Abstract
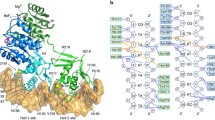
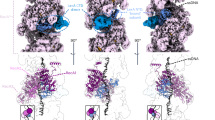
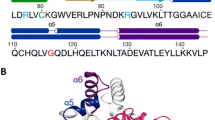
The multiple antibiotic resistance regulatory protein (MarR) binds to two promoter sites on the marO operator in Escherichia coli. Our study showed that more than one MarR dimer proteins bound to either of its two promoter sites (Site I and Site II), suggesting that MarR might form higher complexes than homodimers when bound to DNA inside E. coli cells. To further verify this hypothesis, we site-specifically incorporated a photocrosslinking probe at the interface between two MarR dimer proteins. Photolysis in living E. coli cells revealed a covalent linkage between the two interdimer subunits of MarR, suggesting that MarR forms dimer of dimers in vivo.
Similar content being viewed by others
References
Martinez JL, Fajardo A, Garmendia L, Hernandez A, Linares JF, Martínez-Solano L, Sánchez MB. A global view of antibiotic resistance. FEMS Microbiol Rev, 2009, 33(1): 44–65
Perera IC, Grove A. Molecular mechanisms of ligand-mediated attenuation of DNA binding by marr family transcriptional regulators. J Mol Cell Biol, 2010, 2(5): 243–254
Ariza RR, Cohen SP, Bachhawat N, Levy SB, Demple B. Repressor mutations in the marRAB operon that activate oxidative stress genes and multiple antibiotic resistance in Escherichia coli. J Bacteriol, 1994, 176(1): 143–148
Martin RG, Rosner JL. Binding of purified multiple antibiotic-resistance repressor protein (MarR) to mar operator sequences. Proc Nat Acad Sci USA, 1995, 92(12): 5456–5460
Alekshun MN, Levy SB. The mar regulon: Multiple resistance to antibiotics and other toxic chemicals. Trends Microbiol, 1999, 7(10): 410–413
Seoane A, Levy S. Characterization of MarR, the repressor of the multiple antibiotic resistance (mar) operon in Escherichia coli. J Bacteriol, 1995, 177(12): 3414–3419
Alekshun MN, Levy SB, Mealy TR, Seaton BA, Head JF. The crystal structure of MarR, a regulator of multiple antibiotic resistance, at 2.3 A resolution. Nat Struct Mol Biol, 2001, 8(8): 710–714
Brown BM, Sauer RT: Assembly of the arc repressor-operator complex: Cooperative interactions between DNA-bound dimers. Biochem, 1993, 32(5): 1354–1363
White A, Ding X, vanderSpek JC, Murphy JR, Ringe D. Structure of the metal-ion-activated diphtheria toxin repressor/tox operator complex. Nature, 1998, 394(6692): 502–506
Alekshun MN, Levy SB. Alteration of the repressor activity of MarR, the negative regulator of the Escherichia coli marRAB locus, by multiple chemicals in vitro. J Bacteriol, 1999, 181(15): 4669–4672
Zhang M, Lin S, Song X, Liu J, Fu Y, Ge X, Fu X, Chang Z, Chen PR. A genetically incorporated crosslinker reveals chaperone cooperation in acid resistance. Nat Chem Biol, 2011, 7: 671–677
Chen PR, Groff D, Guo JT, Ou WJ, Cellitti S, Geierstanger BH, Schultz PG. A facile system for encoding unnatural amino acids in mammalian cells. Angew Chem In Ed, 2009, 48(22): 4052–4055
Hancock SM, Uprety R, Deiters A, Chin JW: Expanding the genetic code of yeast for incorporation of diverse unnatural amino acids via a pyrrolysyl-tRNA synthetase/tRNA pair. J Am Chem Soc, 2010, 132(42): 14819–14824
Martin RG, Rosner JL. Transcriptional and translational regulation of the marRAB multiple antibiotic resistance operon in Escherichia coli. Mol Microbiol, 2004, 53(1): 183–191
Kumarevel T, Tanaka T, Umehara T, Yokoyama S. ST1710-DNA complex crystal structure reveals the DNA binding mechanism of the MarR family of regulators. Nucleic Acids Res, 2009, 37(14): 4723–4735
Fiorentino G, Ronca R, Cannio R, Rossi M, Bartolucci S. MarR-like transcriptional regulator involved in detoxification of aromatic compounds in sulfolobus solfataricus. J Bacteriol, 2007, 189(20): 7351–7360
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chen, X., Hao, Z. & Chen, P.R. Protein photocrosslinking reveals dimer of dimers formation on MarR protein in Escherichia coli . Sci. China Chem. 55, 106–111 (2012). https://doi.org/10.1007/s11426-011-4437-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4437-1