Abstract
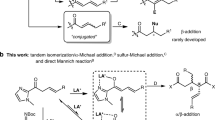
The diastereo- and enantioselective tandem cycloisomerization/[4 + 2]-cycloaddition reaction of cyclopropyl tethered allenyl ketones with α,β-unsaturated carbonyl compounds was realized firstly under mild reaction conditions by using bimetallic silver (I)/chiral metal-N,N′-dioxide catalysts, providing a facile and atom-economic route to enantioenriched polysubstituted furopyran derivatives with good results. Possible transition state models were proposed to explain the stereoinduction.

Similar content being viewed by others
References
Volla CMR, Atodiresei I, Rueping M. Chem Rev, 2014, 114: 2390–2431
Ardkhean R, Caputo DFJ, Morrow SM, Shi H, Xiong Y, Anderson EA. Chem Soc Rev, 2016, 45: 1557–1569
Wasilke JC, Obrey SJ, Baker RT, Bazan GC. Chem Rev, 2005, 105: 1001–1020
Krause N, Winter C. Chem Rev, 2011, 111: 1994–2009
Garayalde D, Nevado C. ACS Catal, 2012, 2: 1462–1479
Miao M, Cao J, Zhang J, Huang X, Wu L. J Org Chem, 2013, 78: 2687–2692
Zhang S, Tang A, Chen P, Zhao Z, Miao M, Ren H. Org Lett, 2020, 22: 848–853
Zhang S, Xie R, Tang A, Chen P, Zhao Z, Miao M, Ren H. Org Lett, 2020, 22: 3056–3061
Miao M, Xu H, Jin M, Chen Z, Xu J, Ren H. Org Lett, 2018, 20: 3096–3100
Miao M, Cao J, Zhang J, Huang X, Wu L. Org Lett, 2012, 14: 2718–2721
Xie R, Zhao Z, Zhao Y, Li R, Yao J, Miao M. Org Lett, 2022, 24: 2220–2225
Teske J, Plietker B. Org Lett, 2018, 20: 2257–2260
Miao M, Luo Y, Xu H, Jin M, Chen Z, Xu J, Ren H. J Org Chem, 2017, 82: 12224–12237
Miao M, Luo Y, Xu H, Chen Z, Xu J, Ren H. Org Lett, 2016, 18: 4292–4295
Xie R, Li R, Zhao Q, Zhao Y, Yao J, Miao M. J Org Chem, 2023, 88: 4778–4789
Zhang S, Tang A, Xie R, Zhao Z, Yao J, Miao M. Org Lett, 2021, 23: 3701–3705
Zhang Y, Wei Y, Shi M. Chem Commun, 2021, 57: 3599–3602
Zhang ZM, Chen P, Li W, Niu Y, Zhao XL, Zhang J. Angew Chem Int Ed, 2014, 53: 4350–4354
Naodovic M, Yamamoto H. Chem Rev, 2008, 108: 3132–3148
Bai XF, Xu Z, Xia CG, Zheng ZJ, Xu LW. ACS Catal, 2015, 5: 6016–6020
Fu W, Tang W. ACS Catal, 2016, 6: 4814–4858
Song C, Wang J, Xu Z. Org Biomol Chem, 2014, 12: 5802–5806
Wang Y, Lu H, Xu PF. Acc Chem Res, 2015, 48: 1832–1844
Patil NT, Shinde VS, Gajula B. Org Biomol Chem, 2012, 10: 211–224
Matsunaga S, Shibasaki M. Chem Commun, 2014, 50: 1044–1057
Li Z, Zhao F, Ou W, Huang P-, Wang X. Angew Chem Int Ed, 2021, 60: 26604–26609
Xiao JA, Peng H, Zhang H, Meng RF, Lin C, Su W, Huang Y. Org Lett, 2022, 24: 8709–8713
Martínez S, Veth L, Lainer B, Dydio P. ACS Catal, 2021, 11: 3891–3915
Liu XH, Lin LL, Feng XM. Acc Chem Res, 2011, 44: 574–587
Liu X, Lin L, Feng X. Org Chem Front, 2014, 1: 298–302
Liu X, Zheng H, Xia Y, Lin L, Feng X. Acc Chem Res, 2017, 50: 2621–2631
Liu X, Dong S, Lin L, Feng X. Chin J Chem, 2018, 36: 791–797
Xie M, Lin L, Feng X. Chem Rec, 2017, 17: 1184–1202
Shao Z, Zhang H. Chem Soc Rev, 2009, 38: 2745–2755
Loh CCJ, Enders D. Chem Eur J, 2012, 18: 10212–10225
Dhiman S, Mishra UK, Ramasastry SSV. Angew Chem Int Ed, 2016, 55: 7737–7741
Xu C, Qiao J, Dong S, Zhou Y, Liu X, Feng X. Chem Sci, 2021, 12: 5458–5463
Fu J, Huo X, Li B, Zhang W. Org Biomol Chem, 2017, 15: 9747–9759
Moyano A, Rios R. Chem Rev, 2011, 111: 4703–4832
Jiang X, Wang R. Chem Rev, 2013, 113: 5515–5546
Marichev KO, Doyle MP. Org Biomol Chem, 2019, 17: 4183–4195
Klier L, Tur F, Poulsen PH, Jørgensen KA. Chem Soc Rev, 2017, 46: 1080–1102
Lu Y, Zhou Y, Lin L, Zheng H, Fu K, Liu X, Feng X. Chem Commun, 2016, 52: 8255–8258
Song X, Chen Y, Lu F, Zhang K, Yu C, Li T, Yao C. Org Biomol Chem, 2022, 20: 1219–1225
Wang Y, Chen Y, Li X, Mao Y, Chen W, Zhan R, Huang H. Org Biomol Chem, 2019, 17: 3945–3950
Huang Y, Zha Z, Wang Z. Org Lett, 2020, 22: 2512–2516
Luo Y, Zhang H, Wang S, Zhou Y, Dong S, Feng X. Org Lett, 2020, 22: 2645–2650
Yao H, Zhou Y, Chen X, Zhang P, Xu J, Liu H. J Org Chem, 2016, 81: 8888–8899
Xiao L, Jin L, Zhao Y, Guo J, Stephan DW. Chem Commun, 2023, 59: 1833–1836
Day J, McKeever-Abbas B, Dowden J. Angew Chem Int Ed, 2016, 55: 5809–5813
Tan C, Chen W, Mu X, Chen Q, Gong J, Luo T, Yang Z. Org Lett, 2015, 17: 2338–2341
Khalaf JK, VanderVelde DG, Datta A. J Org Chem, 2008, 73: 5977–5984
Do H, Kang CW, Cho JH, Gilbertson SR. Org Lett, 2015, 17: 3972–3974
Hirai T, Shibata K, Niwano Y, Shiozaki M, Hashimoto Y, Morita N, Ban S, Tamura O. Org Lett, 2017, 19: 6320–6323
Degorce SL, Bodnarchuk MS, Cumming IA, Scott JS. J Med Chem, 2018, 61: 8934–8943
Wang M, Lu P. Org Chem Front, 2018, 5: 254–259
Dembitsky VM. Phytomedicine, 2014, 21: 1559–1581
CCDC 2239562 (3la), 2239560 (5aa), 2004841 (Lu(OTf)3/ent-L3-PrPr2Br) and 2022853 (Co(BF4)2/L2-PrPr2)
Mo Y, Chen Q, Li J, Ye D, Zhou Y, Dong S, Liu X, Feng X. ACS Catal, 2023, 13: 877–886
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21890723, 22171189) and the Fundamental Research Funds for the Central Universities (YJ201819).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
11426_2023_1667_MOESM1_ESM.pdf
Asymmetric Annulations Based on Cycloisomerization of Cyclopropyl Tethered Allenyl Ketones by Bimetallic Relay Catalysis
Rights and permissions
About this article
Cite this article
He, J., Liu, W., Zhang, J. et al. Asymmetric annulations based on cycloisomerization of cyclopropyl tethered allenyl ketones by bimetallic relay catalysis. Sci. China Chem. 66, 2803–2809 (2023). https://doi.org/10.1007/s11426-023-1667-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1667-9