Abstract
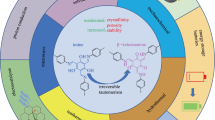
We report the synthesis of covalent organic framework (COF) crystals with organic Lewis acid instead of the conventional use of Brønsted acid or inorganic Lewis acid. Specifically, tris(pentafluorophenyl)borane was applied for the growth of seven imine COFs: TAPB-PDA-, TAPB-2,5-DMTA-, TAPB-2,3-DMTA-, TAPT-PDA-, TAPT-2,5-DMTA-, TAPT-2,3-DMTA-COF with hcb topology and varied in functional groups, as well as a new one, COF-820, with sql topology. All these COFs were obtained at room temperature. Their high crystallinity and porosity demonstrate the versatility of the organic Lewis acid as a catalyst. Bulky organic Lewis acid was found critical for the production of COF-820, while its absence resulted in the formation of a different COF, 4PE-1P-COF, with kgm topology using the same building blocks. Such steric effect, typical for organic catalysts, provides a new way to regulate the topology of COFs and their future design.

Similar content being viewed by others
Change history
20 September 2022
An Erratum to this paper has been published: https://doi.org/10.1007/s11426-022-1393-1
References
Diercks CS, Yaghi OM. Science, 2017, 355: eaal1585
Ding SY, Wang W. Chem Soc Rev, 2013, 42: 548–568
Guan X, Chen F, Fang Q, Qiu S. Chem Soc Rev, 2020, 49: 1357–1384
Sun T, Wei L, Chen Y, Ma Y, Zhang YB. J Am Chem Soc, 2019, 141: 10962–10966
Guan Q, Wang GB, Zhou LL, Li WY, Dong YB. Nanoscale Adv, 2020, 2: 3656–3733
Jiang J, Zhao Y, Yaghi OM. J Am Chem Soc, 2016, 138: 3255–3265
Kandambeth S, Dey K, Banerjee R. J Am Chem Soc, 2019, 141: 1807–1822
Matsumoto M, Dasari RR, Ji W, Feriante CH, Parker TC, Marder SR, Dichtel WR. J Am Chem Soc, 2017, 139: 4999–5002
Liu Y, Zhu Y, Alahakoon SB, Egap E. ACS Mater Lett, 2020, 2: 1561–1566
Giuseppone N, Schmitt JL, Schwartz E, Lehn JM. J Am Chem Soc, 2005, 127: 5528–5539
Satchell DPN, Satchell RS. Q Rev Chem Soc, 1971, 25: 171–199
Ashley AE, Herrington TJ, Wildgoose GG, Zaher H, Thompson AL, Rees NH, Krämer T, O’Hare D. J Am Chem Soc, 2011, 133: 14727–14740
Beckett MA, Strickland GC, Holland JR, Sukumar Varma K. Polymer, 1996, 37: 4629–4631
Braunschweig H, Krummenacher I, Légaré MA, Matler A, Radacki K, Ye Q. J Am Chem Soc, 2017, 139: 1802–1805
Légaré MA, Pranckevicius C, Braunschweig H. Chem Rev, 2019, 119: 8231–8261
Welch GC, San Juan RR, Masuda JD, Stephan DW. Science, 2006, 314: 1124–1126
Welch GC, Stephan DW. J Am Chem Soc, 2007, 129: 1880–1881
Stephan DW. J Am Chem Soc, 2015, 137: 10018–10032
Zhou TY, Xu SQ, Wen Q, Pang ZF, Zhao X. J Am Chem Soc, 2014, 136: 15885–15888
Ascherl L, Sick T, Margraf JT, Lapidus SH, Calik M, Hettstedt C, Karaghiosoff K, Döblinger M, Clark T, Chapman KW, Auras F, Bein T. Nat Chem, 2016, 8: 310–316
Smith BJ, Overholts AC, Hwang N, Dichtel WR. Chem Commun, 2016, 52: 3690–3693
Gomes R, Bhanja P, Bhaumik A. Chem Commun, 2015, 51: 10050–10053
Wang K, Kang X, Yuan C, Han X, Liu Y, Cui Y. Angew Chem Intl Ed, 2021, 60: 19466–19476
Wang X, Han X, Cheng C, Kang X, Liu Y, Cui Y. J Am Chem Soc, 2022, 144: 7366–7373
Pang ZF, Zhou TY, Liang RR, Qi QY, Zhao X. Chem Sci, 2017, 8: 3866–3870
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22025106, 21971199), the National Key Research and Development Project (2018YFA0704000) and the Innovation Team of Wuhan University (2042017kf0232).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors declare no conflict of interest.
Supporting information
The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
The online version of the original article can be found at https://doi.org/10.1007/s11426-022-1393-1
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Shi, X., Yi, L. & Deng, H. Covalent organic frameworks catalyzed by organic Lewis acid. Sci. China Chem. 65, 1315–1320 (2022). https://doi.org/10.1007/s11426-022-1272-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1272-5