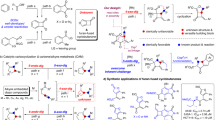
Abstract
Transmetalation of carborane-fused zirconacycles to Cu(II) induces the C-C coupling reaction to form four-membered rings. This serves as a new efficient and general methodology for the generation of a series of carborane-fused cyclobutenes and cyclobutanes. A reaction mechanism involving transmetalation to Cu(II) and reductive elimination is proposed.
Similar content being viewed by others
References
Schore NE. Transition metal-mediated cycloaddition reactions of alkynes in organic synthesis. Chem Rev, 1988, 88: 1081–1119
Trost BM. The atom economy—A search for synthetic efficiency. Science, 1991, 254: 1471–1477
Lautens M, Klute W, Tam W. Transition metal-mediated cycloaddition reactions. Chem Rev, 1996, 96: 49–92
Nakamura I, Yamamoto Y. Transition-metal-catalyzed reactions in heterocyclic synthesis. Chem Rev, 2004, 104: 2127–2198
Rosen BM, Quasdorf KW, Wilson DA, Zhang N, Resmerita AM, Garg NK, Percec V. Nickel-catalyzed cross-couplings involving carbonoxygen bonds. Chem Rev, 2011, 111: 1346–1416
Liu C, Zhang H, Shi W, Lei A. Bond formations between two nucleophiles: transition metal catalyzed oxidative cross-coupling reactions. Chem Rev, 2011, 111: 1780–1824
Wang L, He W, Yu Z. Transition-metal mediated carbon-sulfur bond activation and transformations. Chem Soc Rev, 2013, 42: 599–621
Hawthorne MF. The role of chemistry in the development of boron neutron capture therapy of cancer. Angew Chem Int Ed, 1993, 32: 950–984
Armstrong AF, Valliant JF. The bioinorganic and medicinal chemistry of carboranes: from new drug discovery to molecular imaging and therapy. Dalton Trans, 2007, 4240–4251
Issa F, Kassiou M, Rendina LM. Boron in drug discovery: carboranes as unique pharmacophores in biologically active compounds. Chem Rev, 2011, 111: 5701–5722
Jude H, Disteldorf H, Fischer S, Wedge T, Hawkridge AM, Arif AM, Hawthorne MF, Muddiman DC, Stang PJ. Coordination-driven self-assemblies with a carborane backbone. J Am Chem Soc, 2005, 127: 12131–12139
Dash BP, Satapathy R, Gaillard ER, Maguire JA, Hosmane NS. Synthesis and properties of carborane-appended C 3-symmetrical extended π systems. J Am Chem Soc, 2010, 132: 6578–6587
Yao ZJ, Jin GX. Transition metal complexes based on carboranyl ligands containing N, P, and S donors: synthesis, reactivity and applications. Coord Chem Rev, 2013, 257: 2522–2535
Meng X, Wang F, Jin GX. Construction of M-M bonds in late transition metal complexes. Coord Chem Rev, 2010, 254: 1260–1272
Yao ZJ, Yu WB, Lin YJ, Huang SL, Li ZH, Jin GX. Iridium-mediated regioselective B-H/C-H activation of carborane cage: a facile synthetic route to metallacycles with a carborane backbone. J Am Chem Soc, 2014, 136: 2825–2832
Yao ZJ, Jin GX. Synthesis, reactivity, and structural transformation of mono- and binuclear carboranylamidinate-based 3d metal complexes and metallacarborane derivatives. Organometallics, 2012, 31: 1767–1774
Wee KR, Cho YJ, Jeong S, Kwon S, Lee JD, Suh IH, Kang SO. Carborane-based optoelectronically active organic molecules: wide band gap host materials for blue phosphorescence. J Am Chem Soc, 2012, 134: 17982–17990
Cioran AM, Musteti AD, Teixidor F, Krpetić Z, Prior IA, He Q, Kiely CJ, Brust M, Vinñas C. Mercaptocarborane-capped gold nanoparticles: electron pools and ion traps with switchable hydrophilicity. J Am Chem Soc, 2012, 134: 212–221
Visbal R, Ospino I, López-de-Luzuriaga JM, Laguna A, Gimeno MC. N-heterocyclic carbene ligands as modulators of luminescence in three-coordinate gold(I) complexes with spectacular quantum yields. J Am Chem Soc, 2013, 135: 4712–4715
Shi C, Sun H, Tang X, Lv W, Yan H, Zhao Q, Wang J, Huang W. Variable photophysical properties of phosphorescent iridium(III) complexes triggered by closo- and nido-carborane substitution. Angew Chem Int Ed, 2013, 52: 13434–13438
Shi C, Sun H, Jiang Q, Zhao Q, Wang J, Huang W, Yan H. Carborane tuning of photophysical properties of phosphorescent iridium(III) complexes. Chem Commun, 2013, 49: 4746–4748
Zhu L, Lv W, Liu S, Yan H, Zhao Q, Huang W. Carborane enhanced two-photon absorption of tribranched fluorophores for fluorescence microscopy imaging. Chem Commun, 2013, 49: 10638–10640
Grimes RM. Carboranes. 2nd edn. Amsterdam: Academic Press, 2011
Hosmane NS, Maguire JA. Metallacarboranes of d- and f-block metals. In: Crabtree RH, Mingos DMP, Eds. Comprehensive Organometallic Chemistry III. Oxford: Elsevier, 2007, Vol. 3, Chapter 5.
Xie Z. Advances in the chemistry of metallacarboranes of f-block elements. Coord Chem Rev, 2002, 231: 23–46
Xie Z. Cyclopentadienyl-carboranyl hybrid compounds: a new class of versatile ligands for organometallic chemistry. Acc Chem Res, 2003, 36: 1–9
Xie Z. Group 4 metallocenes incorporating constrained-geometry carboranyl ligands. Coord Chem Rev, 2006, 250: 259–272
Deng L, Xie Z. Advances in the chemistry of carboranes and metallacarboranes with more than 12 vertices. Coord Chem Rev, 2007, 251: 2452–2476
Qiu Z, Ren S, Xie Z. Transition metal-carboryne complexes: synthesis, bonding, and reactivity. Acc Chem Res, 2011, 44: 299–309
Deng L, Chan HS, Xie Z. Synthesis, structure, and reactivity of a zirconocene-carboryne precursor. J Am Chem Soc, 2005, 127: 13774–13775
Deng L, Chan HS, Xie Z. Nickel-mediated regioselective [2+2+2] cycloaddition of carboryne with alkynes. J Am Chem Soc, 2006, 128: 7728–7729
Qiu Z, Xie Z. Nickel-mediated coupling reactions of carboryne with alkenes: a synthetic route to alkenylcarboranes. Angew Chem Int Ed, 2008, 47: 6572–6575
Qiu Z, Xie Z. Nickel-mediated three-component cycloaddition reaction of carboryne, alkenes, and alkynes. J Am Chem Soc, 2009, 131: 2084–2085
Qiu Z, Wang SR, Xie Z. Nickel-catalyzed regioselective [2+2+2] cycloaddition of carboryne with alkynes. Angew Chem Int Ed, 2010, 49: 4649–4652
Qiu Z, Xie Z. Palladium/nickel-cocatalyzed cycloaddition of 1,3-dehydro-o-carborane with alkynes. Facile synthesis of C,B-substituted carboranes. J Am Chem Soc, 2010, 132: 16085–16093
Ren S, Chan HS, Xie Z. Reaction of zirconocene-carboryne precursor with alkynes: an efficient route to zirconacyclopentenes incorporating a carboranyl unit. Organometallics, 2009, 28: 4106–4114
Ren S, Qiu Z, Xie Z. Reaction of zirconocene-carboryne with alkenes: synthesis and structure of zirconacyclopentanes with a carborane auxiliary. Organometallics, 2012, 31: 4435–4441
Ren S, Qiu Z, Xie Z. Synthesis of neutral group 4 metal-carboryne complexes and their reactivity toward unsaturated molecules. Organometallics, 2013, 32: 4292–4300
Ren S, Qiu Z, Xie Z. Transition metal promoted or catalyzed exo-cyclic alkyne insertion via zirconacyclopentene with carborane auxillary: formation of symmetric or unsymmetric benzocarboranes. J Am Chem Soc, 2012, 134: 3242–3254
Ren S, Qiu Z, Xie Z. Three-component [2+2+2] cycloaddition of carboryne, unactivated alkene, and alkyne via zirconacyclopentane mediated by nickel: one-pot synthesis of dihydrobenzocarboranes. Angew Chem Int Ed, 2012, 51: 1010–1013
Quan Y, Zhang J, Xie Z. Three-component [2+2+1] cross-cyclotri-merization of carboryne, unactivated alkene, and trimethylsilylalkyne co-mediated by Zr and Ni. J Am Chem Soc, 2013, 135: 18742–18745
Marek I, Ed. Titanium and Zirconium in Organic Synthesis. Weinhein: Wiley-VCH, 2002.
Negishi E, Takahashi T. Patterns of stoichiometric and catalytic reactions of organozirconium and related complexes of synthetic interest. Acc Chem Res, 1994, 27: 124–130
Negishi E. A quarter of a century of explorations in organozirconium chemistry. Dalton Trans, 2005, 827–848
Ren S, Chan HS, Xie Z. Synthesis, structure, and reactivity of zirconacyclopentene incorporating a carboranyl unit. J Am Chem Soc, 2009, 131: 3862–3863
Lee T, Jeon J, Song KH, Jung I, Baik C, Park KM, Lee SS, Kang SO, Ko J. Generation and trapping reaction of an efficient 1,2-dehydrocarborane precursor, phenyl[o-(trimethylsilyl)carboranyl]iodonium acetate. Dalton Trans, 2004, 933–937
Ho DM, Cunningham RJ, Brewer JA, Bian N, Jones M Jr. Reaction of 1,2-dehydro-o-carborane with acetylenes. Synthesis and structure of a carborane analogue of benzocyclobutadiene. Inorg Chem, 1995, 34: 5274–5278
Sheldrick GM. SADABS: program for empirical absorption correction of area detector data. Dissertation for the Doctoral Degree. Germany: University of Göttingen, 1996
Sheldrick GM. SHELXTL 5.10 for Windows NT: Structure Determination Software Programs. Bruker Analytical X-ray Systems, Inc., Madison, Wisconsin, USA, 1997
L’Esperance RP, Li ZH, Van Engen D, Jones M Jr. New syntheses of 1,2-ethano-o-carborane and the structure of 9-chloro-1,2-ethano-o-carborane. Inorg Chem, 1989, 28: 1823–1826
Allen FH. The Geometry of small rings. VI. Geometry and bonding in cyclobutane and cyclobutene. Acta Cryst, 1984, B40: 64–72
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dedicated to Professor Qian Changtao on the occasion of his 80th birthday.
WANG ShaoWu received his BSc and MS degrees from Anhui Normal University in 1985 and 1992, respectively. After obtaining a PhD from The Chinese University of Hong Kong in 1999, he spent one year as a postdoctoral fellow at The Chinese University of Hong Kong. He then joined the faculty of the Anhui Normal University as a full professor of chemistry. He serves as the vice-president of the Anhui Normal University since June 2009, and has co-authored about 110 publications in peer-reviewed journals, 5 patents, and received two 2nd prize awards from the State Commission of Education and Anhui Province. His research interests include organolanthanide chemistry and organic synthesis.
XIE ZuoWei obtained a B.Sc. from Hangzhou University in 1983, a M.Sc. from Shanghai Institute of Organic Chemistry (SIOC) in 1986, and his Ph.D. in 1990, working in a joint program between SIOC and the Technische Universität Berlin under the co-supervision of Profs. Changtao Qian, Yaozeng Huang and Herbert Schumann. He is a Choh-Ming Li Professor of Chemistry in The Chinese University of Hong Kong, a member of Shanghai-Hong Kong Joint Laboratory in Chemical Synthesis and the 2008 recipient of the State Natural Science Prize.
Rights and permissions
About this article
Cite this article
Yuan, Y., Ren, S., Qiu, Z. et al. Synthesis of carborane-fused cyclobutenes and cyclobutanes. Sci. China Chem. 57, 1157–1163 (2014). https://doi.org/10.1007/s11426-014-5112-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-014-5112-0