3D%3D
Yang Y, Li R, Qi M. In vivo analysis of plant promoters and transcription factors by agroinfiltration of tobacco leaves. Plant J, 2000, 22: 534–551 10.1046/j.1365-313x.2000.00760.x
Jefferson R A, Kavanagh T A, Bevan M W. GUS fusions: Beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J, 1987, 6: 3901–3907 3327686, 1:CAS:528:DyaL1cXovV2itQ%3D%3D
Stalberg K, Ellerstom M, Josefsson L G, et al. Deletion analysis of a 2S seed storage protein promoter of Brassica napus in transgenic tobacco. Plant Mol Biol, 1993, 23: 671–683 8251622, 10.1007/BF00021523, 1:STN:280:DyaK2c%2Fnt1OitA%3D%3D
Gustavsson H O, Ellerström M, Stålberg K, et al. Distinct sequence elements in a napin promoter interact in vivo with DNA-binding proteins from Brassica napus. Physiol Plant, 1991, 82: 205–212 10.1111/j.1399-3054.1991.tb00082.x, 1:CAS:528:DyaK3MXks1Kjtro%3D
Bobb A J, Chern M S, Bustos M M. Conserved RY-repeats mediate transactivation of seed-specific promoters by the developmental regulator PvALF. Nucleic Acids Res, 1997, 25: 641–647 9016607, 10.1093/nar/25.3.641, 1:CAS:528:DyaK2sXhtF2ltL4%3D
Dickinson C D, Evans R P, Neilsen N C. RY repeats are conserved in the 5’-flanking regions of legume seed-protein genes. Nucleic Acids Res, 1988, 16: 371 3340541, 10.1093/nar/16.1.371, 1:CAS:528:DyaL1cXhtVOgsbo%3D
Hattori T, Terada T, Hamasuna S. Regulation of the Osem gene by abscisic acid and the transcriptional activator VP1: Analysis of cis-acting promoter elements required for regulation by abscisic acid and VP1. Plant J, 1995, 7: 913–925 7599651, 10.1046/j.1365-313X.1995.07060913.x, 1:CAS:528:DyaK2MXnsF2ltrk%3D
Kao C Y, Cocciolone S M, Vasil I K, et al. Localization and interaction of the cis-acting elements for abscisic acid, VIVIPAROUS1, and light activation of the C1 gene of maize. Plant Cell, 1996, 8: 1171–1179 8768375, 10.1105/tpc.8.7.1171, 1:CAS:528:DyaK28XksFKkurg%3D
Schmid M, Davison T S, Henz S R, et al. A gene expression map of Arabidopsis thaliana development. Nat Genet, 2005, 37: 501–506 15806101, 10.1038/ng1543, 1:CAS:528:DC%2BD2MXjsF2ksrg%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Grants from Toyota Motor Corporation of Japan and the National Special Project of Transgenic Organisms (Grant No. 2008ZX08010-001) of the Chinese Government.
Rights and permissions
About this article
Cite this article
Che, N., Yang, Y., Li, Y. et al. Efficient LEC2 activation of OLEOSIN expression requires two neighboring RY elements on its promoter. SCI CHINA SER C 52, 854–863 (2009). https://doi.org/10.1007/s11427-009-0119-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-009-0119-z