Abstract
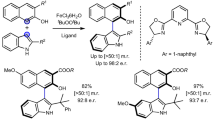
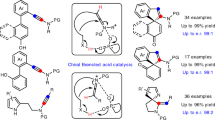
A highly efficient enantioselective construction of heterobiaryl N-oxides was developed. A series of axially chiral heterobiaryl N-oxides were generated via the cascade reaction of aminobenzamides with heterobiaryl aldehydes in the presence of chiral phosphoric acids. A number of atropisomers were afforded in moderate to good yields with excellent enantioselectivities and diastereoselectivities. Preliminary results demonstrate that the heterobiaryl N-oxides can be utilized as efficient chiral ligands in asymmetric catalysis.

Similar content being viewed by others
References
Bringmann G, Gulder T, Gulder TAM, Breuning M. Chem Rev, 2011, 111: 563–639
Kozlowski MC, Morgan BJ, Linton EC. Chem Soc Rev, 2009, 38: 3193–3207
Smyth JE, Butler NM, Keller PA. Nat Prod Rep, 2015, 32: 1562–1583
Zask A, Murphy J, Ellestad GA. Chirality, 2013, 25: 265–274
Pu L. Acc Chem Res, 2012, 45: 150–163
Xue M, Li B, Qiu S, Chen B. Mater Today, 2016, 19: 503–515
Collins BSL, Kistemaker JCM, Otten E, Feringa BL. Nat Chem, 2016, 8: 860–866
Erbas-Cakmak S, Leigh DA, McTernan CT, Nussbaumer AL. Chem Rev, 2015, 115: 10081–10206
Qi LW, Mao JH, Zhang J, Tan B. Nat Chem, 2018, 10: 58–64
Jiang PY, Fan KF, Li S, Xiang SH, Tan B. Nat Commun, 2021, 12: 2384
Zhang HH, Wang CS, Li C, Mei GJ, Li Y, Shi F. Angew Chem Int Ed, 2017, 56: 116–121
Howard RH, Alonso-Moreno C, Broomfield LM, Hughes DL, Wright JA, Bochmann M. Dalton Trans, 2009, 8667
Rokade BV, Guiry PJ. J Org Chem, 2019, 84: 5763–5772
Rokade BV, Guiry PJ. ACS Catal, 2017, 7: 2334–2338
Akiyama T. Chem Rev, 2007, 107: 5744–5758
Li Y, Kwong F, Yu W, Chan A. Coord Chem Rev, 2007, 251: 2119–2144
Chen Y, Yekta S, Yudin AK. Chem Rev, 2003, 103: 3155–3212
Cheng D, Shao Y. Adv Synth Catal, 2020, 362: 3081–3099
Pais VF, Ramírez-López P, Romero-Arenas A, Collado D, Nájera F, Pérez-Inestrosa E, Fernández R, Lassaletta JM, Ros A, Pischel U. J Org Chem, 2016, 81: 9605–9611
Malkov AV, Dufková L, Farrugia L, Kocovský P. Angew Chem Int Ed, 2003, 42: 3674–3677
Malkov AV, Ramírez-López P, Biedermannová(née Bendová) L, Rulíšek L, Dufková L, Kotora M, Zhu F, Kočovský P. J Am Chem Soc, 2008, 130: 5341–5348
Malkov AV, Kočovský P. Eur J Org Chem, 2007, 2007(1): 29–36
Nakajima M, Saito M, Shiro M, Hashimoto S. J Am Chem Soc, 1998, 120: 6419–6420
Shimada T, Kina A, Ikeda S, Hayashi T. Org Lett, 2002, 4: 2799–2801
Shimada T, Kina A, Hayashi T. J Org Chem, 2003, 68: 6329–6337
Murray JI, Flodén NJ, Bauer A, Fessner ND, Dunklemann DL, Bob-Egbe O, Rzepa HS, Bürgi T, Richardson J, Spivey AC. Angew Chem Int Ed, 2017, 56: 5760–5764
Xie MS, Shan M, Li N, Chen YG, Wang XB, Cheng X, Tian Y, Wu XX, Deng Y, Qu GR, Guo HM. ACS Catal, 2022, 12: 877–891
Xie MS, Huang B, Li N, Tian Y, Wu XX, Deng Y, Qu GR, Guo HM. J Am Chem Soc, 2020, 142: 19226–19238
Xie MS, Zhang YF, Shan M, Wu XX, Qu GR, Guo HM. Angew Chem Int Ed, 2019, 58: 2839–2843
Derdau V, Laschat S, Hupe E, König WA, Dix I, Jones PG. Eur J Inorg Chem, 1999, 1999: 1001–1007
Karayannis N, Pytlewski L L, Mikulski CM. Coord Chem Rev, 1973, 11: 93–159
Nakajima M, Sasaki Y, Shiro M, Hashimoto S. Tetrahedron-Asymmetry, 1997, 8: 341–344
Nakajima M, Sasaki Y, Iwamoto H, Hashimoto S. Tetrahedron Lett, 1998, 39: 87–88
Staniland S, Adams RW, McDouall JJW, Maffucci I, Contini A, Grainger DM, Turner NJ, Clayden J. Angew Chem Int Ed, 2016, 55: 10755–10759
Zheng J, You SL. Angew Chem Int Ed, 2014, 53: 13244–13247
Ma C, Jiang F, Sheng FT, Jiao Y, Mei GJ, Shi F. Angew Chem Int Ed, 2019, 58: 3014–3020
Guo D, Zhang J, Zhang B, Wang J. Org Lett, 2018, 20: 6284–6288
Carmona JA, Rodríguez-Franco C, López-Serrano J, Ros A, Iglesias-Sigüenza J, Fernández R, Lassaletta JM, Hornillos V. ACS Catal, 2021, 11: 4117–4124
Mori K, Itakura T, Akiyama T. Angew Chem Int Ed, 2016, 55: 11642–11646
Zhang J, Wang J. Angew Chem Int Ed, 2018, 57: 465–469
Hornillos V, Carmona JA, Ros A, Iglesias-Sigüenza J, López-Serrano J, Fernández R, Lassaletta JM. Angew Chem Int Ed, 2018, 57: 3777–3781
Hong X, Guo J, Liu J, Cao W, Wei C, Zhang Y, Zhang X, Fu Z. Sci China Chem, 2022, 65: 905–911
Akiyama T, Itoh J, Yokota K, Fuchibe K. Angew Chem Int Ed, 2004, 43: 1566–1568
Uraguchi D, Terada M. J Am Chem Soc, 2004, 126: 5356–5357
Parmar D, Sugiono E, Raja S, Rueping M. Chem Rev, 2014, 114: 9047–9153
Cheng X, Vellalath S, Goddard R, List B. J Am Chem Soc, 2008, 130: 15786–15787
Rueping M, Antonchick AP, Sugiono E, Grenader K. Angew Chem Int Ed, 2009, 48: 908–910
Huang D, Li X, Xu F, Li L, Lin X. ACS Catal, 2013, 3: 2244–2247
CCDC 2167850 (3al) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre viahttps://www.ccdc.cam.ac.uk/data_request/cif
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21871160, 21672121, 22071130) and the fellowship of Tsinghua-Peking Centre for Life Sciences (CLS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at https://chem.scichina.com and https://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Yuan, X., Wang, J. Atropoenantioselective synthesis of heterobiaryl N-oxides via dynamic kinetic resolution. Sci. China Chem. 65, 2512–2516 (2022). https://doi.org/10.1007/s11426-022-1402-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1402-9