Abstract
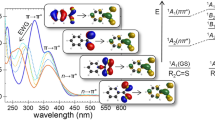
Twenty five samples of 4,4′-disubstituted stilbene derivatives were synthesized, and their UV absorption max wavelengths were determined in over 10 kinds of solvents including cyclohexane, ether, chloroform, acetonitrile and ethanol, in which 242 experimental data were recorded. The effects of substituents and solvents on the energy of their UV absorption max wavelengths were discussed. The research results showed: the energy of UV absorption max wavelengths of 4,4′-disubstituted stilbenes was mainly affected by their intramolecular structure (substituent effect) in a given solvent, that is, the energy is dominated by both of excited-state substituent parameter σ exCC and polar substituent constant σ p. While their energy was dominated by the substituent effect and solvent effect in different kinds of solvents. An equation quantifying the energy of UV absorption max wavelengths of 4,4′-disubstituted stilbenes was developed. In addition, it is found that the n-octanol/water partition coefficient (logP) is more effective than the solvatochromic dye (E T(30)) in scaling the solvent effect. The equation employed the parameter logP has a better correlation and more specific physical meaning. Further, the energies of UV absorption max wavelengths of some reported compounds were predicted by the obtained equation, which are in agreement with their experimental values.
Similar content being viewed by others
References
Waldeck DH. Photoisomerization dynamics of stilbenes. Chem Rev, 1991, 91: 415–436
Arai T, Tokumaru K. Photochemical one-way adiabatic isomerization of aromatic olefins. Chem Rev, 1993, 93: 23–39
Whitten D. Photochemistry and photophysics of trans-stilbene and related alkenes in surfactant assemblies. Acc Chem Res, 1993, 26: 502–509
Saltiel J. Perdeuteriostilbene. The role of phantom states in the cis-trans photoisomerization of stilbenes. J Am Chem Soc, 1967, 89: 1036–1037
I-Ren L, Luis B, Ahmed HZ. Direct observation of the primary bondtwisting dynamics of stilbene anion radical. J Am Chem Soc, 2008, 130: 6708–6709
Jason Q, Todd JM. Ab initio study of cis-trans photoisomerization in stilbene and ethylene. J Phys Chem A, 2003, 107: 829–837
Choi CH, Kertesz M. Conformational information from vibrational spectra of styrene, trans-stilbene, and cis-stilbene. J Phys Chem A, 1997, 101: 3823–3831
Improta R, Santoro F. Excited-STate behavior of trans- and cis-isomers of stilbene and stiff stilbene: A TD-DFT study. J Phys Chem A, 2005, 109: 10058–10067
Durbeej B, Eriksson LA. Photodegradation of substituted stilbene compounds: What colors aging paper yellow? J Phys Chem A, 2005, 109: 5677–5682
Bao J, Weber PM. Ultrafast dynamics of highly excited trans-stilbene: A different twist. J Phys Chem Lett, 2010, 1: 224–227
Langkildet FW, Wilbrandt R. Molecular structure of stilbene in the T1 state. Transient resonance Raman spectra of stilbene isotopomers and quantum chemical calculations. J Phys Chem, 1994, 98: 2254–2265
Görner H. Phosphorescence of trans-stllbene, stilbene derivatives, and stilbene-like molecules at 77 K. J Phys Chem, 1989, 93: 1826–1832
Lin C-R, Wang C-N, Ho T-I. Photochemistry of stilbene-amine. Spin-trapping study. J Org Chem, 1991, 56: 5025–5029
Molina V, Mercha’n M, Roos BO. Theoretical study of the electronic spectrum of trans-stilbene. J Phys Chem A, 1997, 101: 3478–3487
Rice JK, Baronavski AP. Ultrafast studies of solvent effects in the isomerization of cis-stilbene. J Phys Chem, 1992, 96: 3359–3366
Cao C, Chen G, Yin Z. Excited-state substituent constants σ exCC from substituted benzenes. J Phys Org Chem, 2008, 21: 808–815
Chen G, Cao C. Effect of the excited-state substituent constant σ exCC on the UV spectra of some 1,4-disubstituted benzenes. Chin J Chem Phys, 2009, 22(4): 366–370
Chen G, Cao C. Substituent effect on the UV spectra of p-disubstituted compounds XPh(CH=CHPh)nY (n = 0, 1, 2). J Phys Org Chem, 2010, 23: 776–782
Reichardt C. Solvatochromic dyes as solvent polarity indicators. Chem Rev, 1994, 2319–2358
Katritzky AR, Fara DC, Yang H, Tämm K. Quantitative measures of solvent polarity. Chem Rev, 2004, 104: 175–198
Katritzky AR, Fara DC, Kuanar M, Hur E, Karelson M. The classification of solvents by combining classical QSPR methodology with principal component analysis. J Phys Chem A, 2005, 109: 10323–10341
Seus EJ, Wilson CV. New synthesis of stilbene and heterocyclic stilbene analogs. J Org Chem, 1961, 26: 5243
Chen GF. Excited-state substituted constant σ exCC constructing and its applications in the UV spectra and FL spectra. Central South University, Ph D. dissertation, 2008
Hansch C, Leo A, Taft RW. A survey of hammett substituent constants and resonance and field parameters. Chem Rev, 1991, 91: 165–195
Klopman G, Li J-Y, Wang S, Dimayuga M. Computer automated logP calculation based on an extended group contribution approach. J Chem Inf Comput Sci, 1994, 34: 752–781
Liu L, Fu Y, Liu R, Li RQ, Guo QX. Hammett equation and generalized Pauling’s electronegativity equation. J Chem Inf Comput Sci, 2004, 44: 652–657
Gao S, Cao C. A new approach on estimation of solubility and n-octanol/water partition coefficient for organohalogen compounds. Int J Mol Sci, 2008, 9: 962–977
Jiang XK, Ji GZ, Wang DZ, Xie JR. Successful application of the spin-delocalization substituent constant σ. to the correlation of the UV spectra of some model compounds with para-Y-substituted phenyl groups. J Phys Org Chem, 1995, 8: 781–790
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cao, C., Chen, G. & Wu, Y. Effects of substituent and solvent on the UV absorption energy of 4,4′-disubstituted stilbenes. Sci. China Chem. 54, 1735–1744 (2011). https://doi.org/10.1007/s11426-011-4379-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4379-7