Abstract
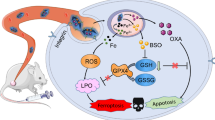
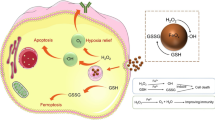
Overexpression of glutathione (GSH) in tumor cells greatly inhibits the therapy effect of traditional ferroptosis inducers; thus, control of the GSH level is an important way to improve the efficacy of ferroptosis. Herein, a kind of nanomotor based on metal organic framework material NH2-MIL-101 is constructed, in which polyethylene glycol (PEG) and glutathione hydrolase γ-glutamyltransferase (GGT) are asymmetrically modified to obtain mPEG@MIL-101@GGT nanomotors (PMG NMs). The nanomotor proposed in this article can be induced by overexpressing GSH in tumors to form chemotactic effects through the specific affinity between enzymes and substrates. Results indicate that the tail structure provided by PEG and the affinity between GGT and GSH can enable the stable chemotaxis behavior of nanomotors in a complex environment, thus enriching and penetrating deeply at the tumor site. In addition, after loading the ferroptosis inducer Erastin, the system shows a highly effective induction effect of tumor ferroptosis. Erastin in the system can effectively inhibit the synthesis of GSH, and PMG NMS can react with GSH through Fe3+ and GGT to promote GSH depletion. The produced Fe2+ can generate excessive reactive oxygen species through Fenton reaction, which further promotes the death of tumor cells. Meantime, the chemotaxis behavior of the nanomotors based on the endogenous biochemical reaction of GGT-catalyzed GSH hydrolysis can endow nanomotors with the enhanced delivery and penetration ability in tumors, thus collaboratively enhancing the ferroptosis effect. This strategy designed according to the physiological characteristics of tumors has good biosafety and treatment effect, providing new perspectives for micro/nanomotor and tumor treatment.

Similar content being viewed by others
References
Wu J, Minikes AM, Gao M, Bian H, Li Y, Stockwell BR, Chen ZN, Jiang X. Nature, 2019, 572: 402–406
Yi J, Zhu J, Wu J, Thompson CB, Jiang X. Proc Natl Acad Sci USA, 2020, 117: 31189–31197
Doll S, Freitas FP, Shah R, Aldrovandi M, da Silva MC, Ingold I, Goya Grocin A, Xavier da Silva TN, Panzilius E, Scheel CH, Mourão A, Buday K, Sato M, Wanninger J, Vignane T, Mohana V, Rehberg M, Flatley A, Schepers A, Kurz A, White D, Sauer M, Sattler M, Tate EW, Schmitz W, Schulze A, O’Donnell V, Proneth B, Popowicz GM, Pratt DA, Angeli JPF, Conrad M. Nature, 2019, 575: 693–698
Shen Z, Song J, Yung BC, Zhou Z, Wu A, Chen X. Adv Mater, 2018, 30: 1704007
Zhou Z, Wu H, Yang R, Xu A, Zhang Q, Dong J, Qian C, Sun M. Sci Adv, 2020, 6: eabc4373
Sun Y, Zheng Y, Wang C, Liu Y. Cell Death Dis, 2018, 9: 753
Zheng J, Conrad M. Cell Metab, 2020, 32: 920–937
Zheng DW, Lei Q, Zhu JY, Fan JX, Li CX, Li C, Xu Z, Cheng SX, Zhang XZ. Nano Lett, 2017, 17: 284–291
Du JZ, Li HJ, Wang J. Acc Chem Res, 2018, 51: 2848–2856
Wan M, Chen H, Wang Q, Niu Q, Xu P, Yu Y, Zhu T, Mao C, Shen J. Nat Commun, 2019, 10: 966
Meng F, Hennink WE, Zhong Z. Biomaterials, 2009, 30: 2180–2198
Wang L, Hortelão AC, Huang X, Sánchez S. Angew Chem Int Ed, 2019, 58: 7992–7996
Wan M, Li T, Chen H, Mao C, Shen J. Angew Chem Int Ed, 2021, 60: 13158–13176
Tang J, Yao C, Gu Z, Jung S, Luo D, Yang D. Angew Chem Int Ed, 2020, 59: 2490–2495
Schmidt CK, Medina-Sánchez M, Edmondson RJ, Schmidt OG. Nat Commun, 2020, 11: 5618
Wan MM, Chen H, Da Wang Z, Liu ZY, Yu YQ, Li L, Miao ZY, Wang XW, Wang Q, Mao C, Shen J, Wei J. Adv Sci, 2021, 8: 2002525
Wan M, Liu Z, Li T, Chen H, Wang Q, Chen T, Tao Y, Mao C. Angew Chem Int Ed, 2021, 60: 16139–16148
Chen H, Shi T, Wang Y, Liu Z, Liu F, Zhang H, Wang X, Miao Z, Liu B, Wan M, Mao C, Wei J. J Am Chem Soc, 2021, 143: 12025–12037
Zhou Q, Shao S, Wang J, Xu C, Xiang J, Piao Y, Zhou Z, Yu Q, Tang J, Liu X, Gan Z, Mo R, Gu Z, Shen Y. Nat Nanotechnol, 2019, 14: 799–809
Wang G, Zhou Z, Zhao Z, Li Q, Wu Y, Yan S, Shen Y, Huang P. ACS Nano, 2020, 14: 4890–4904
Sengupta S, Dey KK, Muddana HS, Tabouillot T, Ibele ME, Butler PJ, Sen A. J Am Chem Soc, 2013, 135: 1406–1414
Sengupta S, Spiering MM, Dey KK, Duan W, Patra D, Butler PJ, Astumian RD, Benkovic SJ, Sen A. ACS Nano, 2014, 8: 2410–2418
Horcajada P, Chalati T, Serre C, Gillet B, Sebrie C, Baati T, Eubank JF, Heurtaux D, Clayette P, Kreuz C, Chang JS, Hwang YK, Marsaud V, Bories PN, Cynober L, Gil S, Férey G, Couvreur P, Gref R. Nat Mater, 2010, 9: 172–178
Huo M, Wang L, Chen Y, Shi J. Nat Commun, 2017, 8: 357
Lee TC, Alarcón-Correa M, Miksch C, Hahn K, Gibbs JG, Fischer P. Nano Lett, 2014, 14: 2407–2412
Bockuviene A, Balciunaite J, Slavuckyte K, Zaliauskiene L, Vareikis A, Makuska R. J Polym Res, 2015, 23: 10
Ji Y, Lin X, Wu Z, Wu Y, Gao W, He Q. Angew Chem Int Ed, 2019, 58: 12200–12205
Hortelao AC, Simó C, Guix M, Guallar-Garrido S, Julián E, Vilela D, Rejc L, Ramos-Cabrer P, Cossío U, Gómez-Vallejo V, Patiño T, Llop J, Sánchez S. Sci Robot, 2021, 6: eabd2823
Glidden M, Muschol M. J Phys Chem C, 2012, 116: 8128–8137
Xuan M, Wu Z, Shao J, Dai L, Si T, He Q. J Am Chem Soc, 2016, 138: 6492–6497
Cheng R, Feng F, Meng F, Deng C, Feijen J, Zhong Z. J Control Release, 2011, 152: 2–12
Circu ML, Stringer S, Rhoads CA, Moyer MP, Aw TY. Biochem Pharmacol, 2009, 77: 76–85
Brechbuhl HM, Gould N, Kachadourian R, Riekhof WR, Voelker DR, Day BJ. J Biol Chem, 2010, 285: 16582–16587
Franco R, Schoneveld OJ, Pappa A, Panayiotidis MI. Arch Physiol Biochem, 2007, 113: 234–258
Chaiswing L, Zhong W, Liang Y, Jones DP, Oberley TD. Free Radical Biol Med, 2012, 52: 452–461
Shi L, Wu W, Duan Y, Xu L, Xu Y, Hou L, Meng X, Zhu X, Liu B. Angew Chem Int Ed, 2020, 59: 19168–19174
Cai L, Yang C, Jia W, Liu Y, Xie R, Lei T, Yang Z, He X, Tong R, Gao H. Adv Funct Mater, 2020, 30: 1909999
Martínez-Menárguez JA. ISRN Cell Biol, 2013, 2013: 1–15
Iversen TG, Frerker N, Sandvig K. J Nanobiotechnol, 2012, 10: 33
Chen Y, Li ZH, Pan P, Hu JJ, Cheng SX, Zhang XZ. Adv Mater, 2020, 32: 2001452
Ma X, Ren X, Guo X, Fu C, Wu Q, Tan L, Li H, Zhang W, Chen X, Zhong H, Meng X. Biomaterials, 2019, 214: 119223
Battaglia AM, Chirillo R, Aversa I, Sacco A, Costanzo F, Biamonte F. Cells, 2020, 9: 1505
Li Y, Xia J, Shao F, Zhou Y, Yu J, Wu H, Du J, Ren X. Biochem Biophysl Res Commun, 2021, 534: 877–884
Niedernhofer LJ, Daniels JS, Rouzer CA, Greene RE, Marnett LJ. J Biol Chem, 2003, 278: 31426–31433
Yu C, Zhou Z, Wang J, Sun J, Liu W, Sun Y, Kong B, Yang H, Yang S. J Hazard Mater, 2015, 283: 519–528
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22175096), the Social Development Project of Jiangsu Natural Science Foundation (BE2019744), the Collaborative Innovation Center of Biomedical Functional Materials, and the Priority Academic Program Development of Jiangsu Higher Education Institution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Supplementary material, approximately 18.0 MB.
Supplementary material, approximately 23.8 MB.
Supplementary material, approximately 2.01 MB.
Supplementary material, approximately 4.20 MB.
Supplementary material, approximately 472 KB.
Supplementary material, approximately 4.50 MB.
Rights and permissions
About this article
Cite this article
Liu, Z., Li, T., Li, N. et al. GSH-induced chemotaxis nanomotors for cancer treatment by ferroptosis strategy. Sci. China Chem. 65, 989–1002 (2022). https://doi.org/10.1007/s11426-021-1208-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-021-1208-6