Abstract
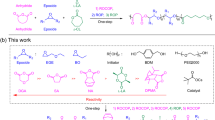
Multicomponent polymerizations have become powerful tools for the construction of sequence-defined polymers. Although the Passerini multicomponent reaction has been widely used in the synthesis of sequence-defined polymers, the tandem usage of the Passerini multicomponent reaction and other multicomponent reactions in one-pot for the synthesis of sequence-defined polymers has not been developed until now. In this contribution, we report the tandem usage of the Passerini three-component reaction and the three-component amine-thiol-ene conjugation reaction in one pot for the synthesis of sequence-defined polymers. The Passerini reaction between methacrylic acid, adipaldehyde, and 2-isocyanobutanoate was carried out, affording a new molecule containing two alkene units. Subsequently, an amine and a thiolactone were added to the reaction system, whereupon the three-component amine-thiol-ene conjugating reaction occurred to yield a sequence-defined polymer. This method offers more rapid access to sequence-defined polymers with high molecular diversity and complexity.
Similar content being viewed by others
References
Matyjaszewski K. Architecturally complex polymers with controlled heterogeneity. Science, 2011, 333: 1104–1105
Ge ZS, Zhou YM, Xu J, Liu HW, Chen DY, Liu SY. High-efficiency preparation of macrocyclic diblock copolymers via selective click reaction in micellar media. J Am Chem Soc, 2009, 131: 1628–1629
Yan JJ, Wang ZK, Lin XS, Hong CY, Liang HJ, Pan CY, You YZ. Polymerizing nonfluorescent monomers without incorporating any fluorescent agent produces strong fluorescent polymers. Adv Mater, 2012, 24: 5617–5624
Kakuchi R. Multicomponent reactions in polymer synthesis. Angew Chem Int Ed, 2014, 53: 46–48
Guan ZB, Cotts PM, McCord EF, McLain SJ. Chain walking: a new strategy to control polymer topology. Science, 1999, 283: 2059–2062
You YZ, Yu ZQ, Cui MM, Hong CY. Preparation of photoluminescent nanorings with controllable bioreducibility and stimuli-responsiveness. Angew Chem Int Ed, 2010, 49: 1099–1102
Lutz JF, Ouchi M, Liu DR, Sawamoto M. Sequence-controlled polymers. Science, 2013, 341: 628–636
Kreye O, Toth T, Meier MAR. Introducing multicomponent reactions to polymer science: Passerini reactions of renewable monomers. J Am Chem Soc, 2011, 133: 1790–1792
Li XJ, Qian YF, Liu T, Hu XL, Zhang GY, You YZ, Liu S. Amphiphilic multiarm star block copolymer-based multifunctional unimolecular micelles for cancer targeted drug delivery and MR imaging. Biomaterials, 2011, 32: 6595–6605
Deng XX, Cui Y, Du FS, Li ZC. Functional highly branched polymers from multicomponent polymerization (MCP) based on the ABC type Passerini reaction. Polym Chem, 2014, 5: 3316–3320
Hu XL, Liu GH, Li Y, Wang XR, Liu SY. Cell-penetrating hyperbranched polyprodrug amphiphiles for synergistic reductive milieu-triggered drug release and enhanced magnetic resonance signals. J Am Chem Soc, 2015, 137: 362–368
Hong CY, You YZ, Wu DC, Liu Y, Pan CY. Thermal control over the topology of cleavable polymers: from linear to hyperbranched structures. J Am Chem Soc, 2007, 129: 5354–5355
Yan DY, Muller AHE, Matyjaszewski K. Molecular parameters of hyperbranched polymers made by self-condensing vinyl polymerization. 2. Degree of branching. Macromolecules, 1997, 30: 7024–7033
Wang RB, Zhou LZ, Zhou YF, Li GL, Zhu XY, Gu HC, Jiang XL, Li HQ, Wu JL, He L, Guo XQ, Zhu BS, Yan DY. Synthesis and gene delivery of poly(amido amine)s with different branched architecture. Biomacromolecules, 2010, 11: 489–495
Yan DY, Zhou ZP. Molecular weight distribution of hyperbranched polymers generated from polycondensation of AB(2) type monomers in the presence of multifunctional core moieties. Macromolecules, 1999, 32: 819–824
Han J, Zheng YC, Zhao B, Li SP, Zhang YC, Gao C. Sequentially hetero-functional, topological polymers by step-growth thiolyne approach. Sci Rep, 2014, 4: 4387
Li SP, Han J, Gao C. High-density and hetero-functional group engineering of segmented hyperbranched polymers via click chemistry. Polym Chem, 2013, 4: 1774–1787
Han J, Zhao B, Gao YQ, Tang AJ, Gao C. Sequential click synthesis of hyperbranched polymers via the A(2)+CB2 approach. Polym Chem, 2011, 2: 2175–2178
Yu ZQ, Xu XM, Hong CY, Wu DC, You YZ. A Responsive hyperbranched polymer not only can self-immolate but also can self-cross-link. Macromolecules, 2014, 47: 4136–4143
Yan JJ, Hong CY, You YZ. An easy method to convert the topologies of macromolecules after polymerization. Macromolecules, 2011, 44: 1247–1251
Deng XX, Li L, Li ZL, Lv A, Du FS, Li ZC. Sequence regulated poly(ester-amide)s based on Passerini reaction. Acs Macro Lett, 2012, 1: 1300–1303
Li L, Lv A, Deng XX, Du FS, Li ZC. Facile synthesis of photo-cleavable polymers via Passerini reaction. Chem Commun, 2013, 49: 8549–8551
Lv A, Deng XX, Li L, Li ZL, Wang YZ, Du FS, Li ZC. Facile synthesis of multi-block copolymers containing poly(ester-amide) segments with an ordered side group sequence. Polym Chem, 2013, 4: 3659–3662
Li L, Deng XX, Li ZL, Du FS, Li ZC. Multifunctional photodegradable polymers for reactive micropatterns. Macromolecules, 2014, 47: 4660–4667
Yan JJ, Wang D, Wu DC, You YZ. Synthesis of sequence-ordered polymers via sequential addition of monomers in one pot. Chem Commun, 2013, 49: 6057–6059
Kan XW, Deng XX, Du FS, Li ZC. Concurrent oxidation of alcohols and the Passerini three-component polymerization for the synthesis of functional poly(ester amide)s. Macromol Chem Phys, 2014, 215: 2221–2228
Liu YJ, Gao M, Lam JWY, Hu RR, Tang BZ. Copper-catalyzed poly coupling of diynes, primary amines, and aldehydes: a new one-pot multicomponent polymerization tool to functional polymers. Macromolecules, 2014, 47: 4908–4919
Deng HQ, Hu RR, Zhao EG, Chan CYK, Lam JWY, Tang BZ. One-pot three-component tandem polymerization toward functional poly(arylene thiophenylene) with aggregation-enhanced emission characteristics. Macromolecules, 2014, 47: 4920–4929
Espeel P, Carrette LLG, Bury K, Capenberghs S, Martins JC, Du Prez FE, Madder A. Multifunctionalized sequence-defined oligomers from a single building block. Angew Chem Int Ed, 2013, 52: 13261–13264
Espeel P, Du Prez FE. One-pot multi-step reactions based on thiolactone chemistry: a powerful synthetic tool in polymer science. Eur Polym J, 2015, 62: 247–272
Espeel P, Goethals F, Driessen F, Nguyen LTT, Du Prez FE. One-pot, additive-free preparation of functionalized polyurethanes via aminethiol-ene conjugation. Polym Chem, 2013, 4: 2449–2456
Espeel P, Goethals F, Du Prez FE. One-pot multistep reactions based on thiolactones: extending the realm of thiol-ene chemistry in polymer synthesis. J Am Chem Soc, 2011, 133: 1678–1681
Yu L, Wang LH, Hu ZT, You YZ, Wu DC, Hong CY. Sequential Michael addition thiol-ene and radical-mediated thiol-ene reactions in one-pot produced sequence-ordered polymers. Polym Chem, 2015, 6: 1527–1532
Yan JJ, Sun JT, You YZ, Wu DC, Hong CY. Growing hyperbranched polymers using natural sunlight. Sci Rep, 2013, 3: 2841
Ma XP, Tang JB, Shen YQ, Fan MH, Tang HD, Radosz M. Facile synthesis of polyester dendrimers from sequential click coupling of asymmetrical monomers. J Am Chem Soc, 2009, 131: 14795–14803
Shanmuganathan K, Sankhagowit RK, Iyer P, Ellison CJ. Thiol-ene chemistry: a greener approach to making chemically and thermally stable fibers. Chem Mater, 2011, 23: 4726–4732
Zhang Z, You YZ, Wu DC, Hong CY. A novel multicomponent reaction and its application in sequence-ordered functional polymer synthesis. Polymer, 2015, 64: 221–226
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, L., Zhang, Z., Cheng, B. et al. Two tandem multicomponent reactions for the synthesis of sequence-defined polymers. Sci. China Chem. 58, 1734–1740 (2015). https://doi.org/10.1007/s11426-015-5448-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5448-0