Abstract
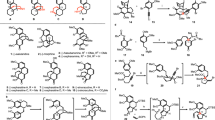
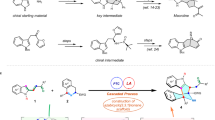
A proposed strategy for constructing the nine-membered azonane ring system embedded in the isotwistane framework of palhinine A has been preliminarily studied on the basis of an azidoketol fragmentation reaction together with a SmI2-mediated pinacol coupling. Albeit negative results for building the azonane ring scaffold via a tertiary azidoketol fragmentation, the present exploration strategically provided an endeavor to probe the synthetically challenging issue in the synthetic study of palhinine A.
Similar content being viewed by others
References
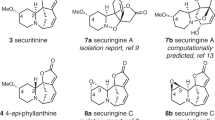
Zhao FW, Sun QY, Yang FM, Hu GW, Luo JF, Tang GH, Wang YH, Long CL. Org Lett, 2010, 12: 3922–3925
Dong LB, Gao X, Liu F, He J, Wu XD, Li Y, Zhao QS. Org Lett, 2013, 15: 3570–3573
Wang XJ, Li L, Yu SS, Ma SG, Qu J, Liu YB, Li Y, Wang Y, Tang W. Fitoterapia, 2013, 91: 74–81
Ayer WA. Nat Prod Rep, 1991, 8: 455–463
Ayer WA, Trifonov LS. Lycopodium alkaloids. In: Cordell GA, Brossi A, Eds. The Alkaloids: Chemistry and Pharmacology. Vol 45. San Diego: CA, 1994. 233–266
Kobayashi J, Morita H. The lycopodium alkaloids. In: Cordell GA, Eds. The Alkaloids: Chemistry and Biology. Vol 61. San Diego: CA, 2005. 1–57
Ma X, Gang DR. Nat Prod Rep, 2004, 21: 752–772
Hirasawa Y, Kobayashi J, Morita H. Heterocycles, 2009, 77: 679–729
Zhao C, Zheng H, Jing P, Fang B, Xie X, She X. Org Lett, 2012, 14: 2293–2295
Zhang GB, Wang FX, Du JY, Qu H, Ma XY, Wei MX, Wang CT, Li Q, Fan CA. Org Lett, 2012, 14: 3696–3699
Gaugele D, Maier ME. Synlett, 2013, 24: 955–958
Sizemore N, Rychnovsky SD. Org Lett, 2014, 16: 688–691
Zhang GB. Synthetic studies towards the lycopodium alkaloid palhinine A. Doctoral Degree. Lanzhou: Lanzhou University, 2013
Several attempts for the direct construction of the nine-membered azonane ring system using N-alkylation or Mitsunobu reaction failed at this stage.
Prantz K, Mulzer J. Chem Rev, 2010, 110: 3741–3766
Drahl MA, Manpadi M, Williams LJ. Angew Chem Int Ed, 2013, 52: 11222–11251
Pearson WH. J Heterocyclic Chem, 1996, 33: 1489–1496
Bräse S, Gil C, Knepper K, Zimmermann V. Angew Chem Int Ed, 2005, 44: 5188–5240
Lang S, Murphy JA. Chem Soc Rev, 2006, 35: 146–156
Nyfeler E, Renaud P. Chimia, 2006, 60: 276–284
Wrobleski A, Coombs TC, Huh CW, Li SW, Aubé J. Org React, 2012, 78: 1–320
Tanimoto H, Kakiuchi K. Nat Prod Commun, 2013, 8: 1021–1034
Soderquist JA. Aldrichimica Acta, 1991, 24: 15–23
Krief A, Laval AM. Chem Rev, 1999, 99: 745–778
Kagan HB. Tetrahedron, 2003, 59: 10351–10372
Beemelmanns C, Reissig HU. Chem Soc Rev, 2011, 40: 2199–2210
Szostak M, Procter DJ. Angew Chem Int Ed, 2011, 50: 7737–7739
The unpublished details on the synthesis of (±)-3 from the known compound (±)-2 will be reported in due course
CCDC 1450792 for (±)-8 and CCDC 1450793 for (±)-13 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif
Sasaki M, Murae T, Takahashi T. J Org Chem, 1990, 55: 528–540
Lu TJ, Lin CK. J Org Chem, 2011, 76: 1621–1633
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, FX., Zhang, PL., Wang, HB. et al. A strategic study towards constructing the nine-membered azonane ring system of palhinine A via an azidoketol fragmentation reaction. Sci. China Chem. 59, 1188–1196 (2016). https://doi.org/10.1007/s11426-016-0059-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-016-0059-7