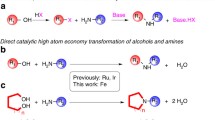
Abstract
An oxidative amidation of tertiary amines with carboxylic acids has been developed in the presence of FeCl3·6H2O as catalyst and oxygen as oxidant. A variety of tertiary amides were obtained in good to excellent yields from inexpensive and readily available reagents. The possible reaction pathways were investigated.
Similar content being viewed by others
References
Cupido T, Tulla-Puche J, Spengler J, Albericio F. The synthesis of naturally occuring peptides and their analogs. Curr Opin Drug Discovery Dev, 2007, 10: 768–783
Bode JW. Emerging methods in amide- and peptide-bond formation. Curr Opin Drug Discovery Dev, 2006, 9: 765–775
Humphrey JM, Chamberlin AR. Chemical synthesis of natural product peptides: coupling methods for the incorporation of noncoded amino acids into peptides. Chem Rev, 1997, 97: 2243–2266
Valeur E, Bradley M. Amide bond formation: beyond the myth of coupling reagents. Chem Soc Rev, 2009, 38: 606–631
Larock RC. Comprehensive Organic Transformations. New York: VCH, 1999
Gunanathan C, Yehoshoa BD, Milstein D. Direct synthesis of amides from alcohols and amines with liberation of H2. Science, 2007, 317: 790–792
Ghosh SC, Muthaiah S, Zhang Y, Xu X, Hong SH. Direct amide synthesis from alcohols and amines by phosphine-free ruthenium catalyst systems. Adv Synth Catal, 2009, 351: 2643–2649
Wang Y, Zhu D, Tang L, Wang S, Wang Z. Highly efficient amide synthesis from alcohols and amines by virtue of a water-soluble Gold/DNA catalyst. Angew Chem Int Ed, 2011, 50: 8917–8921
Soule JF, Miyamura H, Kobayashi S. Powerful amide synthesis from alcohols and amines under aerobic conditions catalyzed by gold or gold/iron, -nickel or -cobalt nanoparticles. J Am Chem Soc, 2011, 133: 18550–18553
Kegnæs S, Mielby J, Mentzel UV, Jensen T, Fristrup P, Riisager A. One-pot synthesis of amides by aerobic oxidative coupling of alcohols or aldehydes with amines using supported gold and base as catalysts. Chem Commun, 2012, 48: 2427–2429
Nakagawa K, Inoue H, Minami K. Oxidation with nickel peroxide. A new synthesis of amides from aldehydes or alcohols. Chem Commun, 1966: 17–18
Nakagawa K, Mineo S, Kawamura S, Horikawa M, Tokumoto T, Mori O. Oxidation with nickel peroxide. XII. Synthesis of nitriles from aldehydes. Synth Commun, 1979, 9: 529–531
Murahashi SI, Naota T, Saito E. Ruthenium-catalyzed amidation of nitriles with amines. A novel, facile route to amides and polyamides. J Am Chem Soc, 1986, 108: 7846–7847
Cobley CJ, van den Heuvel M, Abbadi A, de Vries JG. Platinum catalysed hydrolytic amidation of unactivated nitriles. Tetrahedron Lett, 2000, 41: 2467–2470
Allen CL, Lapkin AA, Williams JMJ. An iron-catalysed synthesis of amides from nitriles and amines. Tetrahedron Lett, 2009, 50: 4262–4264
Callens E, Burton AJ, Barrett AGM. Synthesis of amides using the Ritter reaction with bismuth triflate catalysis. Tetrahedron Lett, 2006, 47: 8699–8701
Martinelli JR, Clark TP, Watson DA, Munday RH, Buchwald SL. Palladium-catalyzed aminocarbonylation of aryl chlorides at atmospheric pressure: the dual role of sodium phenoxide. Angew Chem Int Ed, 2007, 46: 8460–8463
Brennfuhrer A, Neumann H, Beller M. Palladium-catalyzed carbonylation reactions of aryl halides and related compounds. Angew Chem Int Ed, 2009, 48: 4114–4133
Dang TT, Zhu Y, Ghosh SC, Chen A, Chai CLL, Seayad AM. Atmospheric pressure aminocarbonylation of aryl iodides using palladium nanoparticles supported on MOF-5. Chem Commun, 2012, 48: 1805–1807
Li Y, Jia F, Li Z. Iron-catalyzed oxidative amidation of tertiary amines with aldehydes. Chem Eur J, 2013, 19: 82–87
Mai WP, Song G, Yuan JW, Yang LR, Sun GC, Xiao YM, Mao P, Qu LB. nBu4NI-catalyzed unexpected amide bond formation between aldehydes and aromatic tertiary amines. RSC Adv, 2013, 3: 3869–3892
Grierson D. The polonovski reaction. In: Organic Reactions. Weinheim: John Wiley & Sons, Inc., 2004
Cooley JH, Evain EJ. Amine dealkylations with acyl chlorides. Synthesis, 1989: 1–7
Machara A, Cox DP, Hudlicky T. Direct synthesis of naltrexone by palladium-catalyzed N-demethylation/acylation of oxymorphone: the benefit of C-H activation and the intramolecular acyl transfer from C-14 hydroxy. Adv Synth Catal, 2012, 354: 2713–2718
Carroll RJ, Leisch H, Scocchera E, Hudlicky T, Cox DP. Palladium-catalyzed N-demethylation/N-acylation of some morphine and tropane alkaloids. Adv Synth Catal, 2008, 350: 2984–2992
Khai BT, Arcelli AJ. Homogeneous transition-metal catalysis. Cleavage of the C-N bond of tertiary amines by acid anhydrides in the presence of transition metal ions. Organomet Chem, 1983, 252: c9–c13
Mariella RP, Brown KH. A novel SN1 displacement: the reaction of tertiary amines with acetic anhydride. Can J Chem, 1971, 49: 3348–3351
Murata S, Suzuki K, Tamatani A, Miura M, Nomura M. Oxidative dealkylation of 4-substituted N,N-dialkylanilines with molecular oxygen in the presence of acetic anhydride promoted by cobalt(II) or copper(I) chloride. J Chem Soc Perkin Trans 1, 1992: 1387–1392
Li Y, Ma L, Li Z. Amide bond formation through iron-catalyzed oxidative amidation of tertiary amines with anhydrides. J Org Chem, 2013, 78: 5638–5642
Chen X, Chen T, Li Q, Zhou Y, Han LB, Yin SF. Copper-catalyzed aerobic oxidative inert C-C and C-N bond cleavage: a new strategy for the synthesis of tertiary amides. Chem Eur J, 2014, 20: 12234–12238
Zhang C, Xu Z, Shen T, Wu G, Zhang LG, Jiao N. Lewis acid promoted highly diastereoselective petasis Borono-Mannich reaction: efficient synthesis of optically active b,g-unsaturated a-amino acids. Org Lett, 2012, 14: 2062–2065
Qin C, Wang Z, Chen F, Yang O, Jiao N. Iron-catalyzed C-H and C-C bond cleavage: a direct approach to amides from simple hydrocarbons. Angew Chem Int Ed, 2011, 50: 12595–12599
Zhang C, Xu Z, Zhang L, Jiao N. Copper-catalyzed aerobic oxidative coupling of aryl acetaldehydes with anilines leading to a-ketoamides. Angew Chem Int Ed, 2011, 50: 11088–11092
Bao YS, Zhaorigetu B, Agula B, Baiyin M, Jia M. Aminolysis of aryl ester using tertiary amine as amino donor via C-O and C-N bond activations. J Org Chem, 2014, 79: 803–808
Murata S, Suzuki K, Tamatani A, Miura M, Nomura M. Oxidative dealkylation of 4-substituted N,N-dialkylanilines with molecular oxygen in the presence of acetic anhydride promoted by cobalt(II) or copper(I) chloride. J Chem Soc Perkin Trans 1, 1992: 1387–1392
Murata S, Tamatani A, Suzuki K, Miura M, Nomura M. Cobalt(II) chloride catalyzed oxidation of 4-substituted N,N-dialkylanilines with molecular oxygen in the presence of acetic anhydride. Chem Lett, 1990, 19: 757–760
Hao W, Xi ZF. Palladium-catalyzed one-pot three- or four-component coupling of aryl iodides, alkynes, and amines through C-N bond cleavage: efficient synthesis of indole derivatives. Chem Eur J, 2014, 20: 2605–2612
Gu LJ, Wang W, Li GP. Synthesis of 2-arylbenzoxazoles through oxidation of C-H bonds adjacent to oxygen atoms. Eur J Org Chem, 2014: 319–322
Du YD, Tse CW, Xu ZJ, Liu Y, Che CM. [FeIII(TF4DMAP)OTf] catalysed anti-Markovnikov oxidation of terminal aryl alkenes to aldehydes and transformation of methyl aryl tertiary amines to formamides with H2O2 as a terminal oxidant. Chem Commun, 2014. 50: 12669–12672
Huo CD, Tang J. Aerobic oxidative mannich reaction promoted by catalytic amounts of stable radical cation salt. J Org Chem, 2014, 79: 9860–9864
Bao YS, Jia ML. Aminolysis of aryl ester using tertiary amine as amino donor via C-O and C-N bond activations. J Org Chem, 2014, 79: 803–808
Wang F, Luo CP, Deng GJ, Yang L. C(sp3)-C(sp3) bond formation via copper/Brønsted acid co-catalyzed C(sp3)-H bond oxidative cross-dehydrogenative-coupling (CDC) of azaarenes. Green Chem, 2014, 16: 2428–2431
Zhang C, Jiao N. Copper-catalyzed aerobic oxidative dehydrogenative coupling of anilines leading to aromatic azo compounds using dioxygen as an oxidant. Angew Chem Int Ed, 2010, 49: 6174–6177
Zhang C, Jiao N. Dioxygen activation under ambient conditions: Cu-catalyzed oxidative amidation-diketonization of terminal alkynes leading to α-ketoamides. J Am Chem Soc, 2010, 132: 28–29
Li Y, Ma L, Li Z. Progress in the transition-metal catalyzed oxidation of tertiary amines. Chin J Org Chem, 2013, 33: 704–714
Liu W, Liu J, Ogawa D, Nishihara Y, Guo X, Li Z. Iron-catalyzed oxidation of tertiary amines: synthesis of β-1,3-dicarbonyl aldehydes by three-component C-C couplings. Org Lett, 2011, 13: 6272–6275
Li H, He Z, Guo X, Li W, Zhao X, Li Z. Iron-catalyzed selective oxidation of N-methyl amines: highly efficient synthesis of methylene-bridged bis-1,3-dicarbonyl compounds. Org Lett, 2009, 11: 4176–4179
Lei Z, Ye J, Sun J, Shi ZJ. Direct alkenyl C-H functionalization of cyclic enamines with carboxylic acids via Rh catalysis assisted by hydrogen bonding. Org Chem Front, 2014, 1: 634–638
Zheng Q, Wang W, Jiang G, Yu ZX. CuI-catalyzed C1-alkynylation of tetrahydroisoquinolines (THIQs) by A3 reaction with tunable iminium ions. Org Lett, 2013, 15: 5928–5931
Liu J, Liu Q, Yi H, Qin C, Bai R, Qi X, Lan Y, Lei A. Visible-light-mediated decarboxylation/oxidative amidation of a-keto acids with amines under mild reaction conditions using O2. Angew Chem Int Ed, 2014, 53: 502–506
Takahashi H, Kashiwa N, Hashimoto Y, Nagasawa K. Novel Mannich-type nucleophilic substitution reaction with tertiary aromatic amines. Tetrahedron Lett, 2002, 43: 2935–2938
Penn JH, Owens WH, Petersen JL, Finklea HO, Snider DA. Mixed anhydrides: physical properties influenced by molecular structure. J Org Chem, 1993, 58: 2128–2133
Chen H, Groot MJ, Vermeulen PE, Hanzlik RP. Oxidative N-dealkylation of p-cyclopropyl-N,N-dimethylaniline. A substituent effect on a radical-clock reaction rationalized by ab initio calculations on radical cation intermediates. J Org Chem, 1997, 62: 8227–8230
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ma, L., Li, Y. & Li, Z. Iron-catalyzed aerobic oxidative amidation of tertiary amines with carboxylic acids. Sci. China Chem. 58, 1310–1315 (2015). https://doi.org/10.1007/s11426-015-5368-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5368-z