Abstract
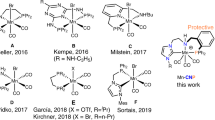
Noble metal catalysts are generally expensive, and thus, abundant 3d metals recently received significant attention as catalysts in catalytic hydrogenation. Mn catalysts are widely applied in transfer hydrogenations, but the reported catalyst loadings remain up to three orders of magnitude higher than noble metals. Thus, catalyst consumption should be overcome before 3d metal catalytic systems may be utilized practically in industry. Here, a catalytic system based on novel, scalable triazole N5-ligands coordinated to Mn is presented for use in transfer hydrogenations. Based on pre-activation via dehydrohalogenation, an unprecedented, efficient catalytic system operating via synergistic H-bond auxiliary activation was established. The Mn catalysts are practical at metal concentrations 0.0001 mol%, generating alcohol with turnover number (TON) up to 857,200, thus approaching loadings more conventionally observed in precious-metal-based systems. Notably, using this protocol, several pharmaceuticals may be easily synthesized in one pot.

Similar content being viewed by others
References
Weissermel K, Arpe HJ. Industrial Organic Chemistry. Weinheim: Wiley-VCH, 2003
de Vries JG, Elsevier CJ. The Handbook of Homogeneous Hydrogenation. Weinhem: Wiley-VCH, 2007, Vol. 1
Crabtree RH. Chem Rev, 2015, 115: 127–150
Deng D, Hu B, Yang M, Chen D. Organometallics, 2018, 37: 2386–2394
Vispute TP, Zhang H, Sanna A, Xiao R, Huber GW. Science, 2010, 330: 1222–1227
Barta K, Ford PC. Acc Chem Res, 2014, 47: 1503–1512
Kumar A, Daw P, Milstein D. Chem Rev, 2022, 122: 385–441
Elangovan S, Topf C, Fischer S, Jiao H, Spannenberg A, Baumann W, Ludwig R, Junge K, Beller M. J Am Chem Soc, 2016, 138: 8809–8814
Kallmeier F, Irrgang T, Dietel T, Kempe R. Angew Chem Int Ed, 2016, 55: 11806–11809
Widegren MB, Harkness GJ, Slawin AMZ, Cordes DB, Clarke ML. Angew Chem Int Ed, 2017, 56: 5825–5828
Garbe M, Junge K, Walker S, Wei Z, Jiao H, Spannenberg A, Bachmann S, Scalone M, Beller M. Angew Chem Int Ed, 2017, 56: 11237–11241
Li YY, Yu SL, Shen WY, Gao JX. Acc Chem Res, 2015, 48: 2587–2598
Wang D, Astruc D. Chem Rev, 2015, 115: 6621–6686
Valyaev DA, Lavigne G, Lugan N. Coord Chem Rev, 2016, 308: 191–235
Alig L, Fritz M, Schneider S. Chem Rev, 2019, 119: 2681–2751
Irrgang T, Kempe R. Chem Rev, 2019, 119: 2524–2549
Casey CP, Strotman NA, Beetner SE, Johnson JB, Priebe DC, Guzei IA. Organometallics, 2006, 25: 1236–1244
Bower JF, Skucas E, Patman RL, Krische MJ. J Am Chem Soc, 2007, 129: 15134–15135
Guillena G, Ramón DJ, Yus M. Chem Rev, 2010, 110: 1611–1641
Choi J, MacArthur AHR, Brookhart M, Goldman AS. Chem Rev, 2011, 111: 1761–1779
Crabtree RH. Organometallics, 2011, 30: 17–19
Marr AC. Catal Sci Technol, 2012, 2: 279–287
Pan S, Shibata T. ACS Catal, 2013, 3: 704–712
Obora Y. ACS Catal, 2014, 4: 3972–3981
Yang Q, Wang Q, Yu Z. Chem Soc Rev, 2015, 44: 2305–2329
Quintard A, Rodriguez J. ChemSusChem, 2016, 9: 28–30
Xie Y, Ben-David Y, Shimon LJW, Milstein D. J Am Chem Soc, 2016, 138: 9077–9080
Langer R, Leitus G, Ben-David Y, Milstein D. Angew Chem Int Ed, 2011, 50: 2120–2124
Langer R, Iron MA, Konstantinovski L, Diskin-Posner Y, Leitus G, Ben-David Y, Milstein D. Chem Eur J, 2012, 18: 7196–7209
Fleischer S, Zhou S, Junge K, Beller M. Angew Chem Int Ed, 2013, 52: 5120–5124
Gorgas N, Stöger B, Veiros LF, Pittenauer E, Allmaier G, Kirchner K. Organometallics, 2014, 33: 6905–6914
Zell T, Ben-David Y, Milstein D. Catal Sci Technol, 2015, 5: 822–826
Gorgas N, Stöger B, Veiros LF, Kirchner K. ACS Catal, 2016, 6: 2664–2672
Maji B, Barman M. Synthesis, 2017, 49: 3377–3393
Garbe M, Junge K, Beller M. Eur J Org Chem, 2017, 2017(30): 4344–4362
Espinosa-Jalapa NA, Nerush A, Shimon LJW, Leitus G, Avram L, Ben-David Y, Milstein D. Chem Eur J, 2017, 23: 5934–5938
van Putten R, Uslamin EA, Garbe M, Liu C, Gonzalez-de-Castro A, Lutz M, Junge K, Hensen EJM, Beller M, Lefort L, Pidko EA. Angew Chem Int Ed, 2017, 56: 7531–7534
Papa V, Cabrero-Antonino JR, Alberico E, Spanneberg A, Junge K, Junge H, Beller M. Chem Sci, 2017, 8: 3576–3585
Glatz M, Stöger B, Himmelbauer D, Veiros LF, Kirchner K. ACS Catal, 2018, 8: 4009–4016
Yang W, Chernyshov IY, van Schendel RKA, Weber M, Müller C, Filonenko GA, Pidko EA. Nat Commun, 2021, 12: 12
Knölker HJ, Baum E, Goesmann H, Klauss R. Angew Chem Int Ed, 1999, 38: 2064–2066
Casey CP, Guan H. J Am Chem Soc, 2007, 129: 5816–5817
Sonnenberg JF, Coombs N, Dube PA, Morris RH. J Am Chem Soc, 2012, 134: 5893–5899
Tuck CO, Pérez E, Horváth IT, Sheldon RA, Poliakoff M. Science, 2012, 337: 695–699
Zuo W, Lough AJ, Li YF, Morris RH. Science, 2013, 342: 1080–1083
Zhang G, Vasudevan KV, Scott BL, Hanson SK. J Am Chem Soc, 2013, 135: 8668–8681
Gunanathan C, Milstein D. Science, 2013, 341: 1229712
Michlik S, Kempe R. Nat Chem, 2013, 5: 140–144
Jeletic MS, Mock MT, Appel AM, Linehan JC. J Am Chem Soc, 2013, 135: 11533–11536
Li Y, Yu S, Wu X, Xiao J, Shen W, Dong Z, Gao J. J Am Chem Soc, 2014, 136: 4031–4039
Yan T, Feringa BL, Barta K. Nat Commun, 2014, 5: 5602
Qu S, Dang Y, Song C, Wen M, Huang KW, Wang ZX. J Am Chem Soc, 2014, 136: 4974–4991
Chakraborty S, Dai H, Bhattacharya P, Fairweather NT, Gibson MS, Krause JA, Guan H. J Am Chem Soc, 2014, 136: 7869–7872
Gärtner D, Welther A, Rad BR, Wolf R, Jacobi von Wangelin A. Angew Chem Int Ed, 2014, 53: 3722–3726
Korstanje TJ, Ivar van der Vlugt J, Elsevier CJ, de Bruin B. Science, 2015, 350: 298–302
Deibl N, Kempe R. J Am Chem Soc, 2016, 138: 10786–10789
De Luca L, Mezzetti A. Angew Chem Int Ed, 2017, 56: 11949–11953
Yuwen J, Chakraborty S, Brennessel WW, Jones WD. ACS Catal, 2017, 7: 3735–3740
Yang W, Chernyshov IY, Weber M, Pidko EA, Filonenko GA. ACS Catal, 2022, 12: 10818–10825
Balaraman E, Khaskin E, Leitus G, Milstein D. Nat Chem, 2013, 5: 122–125
Elangovan S, Garbe M, Jiao H, Spannenberg A, Junge K, Beller M. Angew Chem Int Ed, 2016, 55: 15364–15368
Mastalir M, Glatz M, Pittenauer E, Allmaier G, Kirchner K. J Am Chem Soc, 2016, 138: 15543–15546
Elangovan S, Neumann J, Sortais JB, Junge K, Darcel C, Beller M. Nat Commun, 2016, 7: 12641
Peña-López M, Piehl P, Elangovan S, Neumann H, Beller M. Angew Chem Int Ed, 2016, 55: 14967–14971
Owen AE, Preiss A, McLuskie A, Gao C, Peters G, Bühl M, Kumar A. ACS Catal, 2022, 12: 6923–6933
Perez M, Elangovan S, Spannenberg A, Junge K, Beller M. Chem-SusChem, 2017, 10: 83–86
Deibl N, Kempe R. Angew Chem Int Ed, 2017, 56: 1663–1666
Mastalir M, Pittenauer E, Allmaier G, Kirchner K. J Am Chem Soc, 2017, 139: 8812–8815
Kumar A, Espinosa-Jalapa NA, Leitus G, Diskin-Posner Y, Avram L, Milstein D. Angew Chem Int Ed, 2017, 56: 14992–14996
Nguyen DH, Trivelli X, Capet F, Paul JF, Du-meignil F, Gauvin RM. ACS Catal, 2017, 7: 2022–2032
Chakraborty S, Das UK, Ben-David Y, Milstein D. J Am Chem Soc, 2017, 139: 11710–11713
Kar S, Goeppert A, Kothandaraman J, Prakash GKS. ACS Catal, 2017, 7: 6347–6351
Bertini F, Glatz M, Gorgas N, Stöger B, Peruzzini M, Veiros LF, Kirchner K, Gonsalvi L. Chem Sci, 2017, 8: 5024–5029
Zirakzadeh A, de Aguiar SRMM, Stöger B, Widhalm M, Kirchner K. ChemCatChem, 2017, 9: 1744–1748
Wang Y, Wang M, Li Y, Liu Q. Chem, 2021, 7: 1180–1223
Waiba S, Maiti M, Maji B. ACS Catal, 2022, 12: 3995–4001
Kandepi VVKM, Cardoso JMS, Peris E, Royo B. Organometallics, 2010, 29: 2777–2782
Dubey A, Nencini L, Fayzullin RR, Nervi C, Khusnutdinova JR. ACS Catal, 2017, 7: 3864–3868
Bruneau-Voisine A, Wang D, Dorcet V, Roisnel T, Darcel C, Sortais JB. Org Lett, 2017, 19: 3656–3659
Martínez-Ferraté O, Werlé C, Franciò G, Leitner W. ChemCatChem, 2018, 10: 4514–4518
Ganguli K, Shee S, Panja D, Kundu S. Dalton Trans, 2019, 48: 7358–7366
Zhang C, Hu B, Chen D, Xia H. Organometallics, 2019, 38: 3218–3226
Shvydkiy NV, Vyhivskyi O, Nelyubina YV, Perekalin DS. ChemCatChem, 2019, 11: 1602–1605
Wang L, Lin J, Sun Q, Xia C, Sun W. ACS Catal, 2021, 11: 8033–8041
Filonenko GA, van Putten R, Hensen EJM, Pidko EA. Chem Soc Rev, 2018, 47: 1459–1483
Liu C, Wang M, Xu Y, Li Y, Liu Q. Angew Chem Int Ed, 2022, 61: e202202814
Das K, Waiba S, Jana A, Maji B. Chem Soc Rev, 2022, 51: 4386–4464
Torres-Calis A, García JJ. ACS Omega, 2022, 7: 37008–37038
Zhang C, Liang Z, Jia X, Wang M, Zhang G, Hu ML. Chem Commun, 2020, 56: 14215–14218
See the Supporting Information for details
CCDC: 2204257 (MnN10Br2), 2204258 (MnN10Cl2), 2204256 (D3), and 2204263 (D16) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre viahttp://www.ccdc.cam.ac.uk/data_request/cif
Fu S, Shao Z, Wang Y, Liu Q. J Am Chem Soc, 2017, 139: 11941–11948
Guillena G, Ramón DJ, Yus M. Chem Rev, 2010, 110: 1611–1641
Kallmeier F, Kempe R. Angew Chem Int Ed, 2018, 57: 46–60
Zhang C, Liang Q, Yang W, Zhang G, Hu M, Zhang G. Green Chem, 2022, 24: 7368–7375
Zhang J, Chen P, Yuan B, Ji W, Cheng Z, Qiu X. Science, 2013, 342: 611–614
Zhao D, Huang J, Zhong Y, Li K, Zhang L, Cai J. Adv Funct Mater, 2016, 26: 6279–6287
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22002067, 22202228), the Hundred-Talent Program of the Chinese Academy of Sciences, the Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province (20220052), the Science and Technology Project of Shanxi Province (202103021223457), and the State Key Laboratory of Coal Conversion, Institute of Coal Chemistry, Chinese Academy of Sciences (2021BWZ011).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of interest
The authors declare no conflict of interest.
Supporting information
The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic supplementary material
11426_2022_1576_MOESM1_ESM.pdf
Supporting Information for: Triazole Backbone Ligand in an Unprecedented Efficient Manganese Catalyst for Use in Transfer Hydrogenation
Rights and permissions
About this article
Cite this article
Liang, Q., Zhang, C., Wang, F. et al. Triazole backbone ligand in an unprecedented efficient manganese catalyst for use in transfer hydrogenation. Sci. China Chem. 66, 2028–2036 (2023). https://doi.org/10.1007/s11426-022-1576-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1576-5