Abstract
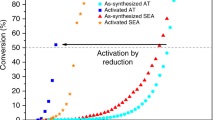
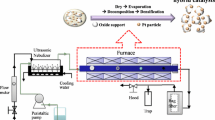
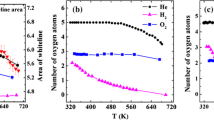
Monodispersed Pt colloids with a mean size of 2 nm were deposited uniformly on the {110} facets of a rod-shaped rutile TiO2, forming a well-defined Pt/TiO2 system. Oxidative treatment of this precursor at elevated temperatures re-dispersed the Pt particles into clusters and single-atoms. Air-calcination at 673 K partially oxidized the Pt particle surface, while calcination at 773 K yielded PtOx clusters of 1.6 nm in 7–8 atomic layers. Further calcination at 873 K formed a mixture of raft-like PtOx clusters (1.6 nm, 1–2 atomic layers) and cationic single-atoms. When tested for CO oxidation at 373 K, the Pt particles showed a higher activity than the PtOx clusters, whereas the cationic single-atoms were much less active. Subsequent H2-reduction at 473 K converted the partially oxidized Pt particles into the metallic species, but they were encapsulated by TiO2−x overlayers because of the strong metal–support interactions, which decreased the activity dramatically. H2-reduction of the PtOx clusters at 473 K enhanced the fraction of metallic Pt species without changing the size and geometry, and promoted the activity substantially. H2-treatment of Pt single-atoms at 473 K increased the activity only moderately because most Pt species still kept at cationic species. These results straightforwardly differentiated the catalytic behavior of Pt particles, clusters and single-atoms at the same metal loading and over the same TiO2 support, and further demonstrated that the electronic structures of Pt entities played a decisive role in the catalytic oxidation, in addition to the specified sizes.

Similar content being viewed by others
References
Therrien AJ, Hensley AJR, Marcinkowski MD, Zhang R, Lucci FR, Coughlin B, Schilling AC, McEwen JS, Sykes ECH. Nat Catal, 2018, 1: 192–198
van Deelen TW, Mejía CH, de Jong KP. Nat Catal, 2019, 2: 955–970
Mitchell S, Pérez-Ramírez J. Nat Commun, 2020, 11: 4302
Giulimondi V, Mitchell S, Pérez-Ramírez J. ACS Catal, 2023, 13: 2981–2997
Liu L, Corma A. Trends Chem, 2020, 2: 383–400
Santos VP, Carabineiro SAC, Tavares PB, Pereira MFR, Órfão JJM, Figueiredo JL. Appl Catal B-Environ, 2010, 99: 198–205
Royer S, Duprez D. ChemCatChem, 2011, 3: 24–65
Freund HJ, Meijer G, Scheffler M, Schlögl R, Wolf M. Angew Chem Int Ed, 2011, 50: 10064–10094
Lin J, Wang X, Zhang T. Chin J Catal, 2016, 37: 1805–1813
Kim GJ, Kwon DW, Hong SC. J Phys Chem C, 2016, 120: 17996–18004
Liu L, Meira DM, Arenal R, Concepcion P, Puga AV, Corma A. ACS Catal, 2019, 9: 10626–10639
Zhang H, Fang S, Hu YH. Catal Rev, 2020, 64: 491–532
Xiao Q, Wang Y, Zhao ZJ, Pei C, Chen S, Gao L, Mu R, Fu Q, Gong J. Sci China Chem, 2020, 63: 1323–1330
DeRita L, Dai S, Lopez-Zepeda K, Pham N, Graham GW, Pan X, Christopher P. J Am Chem Soc, 2017, 139: 14150–14165
Liu L, Corma A. Chem Rev, 2018, 118: 4981–5079
Bonanni S, Aït-Mansour K, Brune H, Harbich W. ACS Catal, 2011, 1: 385–389
Li L, Larsen AH, Romero NA, Morozov VA, Glinsvad C, Abild-Pedersen F, Greeley J, Jacobsen KW, Nørskov JK. J Phys Chem Lett, 2012, 4: 222–226
Ellaby T, Briquet L, Sarwar M, Thompsett D, Skylaris CK. J Chem Phys, 2019, 151: 114702
Yoo M, Kang E, Ha H, Yun J, Choi H, Lee JH, Kim TJ, Min J, Choi JS, Lee KS, Jung N, Kim S, Kim C, Yu YS, Kim HY. J Phys Chem Lett, 2022, 13: 1719–1725
Li N, Chen QY, Luo LF, Huang WX, Luo MF, Hu GS, Lu JQ. Appl Catal B-Environ, 2013, 142–143: 523–532
Ding K, Gulec A, Johnson AM, Schweitzer NM, Stucky GD, Marks LD, Stair PC. Science, 2015, 350: 189–192
Aso R, Hojo H, Takahashi Y, Akashi T, Midoh Y, Ichihashi F, Nakajima H, Tamaoka T, Yubuta K, Nakanishi H, Einaga H, Tanigaki T, Shinada H, Murakami Y. Science, 2022, 378: 202–206
Hojo H, Gondo M, Yoshizaki S, Einaga H. Nano Lett, 2022, 22: 145–150
Chua YPG, Gunasooriya GTKK, Saeys M, Seebauer EG. J Catal, 2014, 311: 306–313
Kim SB, Shin JH, Kim GJ, Hong SC. Ind Eng Chem Res, 2022, 61: 14793–14803
Liu J, Ding T, Zhang H, Li G, Cai J, Zhao D, Tian Y, Xian H, Bai X, Li X. Catal Sci Technol, 2018, 8: 4934–4944
Alayon EMC, Singh J, Nachtegaal M, Harfouche M, van Bokhoven JA. J Catal, 2009, 263: 228–238
Yoo M, Kang E, Choi H, Ha H, Choi H, Choi JS, Lee KS, Celestre R, Shapiro DA, Park JY, Kim C, Yu YS, Kim HY. J Mater Chem A, 2022, 10: 5942–5952
DeRita L, Resasco J, Dai S, Boubnov A, Thang HV, Hoffman AS, Ro I, Graham GW, Bare SR, Pacchioni G, Pan X, Christopher P. Nat Mater, 2019, 18: 746–751
Choi H, Lee J, Kim D, Kumar A, Jeong B, Kim KJ, Lee H, Park JY. Catal Sci Technol, 2021, 11: 1698–1708
Bamwenda GR, Tsubota S, Nakamura T, Haruta M. Catal Lett, 1997, 44: 83–87
Resasco J, Yang F, Mou T, Wang B, Christopher P, Resasco DE. ACS Catal, 2019, 10: 595–603
Kuo CT, Lu Y, Kovarik L, Engelhard M, Karim AM. ACS Catal, 2019, 9: 11030–11041
Jones J, Xiong H, DeLaRiva AT, Peterson EJ, Pham H, Challa SR, Qi G, Oh S, Wiebenga MH, Hernández XIP, Wang Y, Datye AK. Science, 2016, 353: 150–154
Nur ASM, Funada E, Kiritoshi S, Matsumoto A, Kakei R, Hinokuma S, Yoshida H, Machida M. J Phys Chem C, 2018, 122: 662–669
Cheng H, Zhu YA, Chen D, Åstrand PO, Li P, Qi Z, Zhou XG. J Phys Chem C, 2014, 118: 23711–23722
Wang G, Xu YS, Qian P, Su YJ. Comput Mater Sci, 2019, 169: 109090
Chang TY, Tanaka Y, Ishikawa R, Toyoura K, Matsunaga K, Ikuhara Y, Shibata N. Nano Lett, 2014, 14: 134–138
Tao F, Dag S, Wang LW, Liu Z, Butcher DR, Bluhm H, Salmeron M, Somorjai GA. Science, 2010, 327: 850–853
Lykhach Y, Kozlov SM, Skála T, Tovt A, Stetsovych V, Tsud N, Dvořák F, Johánek V, Neitzel A, Mysliveček J, Fabris S, Matolín V, Neyman KM, Libuda J. Nat Mater, 2016, 15: 284–288
Macino M, Barnes AJ, Althahban SM, Qu R, Gibson EK, Morgan DJ, Freakley SJ, Dimitratos N, Kiely CJ, Gao X, Beale AM, Bethell D, He Q, Sankar M, Hutchings GJ. Nat Catal, 2019, 2: 873–881
Ma Y, Ge H, Yi S, Yang M, Feng D, Ren Y, Gao J, Qin Y. Sci China Chem, 2022, 65: 2596–2603
Petzoldt P, Eder M, Mackewicz S, Blum M, Kratky T, Günther S, Tschurl M, Heiz U, Lechner BAJ. J Phys Chem C, 2022, 126: 16127–16139
Beck A, Huang X, Artiglia L, Zabilskiy M, Wang X, Rzepka P, Palagin D, Willinger MG, van Bokhoven JA. Nat Commun, 2020, 11: 3220
Wang JG, Li WX, Borg M, Gustafson J, Mikkelsen A, Pedersen TM, Lundgren E, Weissenrieder J, Klikovits J, Schmid M, Hammer B, Andersen JN. Phys Rev Lett, 2005, 95: 256102
Held G, Jones LB, Seddon EA, King DA. J Phys Chem B, 2005, 109: 6159–6163
Miller DJ, Öberg H, Kaya S, Casalongue HS, Friebel D, Anniyev T, Ogasawara H, Bluhm H, Pettersson LGM, Nilsson A. Phys Rev Lett, 2011, 107: 195502
van Spronsen MA, Frenken JWM, Groot IMN. Nat Commun, 2017, 8: 429
Lang R, Xi W, Liu JC, Cui YT, Li T, Lee AF, Chen F, Chen Y, Li L, Li L, Lin J, Miao S, Liu X, Wang AQ, Wang X, Luo J, Qiao B, Li J, Zhang T. Nat Commun, 2019, 10: 234
Han B, Guo Y, Huang Y, Xi W, Xu J, Luo J, Qi H, Ren Y, Liu X, Qiao B, Zhang T. Angew Chem Int Ed, 2020, 59: 11824–11829
Avanesian T, Dai S, Kale MJ, Graham GW, Pan X, Christopher P. J Am Chem Soc, 2017, 139: 4551–4558
Zhou P, Zhang H, Ji H, Ma W, Chen C, Zhao J. Sci China Chem, 2020, 63: 354–360
Tigwell M, Douthwaite M, Smith LR, Dummer NF, Morgan DJ, Bethell D, Taylor SH, Hutchings GJ. J Phys Chem C, 2022, 126: 15651–15661
Qiao B, Wang A, Yang X, Allard LF, Jiang Z, Cui Y, Liu J, Li J, Zhang T. Nat Chem, 2011, 3: 634–641
Kottwitz M, Li Y, Palomino RM, Liu Z, Wang G, Wu Q, Huang J, Timoshenko J, Senanayake SD, Balasubramanian M, Lu D, Nuzzo RG, Frenkel AI. ACS Catal, 2019, 9: 8738–8748
Gao F, Wang Y, Cai Y, Goodman DW. J Phys Chem C, 2009, 113: 174–181
Gustafson J, Balmes O, Zhang C, Shipilin M, Schaefer A, Hagman B, Merte LR, Martin NM, Carlsson PA, Jankowski M, Crumlin EJ, Lundgren E. ACS Catal, 2018, 8: 4438–4445
Li X, Wang X, Sadykov II, Palagin D, Safonova OV, Li J, Beck A, Krumeich F, van Bokhoven JA, Artiglia L. ACS Catal, 2021, 11: 13041–13049
Maurer F, Beck A, Jelic J, Wang W, Mangold S, Stehle M, Wang D, Dolcet P, Gänzler AM, Kübel C, Studt F, Casapu M, Grunwaldt JD. ACS Catal, 2022, 12: 2473–2486
Pedersen TM, Li WX, Hammer B. Phys Chem Chem Phys, 2006, 8: 1566–1574
Miller D, Casalongue HS, Bluhm H, Ogasawara H, Nilsson A, Kaya S. J Am Chem Soc, 2014, 136: 6340–6347
van Spronsen MA, Frenken JWM, Groot IMN. Chem Soc Rev, 2017, 46: 4347–4374
Wang H, Wang L, Xiao FS. Sci China Chem, 2022, 65: 2051–2057
Beck A, Frey H, Huang X, Clark AH, Goodman ED, Cargnello M, Willinger M, van Bokhoven JA. Angew Chem Int Ed, 2023, 62: e202301468
Shirley DA. Chem Phys Lett, 1972, 16: 220–225
Jackson SD, Willis J, Mclellan GD, Webb G, Keegan MBT, Moyes RB, Simpson S, Wells PB, Whyman R. J Catal, 1993, 139: 191–206
Boyanov BI, Morrison TI. J Phys Chem, 1996, 100: 16318–16326
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22002164).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at chem.scichina.com and link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Rights and permissions
About this article
Cite this article
Zhang, X., Shi, W., Han, S. et al. Interplay between geometric and electronic structures of Pt entities over TiO2 for CO oxidation. Sci. China Chem. 67, 705–714 (2024). https://doi.org/10.1007/s11426-023-1874-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1874-0