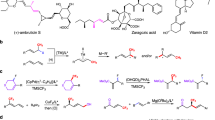
Abstract
C-C bond activation has emerged as a powerful tool for the construction of complex molecules. Herein, we report a dual ligands relay-promoted transformation of unstrained aryl, alkenyl and alkynyl ketones to the corresponding polyfluoroarenes and nitriles via C-C (=O) bond cleavage and subsequent decarboxylative arylation process. Various polyfluoroarene and nitrile products are obtained in one pot under cyanide-free conditions. The protocol features high atom economy, broad functional group tolerance and excellent heterocyclic compatibility. The late-stage functionalization of the drug and natural product demonstrated the synthetic utility of our protocol. Furthermore, the decisive role of the dual ligands was clarified and the mechanistic rationale including the β−C elimination as the rate-limiting step was supported by detailed density functional theory (DFT) studies.

Similar content being viewed by others
References
Meyer EA, Castellano RK, Diederich F. Angew Chem Int Ed, 2003, 42: 1210–1250
Zahn A, Brotschi C, Leumann CJ. Chem Eur J, 2005, 11: 2125–2129
Muller K, Faeh C, Diederich F. Science, 2007, 317: 1881–1886
Purser S, Moore PR, Swallow S, Gouverneur V. Chem Soc Rev, 2008, 37: 320–330
de Candia M, Liantonio F, Carotti A, De Cristofaro R, Altomare C. J Med Chem, 2009, 52: 1018–1028
Sakamoto Y, Suzuki T, Miura A, Fujikawa H, Tokito S, Taga Y. J Am Chem Soc, 2000, 122: 1832–1833
Zacharias P, Gather M, Rojahn M, Nuyken O, Meerholz K. Angew Chem Int Ed, 2007, 46: 4388–4392
Babudri F, Farinola GM, Naso F, Ragni R. Chem Commun, 2007, 43: 1003–1022
Korenaga T, Kosaki T, Fukumura R, Ema T, Sakai T. Org Lett, 2005, 7: 4915–4917
Lafrance M, Rowley CN, Woo TK, Fagnou K. J Am Chem Soc, 2006, 128: 8754–8756
Do HQ, Daugulis O. J Am Chem Soc, 2008, 130: 1128–1129
Wei Y, Kan J, Wang M, Su W, Hong M. Org Lett, 2009, 11: 3346–3349
Fan S, He CY, Zhang X. Chem Commun, 2010, 46: 4926–4928
Fan S, Yang J, Zhang X. Org Lett, 2011, 13: 4374–4377
Dahiya A, Fricke C, Schoenebeck F. J Am Chem Soc, 2020, 142: 7754–7759
Gooßen LJ, Deng G, Levy LM. Science, 2006, 313: 662–664
Gooßen LJ, Rodríguez N, Melzer B, Linder C, Deng G, Levy LM. J Am Chem Soc, 2007, 129: 4824–4833
Gooßen L, Zimmermann B, Knauber T. Angew Chem Int Ed, 2008, 47: 7103–7106
Gooßen LJ, Rodríguez N, Linder C. J Am Chem Soc, 2008, 130: 15248–15249
Rodríguez N, Gooßen LJ. Chem Soc Rev, 2011, 40: 5030–5048
Wei Y, Hu P, Zhang M, Su W. Chem Rev, 2017, 117: 8864–8907
Perry GJP, Larrosa I. Eur J Org Chem, 2017, 2017(25): 3517–3527
Shang R, Fu Y, Wang Y, Xu Q, Yu HZ, Liu L. Angew Chem Int Ed, 2009, 48: 9350–9354
Fu L, Chen Q, Nishihara Y. Org Lett, 2020, 22: 6388–6393
Becht JM, Drian CL. Org Lett, 2008, 10: 3161–3164
Sardzinski LW, Wertjes WC, Schnaith AM, Kalyani D. Org Lett, 2015, 17: 1256–1259
Wei Y, Su W. J Am Chem Soc, 2010, 132: 16377–16379
He CY, Fan S, Zhang X. J Am Chem Soc, 2010, 132: 12850–12852
Li H, Liu J, Sun CL, Li BJ, Shi ZJ. Org Lett, 2011, 13: 276–279
Fleming FF, Yao L, Ravikumar PC, Funk L, Shook BC. J Med Chem, 2010, 53: 7902–7917
Miller JS, Manson JL. Acc Chem Res, 2001, 34: 563–570
Rappoport Z. The Chemistry of the Cyano Group. London: Interscience Publishers, 1970
Fleming FF, Fleming FF. Nat Prod Rep, 1999, 16: 597–606
Larock RC. Comprehensive Organic Transformations: A Guide to Functional Group Preparations. 3rd ed. Hoboken: John Wiley and Sons, 2018
Larock RC, Yao T. Formation of Nitriles, Carboxylic acids, and Derivatives by Oxidation, Substitution, and Addition. Comprehensive Organic Transformations. 3rd ed. Wiley: Weinheim, 2018
Anbarasan P, Schareina T, Beller M. Chem Soc Rev, 2011, 40: 5049–5067
Sundermeier M, Zapf A, Mutyala S, Baumann W, Sans J, Weiss S, Beller M. Chem Eur J, 2003, 9: 1828–1836
Kim J, Kim HJ, Chang S. Angew Chem Int Ed, 2012, 51: 11948–11959
Culkin DA, Hartwig JF. J Am Chem Soc, 2002, 124: 9330–9331
Wu L, Hartwig JF. J Am Chem Soc, 2005, 127: 15824–15832
You J, Verkade JG. Angew Chem Int Ed, 2003, 42: 5051–5053
Chen Y, Xu L, Jiang Y, Ma D. Angew Chem Int Ed, 2021, 60: 7082–7086
Ping Y, Ding Q, Peng Y. ACS Catal, 2016, 6: 5989–6005
Jun CH. Chem Soc Rev, 2004, 33: 610–618
Tobisu M, Chatani N. Chem Soc Rev, 2008, 37: 300–307
Nakao Y, Hiyama T. Pure Appl Chem, 2008, 80: 1097–1107
Ruhland K. Eur J Org Chem, 2012, 2012(14): 2683–2706
Chen F, Wang T, Jiao N. Chem Rev, 2014, 114: 8613–8661
Liu H, Feng M, Jiang X. Chem Asian J, 2014, 9: 3360–3389
Souillart L, Cramer N. Chem Rev, 2015, 115: 9410–9464
Murakami M, Ishida N. J Am Chem Soc, 2016, 138: 13759–13769
Tsang ASK, Kapat A, Schoenebeck F. J Am Chem Soc, 2016, 138: 518–526
Kim DS, Park WJ, Jun CH. Chem Rev, 2017, 117: 8977–9015
Song F, Gou T, Wang BQ, Shi ZJ. Chem Soc Rev, 2018, 47: 7078–7115
To CT, Chan KS. Eur J Org Chem, 2019, 2019(39): 6581–6591
Deng L, Dong G. Trends Chem, 2020, 2: 183–198
Hu X, Shao Y, Xie H, Chen X, Chen F, Ke Z, Jiang H, Zeng W. ACS Catal, 2020, 10: 8402–8408
Walter MW. Nat Prod Rep, 2002, 19: 278–291
Ertl P, Schuhmann T. J Nat Prod, 2019, 82: 1258–1263
Huang H, Ji X, Wu W, Jiang H. Chem Soc Rev, 2015, 44: 1155–1171
Huang H, Cai J, Deng GJ. Org Biomol Chem, 2016, 14: 1519–1530
Walton JC. Acc Chem Res, 2014, 47: 1406–1416
Blake JA, Pratt DA, Lin S, Walton JC, Mulder P, Ingold KU. J Org Chem, 2004, 69: 3112–3120
Kitamura M, Narasaka K. Chem Record, 2002, 2: 268–277
Tsutsui H, Narasaka K. Chem Lett, 1999, 28: 45–46
Faulkner A, Scott JS, Bower JF. J Am Chem Soc, 2015, 137: 7224–7230
Hazelden IR, Ma X, Langer T, Bower JF. Angew Chem Int Ed, 2016, 55: 11198–11202
Bao X, Wang Q, Zhu J. Angew Chem Int Ed, 2017, 56: 9577–9581
Chen C, Hou L, Cheng M, Su J, Tong X. Angew Chem Int Ed, 2015, 54: 3092–3096
Liu S, Liebeskind LS. J Am Chem Soc, 2008, 130: 6918–6919
Shuler SA, Yin G, Krause SB, Vesper CM, Watson DA. J Am Chem Soc, 2016, 138: 13830–13833
Xu F, Shuler SA, Watson DA. Angew Chem Int Ed, 2018, 57: 12081–12085
Xu F, Korch KM, Watson DA. Angew Chem Int Ed, 2019, 58: 13448–13451
Tan Y, Hartwig JF. J Am Chem Soc, 2010, 132: 3676–3677
Nishimura T, Uemura S. J Am Chem Soc, 2000, 122: 12049–12050
Zhao B, Shi Z. Angew Chem Int Ed, 2017, 56: 12727–12731
Yu X, Chen J, Wang P, Yang M, Liang D, Xiao W. Angew Chem Int Ed, 2018, 57: 738–743
Yu X, Zhao Q, Chen J, Chen J, Xiao W. Angew Chem Int Ed, 2018, 57: 15505–15509
Wang T, Wang YN, Wang R, Zhang BC, Yang C, Li YL, Wang XS. Nat Commun, 2019, 10: 5373–5382
Xiao T, Huang H, Anand D, Zhou L. Synthesis, 2020, 52: 1585–1601
Yu XY, Chen JR, Xiao WJ. Chem Rev, 2021, 121: 506–561
Wang PZ, Gao Y, Chen J, Huan XD, Xiao WJ, Chen JR. Nat Commun, 2021, 12: 1815–1825
Chen J, Wang PZ, Lu B, Liang D, Yu XY, Xiao WJ, Chen JR. Org Lett, 2019, 21: 9763–9768
Zuo HD, Zhu SS, Hao WJ, Wang SC, Tu SJ, Jiang B. ACS Catal, 2021, 11: 6010–6019
Zhu C, Chen F, Liu C, Zeng H, Yang Z, Wu W, Jiang H. J Org Chem, 2018, 83: 14713–14722
Xu Y, Qi X, Zheng P, Berti CC, Liu P, Dong G. Nature, 2019, 567: 373–378
Li H, Ma B, Liu Q, Wang M, Wang Z, Xu H, Li L, Wang X, Dai H. Angew Chem Int Ed, 2020, 59: 14388–14393
Xu H, Ma B, Fu Z, Li HY, Wang X, Wang ZY, Li LJ, Cheng TJ, Zheng M, Dai HX. ACS Catal, 2021, 11: 1758–1764
Guo ZQ, Xu H, Wang X, Wang ZY, Ma B, Dai HX. Chem Commun, 2021, 57: 9716–9719
Wang ML, Xu H, Li HY, Ma B, Wang ZY, Wang X, Dai HX. Org Lett, 2021, 23: 2147–2152
Wang ZY, Ma B, Xu H, Wang X, Zhang X, Dai HX. Org Lett, 2021, 23: 8291–8295
Reetz M. Angew Chem Int Ed, 2008, 47: 2556–2588
Duursma A, Hoen R, Schuppan J, Hulst R, Minnaard AJ, Feringa BL. Org Lett, 2003, 5: 3111–3113
Meng G, Wang Z, Chan HSS, Chekshin N, Li Z, Wang P, Yu JQ. J Am Chem Soc, 2023, 145: 8198–8208
Chen H, Wedi P, Meyer T, Tavakoli G, van Gemmeren M. Angew Chem Int Ed, 2018, 57: 2497–2501
Sinha SK, Panja S, Grover J, Hazra PS, Pandit S, Bairagi Y, Zhang X, Maiti D. J Am Chem Soc, 2022, 144: 12032–12042
Fors BP, Buchwald SL. J Am Chem Soc, 2010, 132: 15914–15917
Chen C, Peters JC, Fu GC. Nature, 2021, 596: 250–256
Kim-Lee SH, Mauleón P, Gómez Arrayás R, Carretero JC. Chem, 2021, 7: 2212–2226
Zhang Y, Ma J, Chen J, Meng L, Liang Y, Zhu S. Chem, 2021, 7: 3171–3188
Sun Y, Guo J, Shen X, Lu Z. Nat Commun, 2022, 13: 650
Zhang F, Greaney M. Angew Chem Int Ed, 2010, 49: 2768–2771
Zhao H, Wei Y, Xu J, Kan J, Su W, Hong M. J Org Chem, 2011, 76: 882–893
Hu P, Zhang M, Jie X, Su W. Angew Chem Int Ed, 2012, 51: 227–231
Acknowledgements
This work was supported by the Shanghai Institute of Materia Medica, the Chinese Academy of Sciences, the National Natural Science Foundation of China (21772211, 21920102003), the Institutes for Drug Discovery and Development, Chinese Academy of Sciences (CASIMM0120163006), the Science and Technology Commission of Shanghai Municipality (17JC1405000, 18431907100), the Program of Shanghai Academic Research Leader (19XD1424600), the National Science & Technology Major Project “Key New Drug Creation and Manufacturing Program”, China (2018ZX09711002-006) and the China Postdoctoral Science Foundation (2019M662854).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting information The supporting information is available online at https://chem.scichina.com and https://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Conflict of interest The authors declare no conflict of interest.
Supporting Information For
Rights and permissions
About this article
Cite this article
Wang, ZY., Xu, H., Zhang, X. et al. Dual ligands relay-promoted transformation of unstrained ketones to polyfluoroarenes and nitriles. Sci. China Chem. 66, 2037–2045 (2023). https://doi.org/10.1007/s11426-023-1653-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1653-3