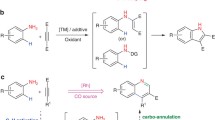
Abstract
A Rh(III)-catalyzed oxidative C–H/C–H cross-coupling between indoles and oxazoles with molecular oxygen as an oxidant has been developed to construct a variety of 2-(indol-2-yl)oxazoles. The chelation-assisted strategy endows this catalytic system with an excellent C2-site selectivity by introducing a pyrimidyl directing group. In addition, polycyclic pyridinium salts are also obtained via Rh(III)-catalyzed C–H activation/cyclization of 2-(indol-2-yl)oxazoles with alkynes.
Similar content being viewed by others
References
Cho SH, Kim JY, Kwak J, Chang S. Chem Soc Rev, 2011, 40: 5068–5083
Hirano K, Miura M. Chem Commun, 2012, 48: 10704–10714
Liu C, Yuan J, Gao M, Tang S, Li W, Shi R, Lei A. Chem Rev, 2015, 115: 12138–12204
Yang Y, Lan J, You J. Chem Rev, 2017, 117: doi: acs.chemrev.6b00567
Xi P, Yang F, Qin S, Zhao D, Lan J, Gao G, Hu C, You J. J Am Chem Soc, 2010, 132: 1822–1824
Mandal D, Yamaguchi AD, Yamaguchi J, Itami K. J Am Chem Soc, 2011, 133: 19660–19663
Han W, Mayer P, Ofial AR. Angew Chem Int Ed, 2011, 50: 2178–2182
Wang Z, Li K, Zhao D, Lan J, You J. Angew Chem Int Ed, 2011, 50: 5365–5369
Nishino M, Hirano K, Satoh T, Miura M. Angew Chem Int Ed, 2012, 51: 6993–6997
Kuhl N, Hopkinson MN, Glorius F. Angew Chem Int Ed, 2012, 51: 8230–8234
Odani R, Hirano K, Satoh T, Miura M. Angew Chem Int Ed, 2014, 53: 10784–10788
Cheng Y, Li G, Liu Y, Shi Y, Gao G, Wu D, Lan J, You J. J Am Chem Soc, 2016, 138: 4730–4738
Tan G, He S, Huang X, Liao X, Cheng Y, You J. Angew Chem Int Ed, 2016, 55: 10414–10418
Cheng Y, Wu Y, Tan G, You J. Angew Chem Int Ed, 2016, 55: 12275–12279
Ackermann L, Vicente R, Kapdi AR. Angew Chem Int Ed, 2009, 48: 9792–9826
Lyons TW, Sanford MS. Chem Rev, 2010, 110: 1147–1169
Yeung CS, Dong VM. Chem Rev, 2011, 111: 1215–1292
Kuhl N, Hopkinson MN, Wencel-Delord J, Glorius F. Angew Chem Int Ed, 2012, 51: 10236–10254
Emsley J. Nature’s Building Blocks: An A-Z Guide to the Elements. Oxford: Oxford University Press,2001. 297–304
Piera J, Bäckvall JE. Angew Chem Int Ed, 2008, 47: 3506–3523
Campbell AN, Stahl SS. Acc Chem Res, 2012, 45: 851–863
Wu W, Jiang H. Acc Chem Res, 2012, 45: 1736–1748
Shi Z, Zhang C, Tang C, Jiao N. Chem Soc Rev, 2012, 41: 3381–3430
Jin LQ, Lei AW. Sci China Chem, 2012, 55: 2027–2035
Chen WL, Gao YR, Mao S, Zhang YL, Wang YF, Wang YQ. Org Lett, 2012, 14: 5920–5923
Zhang G, Yang L, Wang Y, Xie Y, Huang H. J Am Chem Soc, 2013, 135: 8850–8853
Gigant N, Bäckvall JE. Org Lett, 2014, 16: 1664–1667
Huang X, Li X, Zou M, Song S, Tang C, Yuan Y, Jiao N. J Am Chem Soc, 2014, 136: 14858–14865
Warratz S, Kornhaaß C, Cajaraville A, Niepötter B, Stalke D, Ackermann L. Angew Chem Int Ed, 2015, 54: 5513–5517
Liu YH, Liu YJ, Yan SY, Shi BF. Chem Commun, 2015, 51: 11650–11653
Lu Y, Wang HW, Spangler JE, Chen K, Cui PP, Zhao Y, Sun WY, Yu JQ. Chem Sci, 2015, 6: 1923–1927
Shi Y, Wang Z, Cheng Y, Lan J, She Z, You J. Sci China Chem, 2015, 58: 1292–1296
Somei M, Yamada F. Nat Prod Rep, 2004, 21: 278–311
Kang JW, Moseley K, Maitlis PM. J Am Chem Soc, 1969, 91: 5970–5977
Ackermann L, Lygin AV. Org Lett, 2011, 13: 3332–3335
Cho SH, Kim JY, Lee SY, Chang S. Angew Chem Int Ed, 2009, 48: 9127–9130
(d) van Leusen AM, Hoogenboom BE, Siderius H. Tetrahedron Lett, 1972, 13: 2369–2372
Ding Z, Yoshikai N. Angew Chem Int Ed, 2012, 51: 4698–4701
Yang Y, Qiu X, Zhao Y, Mu Y, Shi Z. J Am Chem Soc, 2016, 138: 495–498
Yang Y, Li R, Zhao Y, Zhao D, Shi Z. J Am Chem Soc, 2016, 138: 8734–8737
Sun D, Li B, Lan J, Huang Q, You J. Chem Commun, 2016, 52: 3635–3638
Qin X, Liu H, Qin D, Wu Q, You J, Zhao D, Guo Q, Huang X, Lan J. Chem Sci, 2013, 4: 1964–1969
Li B, Lan J, Wu D, You J. Angew Chem Int Ed, 2015, 54: 14008–14012
Gandeepan P, Cheng CH. Chem Asian J, 2016, 11: 448–460
Sucunza D, Cuadro AM, Alvarez-Builla J, Vaquero JJ. J Org Chem, 2016, 81: 10126–10135
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21672154, 21372164). We thank the Comprehensive Training Platform of Specialized Laboratory, College of chemistry, Sichuan University, for NMR measurements and HRMS measurements.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, Y., Lan, J. Oxygen as an oxidant in rhodium(III) catalyzed oxidative C–H/C–H cross-coupling between indoles and oxazoles. Sci. China Chem. 61, 200–205 (2018). https://doi.org/10.1007/s11426-017-9053-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-017-9053-7