Abstract
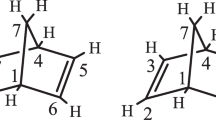
The role of bismuth in the selective oxidation of propene has long been debated. We performed density functional calculations to study the dehydrogenation reaction of propene on Bi2O3 surfaces. Our calculated thermodynamic data reveal that the first dehydrogenation of propene on the most stable (010) surface and the (100) surface are difficult. Our calculations indicate that the barrier of the first hydrogen abstraction on the high Miller index surface (211) is much lower than those on the (100) and (010) surfaces, and is close to the experimental one. Further dehydrogenation is shown to be difficult and production of 1,5-hexadiene through dimerization of allyl is likely, in agreement with the experimental observations.
Similar content being viewed by others
References
Wang L, Peng B, Peng LM, Guo XF, Xie ZK, Ding WP. Mesostructural Bi-Mo-O catalyst: correct structure leading to high performance. Sci Rep, 2013, 3: 2881
Ayame A, Uchida K, Iwataya M, Miyamoto M. X-ray photoelectron spectroscopic study on α- and γ-bismuth molybdate surfaces exposed to hydrogen, propene and oxygen. Appl Catal A, 2002, 227: 7–17
Burrington JD, Kartisek CT, Grasselli RK. Aspects of selective oxidation and ammoxidation mechanisms over bismuth molybdate catalysts: II. Allyl alcohol as a probe for the allylic intermediate. J Catal, 1980, 63: 235–254
Adams CR, Jennings TJ. Mechanism studies of the catalytic oxidation of propylene. J Catal, 1964, 3: 549–558
Snyder TP, Hill CG. The mechanism for the partial oxidation of propylene over bismuth molybdate catalysts. Catal Rev-Sci Eng, 1989, 31: 43–95
Burrington JD, Grasselli RK. Aspects of selective oxidation and ammoxidation mechanisms over bismuth molybdate catalysts. J Catal, 1979, 59: 79–99
Brückman K, Grabowski R, Haber J, Mazurkiewicz A, Sloczynski J, Wiltowski T. The role of different MoO3 crystal faces in elementary steps of propene oxidation. J Catal, 1987, 104: 71–79
Grzybowska B, Haber J, Janas J. Interaction of allyl iodide with molybdate catalysts for the selective oxidation of hydrocarbons. J Catal, 1977, 49: 150–163
Swift HE, Bozik JE, Ondrey JA. Dehydrodimerization of propylene using bismuth oxide as the oxidant. J Catal, 1971, 21: 212–224
Massoth FE, Scarpiello DA. Kinetics of bismuth oxide reduction with propylene. J Catal, 1971, 21: 225–238
Martir W, Lunsford JH. The formation of gas-phase π-allyl radicals from propylene over bismuth oxide and γ-bismuth molybdate catalysts. J Am Chem Soc, 1981, 103: 3728–3732.
Drlscol DJ, Lunsford JH. Kinetic isotope effect in the partial oxidatlon of propylene over Bi2O3. J Phys Chem, 1983, 87: 301–303
Ueda W, Asakawa K, Chen CL, Moro-Oka Y, Ikawa T. Catalytic properties of tricomponent metal oxides having the scheelite structure: I. Role of bulk diffusion of lattice oxide ions in the oxidation of propylene. J Catal, 1986, 101: 360–368
Adams CR, Voge HH, Morgan CZ, Armstrong WE. Oxidation of butylenes and propylene over bismuth molybdate. J Catal, 1964, 3: 379–386
Peacock JM, Parker AJ, Ashmore PG, Hockey JA. The oxidation of propene over bismuth oxide, molybdenum oxide, and bismuth molybdate catalysts: IV. The selective oxidation of propene. J Catal, 1969, 15: 398–406
Mehandru SP, Anderson AB, Brazdil JF. CH bond activation and radical-surface reactions for propylene and methane over α-Bi2O3. J Chem Soc, Faraday Trans, 1987, 83: 463–475
Jang YH, Goddard WA. Selective oxidation and ammoxidation of propene on bismuth molybdates, ab initio calculations. Top Catal, 2001, 15: 273–289
Jang YH, Goddard WA. Mechanism of selective oxidation and ammoxidation of propene on bismuth molybdates from DFT calculations on model clusters. J Phys Chem B, 2002, 106: 5997–6013
Pudar S, Oxgaard J, Chenoweth K, Duin ACT, Goddard WA. Mechanism of selective oxidation of propene to acrolein on bismuth molybdates from quantum mechanical calculations. J Phys Chem C, 2007, 111: 16405–16415
Getsoian AB, Shapovalov V, Bell AT. DFT+U investigation of propene oxidation over bismuth molybdate: active sites, reaction intermediates, and the role of bismuth. J Phys Chem C, 2013, 117: 7123–7137
Kresse G, Hafner J. Ab initio molecular dynamics for liquid metals. Phys Rev B, 1993, 47: 558–561
Perdew JP, Burke K, Ernzerhof M. Generalized gradient approximation made simple. Phys Rev Lett, 1996, 77: 3865–3868
Blöchl PE. Projector augmented-wave method. Phys Rev B, 1994, 50: 17953–17979
Malmros G. The crystal structure of alpha-Bi2O3. Acta Chem Scand, 1970, 24: 384–396
Monkhorst HJ, Pack JD. Special points for Brillouin-zone integrations. Phys Rev B, 1976, 13: 5188–5192
Lei YH, Chen ZX. Density functional study of the stability of various α-Bi2O3 surfaces. J Chem Phys, 2013, 138: 054703
Davad RL. CRC Handbook of Chemistry and Physics. 88th ed. Boca Raton: Taylor & Francies Group, 2007–2008
Haber J, Witko M. Oxidation catalysis-electronic theory revisited. J Catal, 2003, 216: 416–424
Fu G, Xu X, Lu X, Wan HL. Mechanisms of methane activation and transformation on molybdenum oxide based catalysts. J Am Chem Soc, 2005, 127: 3989–3996
Perdew JP, Ruzsinszky A, Csonka GI, Vydrov OA, Scuseria GE, Constantin LA, Zhou X, Burke K. Restoring the density-gradient expansion for exchange in solids and surfaces. Phys Rev Lett, 2008, 100: 136406
Grimme S, Antony J, Ehrlich S, Krieg H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys, 2010, 132: 154104
Brønsted JN. Acid and basic catalysis. Chem Rev, 1928, 5: 231–335
Evans MG, Polanyi M. Inertia and driving force of chemical reactions. Trans Faraday Soc, 1938, 34: 11–24
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lei, YH., Chen, ZX. Theoretical study of propene oxidation on Bi2O3 surfaces. Sci. China Chem. 58, 593–600 (2015). https://doi.org/10.1007/s11426-015-5341-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5341-x