Abstract
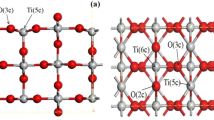
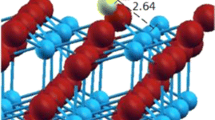
The adsorption and dissociation of CH3OH on TiO2 (110) were studied using density functional theory methods. Our results suggest that CH3OH molecules can adsorb up to 3/4 ML (1 ML=5.2×1014 molecules/cm2) coverage at five-coordinated titanium (Ti5c) sites to form the first layer. In the second layer, the CH3OH is adsorbed at bridge-bonded oxygen, and from the third layer, the CH3OH molecules form a hydrogen-bonded network with each other. The theoretical results show that dissociation of multilayer adsorbed methanol to aldehyde occurs through a stepwise pathway, with easy O-H bond dissociation and rate-determining C-H bond dissociation. The dissociation barriers for 8 or 12 CH3OH molecules on TiO2 are higher than that for low coverage by 0.15–0.21 eV; this suggests that the dissociation of multilayer adsorbed CH3OH is harder.
Similar content being viewed by others
References
Fujishima A, Honda K. Photolysis-decomposition of water at the surface of an irradiated semiconductor. Nature, 1972, 37: 238–245
Oregan B, Grätzel M. A low-cost, high-efficiency solar-cell based on dye-sensitized colloidal TiO2 films. Nature, 1991, 353: 737–740
Linsebigler AL, Lu G, Yates JJT. Photocatalysis on TiO2 surface-principles, mechanisms, and selected results. Chem Rev, 1995, 95: 735–738
Diebold U. The surface science of titanium dioxide. Surf Sci, 2003, 48: 53–229
Fujishima A, Zhang X, Tryk DA. TiO2 photocatalysis and related surface phenomena. Surf Sci Rep, 2008, 63: 515–582
Gundlach L, Ernstorfer R, Willig F. Ultrafast interfacial electron transfer from the excited state of anchored molecules into a semiconductor. Prog Surf Sci Rep, 2007, 82: 355–377
Fujishima A, Zhang X, Tryk DA. TiO2 photocatalysis and related surface phenomena. Surf Sci Rep, 2008, 63: 515–582
Chen X, Mao SS. Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem Rev, 2007, 107: 2891–2959
Henderson MA. A surface science perspective on TiO2 photocatalysis. Surf Sci Rep, 2011, 66: 185–297
Bates SP, Gillan MJ, Kresse G. Adsorption of methanol on TiO2 (110): a first-principles investigation. J Phys Chem B, 1998, 102: 2017–2026
Henderson MA, Otero-Tapia S, Castro ME. The chemistry of methanol on the surface: the TiO2 (110) influence of vacancies and coadsorbed species. Faraday Discuss, 1999, 114: 313–329
Pang CL, Lindsay R, Thornton G. Chemical reactions on rutile TiO2 (110). Chem Soc Rev, 2008, 37: 2328–2353
Onda K, Li B, Zhao J, Petek H. The electronic structure of methanol covered TiO2 (110) surfaces. Surf Sci, 2005, 593: 32–37
Ma Z, Guo Q, Mao X, Ren ZF, Wang X, Xu CB, Yang WS, Dai DX, Zhou CY, Fan HJ, Yang XM. Photocatalytic dissociation of ethanol on TiO2 (110) by near-band-gap excitation. J Phys Chem C, 2013, 117: 10336–10344
Guo Q, Xu C, Ren Z, Yang WS, Ma ZB, Dai DX, Fan HJ, Minton TK, Yang XM. Stepwise photocatalytic dissociation of methanol and water on TiO2 (110). J Am Chem Soc, 2012, 134: 13366–13373
Zhou C, Ren Z, Tan S, Ma ZB, Mao XC, Dai DX, Fan HQ, Fan HQ, Yang XM, LaRue J, Cooper R, Wodtke AM, Wang Z, Li ZY, Wang B, Yang JL, Hou JG. Site-specific photocatalytic splitting of methanol on TiO2 (110). Chem Sci, 2010, 1: 575–580
Phillips KR, Jensen SC, Baron M, Li SC, Friend CM. Sequential photo-oxidation of methanol to methyl formate on TiO2 (110). J Am Chem Soc, 2013, 135: 574–577
Yuan Q, Wu Z, Jin Y, Xu LS, Xiong F, Ma YS, Huang WX. Photocatalytic cross-coupling of methanol and formaldehyde on a rutile TiO2 (110) surface. J Am Chem Soc, 2013, 135: 5212–5219
Guo Q, Xu C, Yang W, Ren ZF, Ma ZB, Dai DX, Minton TK, Yang XM. Methyl formate production on TiO2 (110), initiated by methanol photocatalysis at 400 nm. Phys Chem C, 2013, 117: 5293–5300
Zhang Z, Bondarchuk O, White JM, Kay BD, Dohnalek Z. Imaging adsorbate O-H bond cleavage: methanol on TiO2(110). J Am Chem Soc, 2006, 128: 4198–4199
Zhou C, Ren Z, Tan S, Ma Z, Mao X, Dai D, Fan H, Yang X, LaRue J, Cooper R, Wodtke AM, Wang Z, Li Z, Wang B, Yang J, Hou J. Site-specific photocatalytic splitting of methanol on TiO2 (110). Chem Sci, 2010, 1: 575–580
Sánchez de Armas R, Oviedo J, San Miguel MÁ, Sanz JF. Methanol adsorption and dissociation on TiO2 (110) from first principles calculations. J Phys Chem C, 2007, 111: 10023–10028
Oviedo J, Sánchez de Armas R, San Miguel MÁ, Sanz JF. Methanol and water dissociation on TiO2 (110): the role of surface oxygen. J Phys Chem C, 2008, 112: 17737–17740
Zhao J, Yang J, Petek H. Theoretical study of the molecular and electronic structure of methanol on a TiO2 (110) surface. Phys Rev B, 2009, 80: 235416
Kresse G, Hafner J. Ab initio molecular dynamics for liquid metals. Phys Rev B, 1993, 47: 558–561
Kresse G, Furthmuller J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B, 1996, 54: 11169–11186
Blöchl PE. Projector augmented-wave method. Phys Rev B, 1994, 50: 17953–17979
Perdew JP, Burke K, Ernzerhof M. Generalized gradient approximation made simple. Phys Rev Lett, 1996, 77: 3865–3868
Henkelman G, Uberuaga BP, Jonsson H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J Chem Phys, 2000, 113: 9901–9904
Henkelman G, Jonsson H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J Chem Phys, 2000, 113: 9978–9985
Perdew J, Ernzerhof M, Burke K. Rationale for mixing exact exchange with density functional approximations. J Chem Phys, 1996, 105: 9982–9985
Paier J, Hirsch R, Marsman M, Kresse G. The Perdew-Burke-Ernzerh of exchange-correlation functional applied to the G2-1 test set using a plane-wave basis set. J Chem Phys, 2005, 122: 234102
Zhang X, Wen B, Zhou CY, Ren ZF, Liu LM. First-principles study of methanol oxidation into methyl formate on rutile TiO2(110). J Phys Chem C, 2014, 118: 19859–19868
Kowalski PM, Meyer B, Marx D. Composition, structure, and stability of the rutile TiO2 (110) surface: oxygen depletion, hydroxylation, hydrogen migration, and water adsorption. Phys Rev B, 2009, 79: 115410
Stefanovich EV, Truong TN. Ab initio study of water adsorption on TiO2 (110): molecular adsorption versus dissociative chemisorption. Chem Phys Lett, 1999, 299: 623–629
Monkhorst HJ, Pack JD. Special points for Brillouin-zone integrations. Phys Rev B, 1976, 13: 5188–5192
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, R., Fan, H. The adsorption and dissociation of multilayer CH3OH on TiO2 (110). Sci. China Chem. 58, 614–619 (2015). https://doi.org/10.1007/s11426-015-5342-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5342-9