Abstract
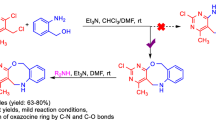
Here, we describe a general and modular strategy for the rapid assembly of benzo[c]phenanthridine (BCP) derivatives using homogeneous gold catalysis. Notably, in contrast to traditional methods based on the specially preformed substrates that have an inherent preference for the formation of this class of compounds with limited flexibility, this protocol is achieved via a selectively intramolecular cascade of a diazo-tethered alkyne and subsequently an intermolecular cyclization with a nitrile to facilitate the successive C-N and C-C bonds formation. This methodology uses readily available nitriles as the nitrogen source to deliver the products in good yield with excellent functional group compatibility. A preliminary anti-tumor activity study of these generated products exhibits high anticancer potency against five tumor cell lines, including HeLa, Mel624, SW-480, 8505C, LAN-1. Besides, we report a catalyst-controlled intermolecular cycloaddition/intramolecular insertion of the substrate with a fulvene to provide fused polycarbocycles containing a seven-membered ring.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
13 September 2022
Funding note is missing during initial upload.
20 September 2022
An Erratum to this paper has been published: https://doi.org/10.1007/s11426-022-1393-1
References
Janin YL, Croisy A, Riou JF, Bisagni E. J Med Chem, 1993, 36: 3686–3692
Lynch MA, Duval O, Sukhanova A, Devy J, MacKay SP, Waigh RD, Nabiev I. Bioorg Med Chem Lett, 2001, 11: 2643–2646
Kumar GS, Hazra S. RSC Adv, 2014, 4: 56518–56531
Nakanishi T, Suzuki M, Saimoto A, Kabasawa T. J Nat Prod, 1999, 62: 864–867
Castillo D, Sauvain M, Rivaud M, Jullian V. Planta Med, 2014, 80: 902–906
Wang Z, Jiang W, Zhang Z, Qian M, Du B. J Ethnopharmacol, 2012, 144: 145–150
Hamoud R, Reichling J, Wink M. J Pharmacy Pharmacol, 2015, 67: 264–273
Krane BD, Fagbule MO, Shamma M, Gözler B. J Nat Prod, 1984, 47: 1–43
Candito DA, Lautens M. Angew Chem Int Ed, 2009, 48: 6713–6716
Fu WC, Wang Z, Chan WTK, Lin Z, Kwong FY. Angew Chem Int Ed, 2017, 56: 7166–7170
Jiang H, An X, Tong K, Zheng T, Zhang Y, Yu S. Angew Chem Int Ed, 2015, 54: 4055–4059
Zhao HB, Liu ZJ, Song J, Xu HC. Angew Chem Int Ed, 2017, 56: 12732–12735
Zhao HB, Xu P, Song J, Xu HC. Angew Chem Int Ed, 2018, 57: 15153–15156
Tietze LF. Chem Rev, 1996, 96: 115–136
Wasilke JC, Obrey SJ, Baker RT, Bazan GC. Chem Rev, 2005, 105: 1001–1020
Nicolaou KC, Edmonds DJ, Bulger PG. Angew Chem Int Ed, 2006, 45: 7134–7186
Lu LQ, Chen JR, Xiao WJ. Acc Chem Res, 2012, 45: 1278–1293
Ardkhean R, Caputo DFJ, Morrow SM, Shi H, Xiong Y, Anderson EA. Chem Soc Rev, 2016, 45: 1557–1569
Tietze LF, Haunert F. Stimulating Concepts in Chemistry. Shibasaki M, Stoddart JF, Vögtle F, Eds. Weinheim: Wiley-VCH, 2000. 39–64
Nicolaou KC, Montagnon T, Snyder SA. Chem Commun, 2003, 1: 551–564
Hoye TR, Dinsmore CJ. J Am Chem Soc, 1991, 113: 4343–4345
Padwa A, Xu SL. J Am Chem Soc, 1992, 114: 5881–5882
Padwa A, Weingarten MD. Chem Rev, 1996, 96: 223–270
Padwa A. J Organomet Chem, 2000, 610: 88–101
Gulevich AV, Dudnik AS, Chernyak N, Gevorgyan V. Chem Rev, 2013, 113: 3084–3213
Archambeau A, Miege F, Meyer C, Cossy J. Acc Chem Res, 2015, 48: 1021–1031
Pei C, Zhang C, Qian Y, Xu X. Org Biomol Chem, 2018, 16: 8677–8685
Padwa A, Kinder FR. J Org Chem, 1993, 58: 21–28
Chuprakov S, Hwang FW, Gevorgyan V. Angew Chem Int Ed, 2007, 46: 4757–4759
Cambeiro F, López S, Varela JA, Saá C. Angew Chem Int Ed, 2012, 51: 723–727
Chattopadhyay B, Gevorgyan V. Angew Chem Int Ed, 2012, 51: 862–872
González-Rodríguez C, Suárez JR, Varela JA, Saá C. Angew Chem Int Ed, 2015, 54: 2724–2728
Le PQ, May JA. J Am Chem Soc, 2015, 137: 12219–12222
Zhang C, Chang S, Qiu L, Xu X. Chem Commun, 2016, 52: 12470–12473
Panne P, Fox JM. J Am Chem Soc, 2007, 129: 22–23
Park EJ, Kim SH, Chang S. J Am Chem Soc, 2008, 130: 17268–17269
Briones JF, Davies HML. J Am Chem Soc, 2012, 134: 11916–11919
Zhang C, Li H, Pei C, Qiu L, Hu W, Bao X, Xu X. ACS Catal, 2019, 9: 2440–2447
Zhou L, Liu Y, Zhang Y, Wang J. Beilstein J Org Chem, 2011, 7: 631–637
Zhang C, Hong K, Pei C, Zhou S, Hu W, Hashmi ASK, Xu X. Nat Commun, 2021, 12: 1182
Witham CA, Mauleón P, Shapiro ND, Sherry BD, Toste FD. J Am Chem Soc, 2007, 129: 5838–5839
Qiu H, Srinivas HD, Zavalij PY, Doyle MP. J Am Chem Soc, 2016, 138: 1808–1811
Jin H, Huang L, Xie J, Rudolph M, Rominger F, Hashmi ASK. Angew Chem Int Ed, 2016, 55: 794–797
Tian X, Song L, Rudolph M, Rominger F, Oeser T, Hashmi ASK. Angew Chem Int Ed, 2019, 58: 3589–3593
Padwa A, Chiacchio U, Fairfax DJ, Kassir JM, Litrico A, Semones MA, Xu SL. J Org Chem, 1993, 58: 6429–6437
Maas G. Angew Chem Int Ed, 2009, 48: 8186–8195
Dao HT, Baran PS. Angew Chem Int Ed, 2014, 53: 14382–14386
Wang HX, Wan Q, Wu K, Low KH, Yang C, Zhou CY, Huang JS, Che CM. J Am Chem Soc, 2019, 141: 9027–9046
Wang HX, Richard Y, Wan Q, Zhou CY, Che CM. Angew Chem Int Ed, 2020, 59: 1845–1850
Thai KM, Bui QH, Tran TD, Huynh TNP. Molecules, 2012, 17: 5690–5712
He W, Li C, Zhang L. J Am Chem Soc, 2011, 133: 8482–8485
Xiao Y, Zhang L. Org Lett, 2012, 14: 4662–4665
Straub BF. Chem Commun, 2004, 1726
Zhang C, Wang G, Zhan L, Yang X, Wang J, Wei Y, Xu S, Shi M, Zhang J. ACS Catal, 2020, 10: 6682–6690
Funding
Funding note Open access funding provided by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Conflict of interest
The authors declare no conflict of interest.
Supporting information The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/
The online version of the original article can be found at https://doi.org/10.1007/s11426-022-1393-1
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, C., Chen, Q., Wang, L. et al. Practical and modular construction of benzo[c]phenanthridine compounds. Sci. China Chem. 65, 1338–1346 (2022). https://doi.org/10.1007/s11426-022-1273-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1273-7