Abstract
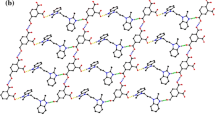
Multifunctional molecules are capable of assembling via different supramolecular synthons, or hydrogen bond motifs, between the same or different functional groups, leading to the possibility of cocrystal. Utilization of the interplay of dimensionality (1-D, 2-D and 3-D), orientation of functional groups of the building blocks, influence of rigid/flexible linking groups, and weak interactions provides an interesting route for the creation of novel supramolecular architectures in the crystal lattice. N-unsubstituted 1H-benzotriazole and carboxylic acid, being self-complementary molecules, offer a broad scope of study of binary compounds based on the complementary combination of H-bonding/donating sites. We report here the construction of three extended molecular networks in cocrystals of the carboxylic acid group of the acid and the 1H-benzotriazole triazole moiety. We have been able to identify four major supramolecualr synthons that would be helpful in the prediction of structural motifs for these kinds of studies. Interestingly, these heterosynthons are strikingly similar, to those of the homosynthons of the individual functional groups. The nature of the aza groups helps to enhance the overall volume of the crystal lattice thus leading to the formation of various supramolecular assemblies. Thermal stability of these compounds has been investigated by thermogravimetric analysis (TGA) of mass loss.
Similar content being viewed by others
References
Aakeröy CB, Salmon DJ. Building co-crystals with molecular sense and supramolecular sensibility. CrystEngComm, 2005, 7: 439–448
Desiraju GR. Crystal engineering: A holistic view. Angew Chem Int Ed, 2007, 46: 8342–8356
Janiak C. Engineering coordination polymers towards applications. Dalton Trans, 2003, 14: 2781–2804
James S. Metal-organic frameworks. Chem Soc Rev, 2003, 32: 276–288
Brammer L. Developments in inorganic crystal engineering. Chem Soc Rev, 2004, 33: 476–489
Moulton B, Zaworotko MJ. From molecules to crystal engineering: Supramolecular isomerism and polymorphism in network solids. Chem Rev, 2001, 101: 1629–1658
Braga D, Brammer L, Champness NR. New trends in crystal engineering. CrystEngComm, 2005, 7: 1–19
Hosseini MW. Molecular tectonics: From molecular recognition of anions to molecular networks. Coord Chem Rev, 2003, 240: 157–166
Lehn J-M. From supramolecular chemistry towards constitutional dynamic chemistry and adaptive chemistry. Chem Soc Rev, 2007, 36: 151–160
Hosseini MW. Molecular tectonics: from simple tectons to complex molecular networks. Acc Chem Res, 2005, 38: 313–323
Bis JA, McLaughlin OL, Vishweshwar P, Zaworotko MJ. Supramolecular heterocatemers and their role in cocrystal design. Cryst Growth Des, 2006, 6: 2648–2650
Aakeröy CB, Beatty AM, Helfrich BA. “Total synthesis” supramolecular style: Design and hydrogen-bond-directed assembly of ternary supermolecules. Angew Chem Int Ed, 2001, 40: 3240–3242
Aakeröy CB, Desper J, Helfrich BA. Heteromeric intermolecular interactions as synthetic tools for the formation of binary co-crystals. CrystEngComm, 2004, 6: 19–24
Sokolov AN, Friscic T, MacGillivray LR. Enforced face-to-face stacking of organic semiconductor building blocks within hydrogen-bonded molecular cocrystals. J Am Chem Soc, 2006, 128: 2806–2807
Desiraju GR, Steiner T. The Weak Hydrogen Bond in Structural Chemistry and Biology. Oxford: Oxford University Press, 1998
Jayaraman A, Balasubramaniam V, Valiyaveettil S. Self-Assembly of pentaphenol adducts: Formation of 3D network and ladder-type supramolecular structures in the solid state. Cryst Growth Des, 2006, 6: 636–642
Aakeröy CB, Schulteiss N, Desper J, Moore C. Balancing intermolecular hydrogen-bond interactions for the directed assembly of binary 1:1 co-crystals. New J Chem, 2006, 30: 1452–1460
Du M, Zhang ZH, Wang XG, Wu HF, Wang Q. Flexible building blocks of N,N′-bis(picolinoyl)hydrazine for hydrogen-bonding directed cocrystallization: structural diversity, concomitant polymorphs, and synthon prediction. Cryst Growth Des, 2006, 6: 1867–1875
Vangala VR, Bhogala BR, Dey A, Desiraju GR, Broder CK, Smith PS, Mondal R, Howard JAK, Wilson CC. Correspondence between molecular functionality and crystal structures supramolecular chemistry of a family of homologated aminophenols. J Am Chem Soc, 2003, 125: 14495–14509
Vangala VR, Mondal R, Broder CK, Howard JAK, Desiraju GR. Dianiline-Diphenol molecular complexes based on supraminol recognition. Cryst Growth Des, 2005, 5: 99–104
Desiraju GR. Crystal engineering: A holistic view. Angew Chem Int Ed, 2007, 46: 8342–8356
Joanna, AB, Vishweshwar P, Weyna D, Zaworotko MJ. Hierarchy of supramolecular synthons: persistent hydroxyl…pyridine hydrogen bonds in cocrystals that contain a cyano acceptor. Mol Pharm, 2007, 4: 401–41
Rajput L, Biradha K. Design of cocrystals via new and robust supramolecular synthon between carboxylic acid and secondary amide: honeycomb network with jailed aromatics. Cryst Growth Des, 2009, 9: 40–42
Dai FN, Sun D, Sun WM, Liu YQ, Sun DF. Conformation variation of tris(2-carboxyethyl)isocyanuric acid induced by cocrystallized N-heterocyclic organic molecules. CrystEngComm, 2012, 14: 1376–1381
Wang L, Xu LY, Xue RF, Lu XF, Chen RX, Tao XT. Cocrystallization of N-donor type compounds with 5-sulfosalicylic acid: The effect of hydrogen-bonding supramolecular architectures. Sci China Chem, 2012, 55: 138–144
Etter MC. Encoding and decoding hydrogen-bond patterns of organic compounds. Acc Chem Res, 1990, 23: 120–126
Desiraju GR. Supramolecular synthons in crystal engineering-a new organic synthesis. Angew Chem Int Ed, 1995, 34: 2311–2327
Etter MC, MacDonald JC, Bernstein J. Graph-set analysis of hydrogen-bond patterns in organic crystals. Acta Crystallogr, 1990, B46: 256–262
Du M, Zhang ZH, Zhao XJ. Cocrystallization of trimesic acid and pyromellitic acid with bent dipyridines. Cryst Growth Des, 2005, 5: 1247–1254
Nichol GS, Clegg W. Classical and weak hydrogen bonding interactions between 4,4′-bipyridine and organic acids: from co-crystal to organic complex. Cryst Growth Des, 2009, 9: 1844–1850
Khan M, Enkelmann V, Brunklaus G. O·H…N heterosynthon: A robust supramolecular unit for crystal engineering. Cryst Growth Des, 2009, 9: 2354–2362
Sutor DJ. Evidence for the existence of C-HO hydrogen bonds in crystals. J Chem Soc, 1963, 1105-1110
Taylor R, Kennard O. Hydrogen-bond geometry in organic crystals. Acc Chem Res, 1984, 17: 320–326
Burrows AD. Crystal engineering using multiple hydrogen bonds. Struct Bonding (Berlin), 2004, 108: 55–96
SAINT Software Reference Manual, Bruker AXS: Madison, WI, 1998
Sheldrick GM. SHELXTL NT Version 51 Program for solution and refinement of crystal structure: University of Gottingen, Germany, 1997
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, L., Zhao, L., Xue, R. et al. Construction of interesting organic supramolecular structures with synthons cooperation in the cocrystals of 1H-benzotriazole and hydroxybenzoic acids. Sci. China Chem. 55, 2515–2522 (2012). https://doi.org/10.1007/s11426-012-4652-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-012-4652-4