Abstract
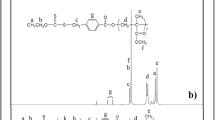
Hyperbranched poly(methyl methacrylate)s (HPMMAs) have been successfully prepared by atom transfer radical copolymerization of MMA and divinylbenzene (DVB). Kinetic study shows complete consumption of the initiator in 0.5 h, and relatively low polymerization rate when DVB content in the feed was high. By analyzing MALDI-TOF spectra of the resulting copolymers, the linear A n B* (n = 0, 1, 2, 3) oligomers were formed in 0.5 h of polymerization, and then the oligomers reacted each other to form dimers, further reactions produced HPMMA. The SEC and NMR spectroscopies were used to trace the polymerization, and the results demonstrate that small amount of the branching reactions occur in the initial polymerization, and the branched polymers are significantly generated past a certain conversion depending upon the feed ratios. Raising the content of DVB in the monomer mixture can increase the pendent vinyl groups of the linear oligo-inimers, leading to gelation at low MMA conversion.
Similar content being viewed by others
References
Kitajyo Y, Nawa Y, Tamaki M, Tani H, Takahashi K, Kaga H, Satoh T, Kakuchi T. A unimolecular nanocapsule: Encapsulation property of amphiphilic polymer based on hyperbranched polythreitol. Polymer, 2007, 48: 4683–4690
Gao Q, Liu PS. Crystalline-amorphous phase transition of hyperbranched polyurethane phase change materials for energy storage. J Mater Sci, 2007, 42: 5661–5665
Fréchet JMJ, Henmi M, Gitsov II, Aoshima S, Leduc M, Grubbs RB. Self-condensing vinyl polymerization: An approach to dendritic materials. Science, 1995, 269: 1080–1083
Jikei M, Kakimoto M. Hyperbranched polymers: A promising new class of materials. Prog Polym Sci, 2001, 26: 1233–1285.
Gao C, Yan D. Hyperbranched polymers: From synthesis to applications. Prog Polym Sci, 2004, 29: 183–275
Flory P. Molecular size distribution in three dimensional polymers. VI. Branched polymers containing A-R-Bf-1 type units. J Am Chem Soc, 1952, 74: 2718–2723
Vanjinathan M, Shanavas A, Raghavan A, Nasar AS. Synthesis and properties of hyperbranched polyurethanes, hyperbranched polyurethane copolymers with and without ether and ester groups using blocked isocyanate monomers. J. Polym Sci Part A: Polym. Chem, 2007, 45: 3877–3893
Zhu ZC, Pan CY. A Feasible synthetic route for linear PTHF-hyperbranched poly(phenyl sulfide) block copolymers Macromol Chem Phys, 2007, 208: 1274–1282
Hong CY, Pan CY. Synthesis and characterization of hyperbranched polyacrylates in the presence of a tetrafunctional initiator with higher reactivity than monomer by self-condensing vinyl polymerization. Polymer, 2001, 42: 9385–9391
He XH, Liang HJ, Pan CY. Monte Carlo simulation of hyper-branched copolymerizations in the presence of a multifunctional initiator. Macromol Theory Simul, 2001, 10: 196–203
Kong LZ, Pan CY. Synthesis and characterization of hyperbranched polymers from the polymerization of glycidyl methacrylate and styrene using Cp2TiCl as a catalyst. Macromol Chem Phys, 2007, 208: 2686–2697
Hawker CJ, Fréchet JMJ, Grubbs RB, Dao JL. Preparation of hyperbranched and star polymers by a “living”, self-condensing free radical polymerization. J Am Chem Soc, 1995, 117: 10763–10764
Yan DY, Hou J, Zhu XY, Kosman JJ, Wu HS. A new approach to control crystallinity of resulting polymers: Self-condensing ring opening polymerization. Macromol Rapid Commun, 2000, 21: 557–561
Matyjaszewski K, Gaynor SG, Kulfan A, Podwika M. Preparation of hyperbranched polyacrylates by atom transfer radical polymerization. 1. Acrylic AB* monomers in “living” radical polymerizations. Macromolecules, 1997, 30: 5192–5194
Zhou P, Yang LP, Pan CY. One-pot synthesis of linear-hyper-branched diblock copolymers via self-condensing vinyl polymerization and ring opening polymerization. J Polym Sci Part A: Polym Chem, 2008, 46: 7628–7636
Simon PFW, Müller AHE. Synthesis of hyperbranched and highly branched methacrylates by self-condensing group transfer copolymerization. Macromolecules, 2001, 34: 6206–6213
O’Brien N, McKee A, Sherrington DC, Slark AT, Titterton A. Facile, versatile and cost effective route to branched vinyl polymers. Polymer, 2000, 41: 6027–6031
Baskaran D. Synthesis of hyperbranched polymers by anionic self-condensing vinyl polymerization. Macromol Chem Phys, 2001, 202: 1569–1575
Baskaran D. Hyperbranched polymers from divinylbenzene and 1,3-diisopropenylbenzene through anionic self-condensing vinyl polymerization. Polymer, 2003, 44: 2213–2220
Isaure F, Cormack PAG, Graham S, Sherrington DC, Armes SP. Synthesis of branched poly(methyl methacrylate)s via controlled/living polymerisations exploiting ethylene glycol dimethacrylate as branching agent. Chem Commun, 2004, 9: 1138–1139
Bütün V, Bannister I, Billingham NC, Sherrington DC, Armes SP. Synthesis and characterization of branched water-soluble homopolymers and diblock copolymers using group transfer polymerization. Macromolecules, 2005, 38: 4977–4982
Li Y, Armes SP. Synthesis and chemical degradation of branched vinyl polymers prepared via ATRP: Use of a cleavable disulfide-based branching agent. Macromolecules, 2005, 38: 8155–8162
Wang AR, Zhu S. Branching and gelation in atom transfer radical polymerization of methyl methacrylate and ethylene glycol dimethacrylate. Polym Eng Sci, 2005, 45: 720–727
Bannister I, Billingham NC, Armes SP, Rannard SP, Findlay P. Development of branching in living radical copolymerization of vinyl and divinyl monomers. Macromolecules, 2006, 39: 7483–7492
Gong HD, Huang WY, Zhang DL, Gong FH, Liu CL, Yang Y, Chen JH, Jiang BB. Studies on the development of branching in ATRP of styrene and acrylonitrile in the presence of divinylbenzene. Polymer, 2008, 49: 4101–4108
Wang AR, Zhu SP. Control of the polymer molecular weight in atom transfer radical polymerization with branching/crosslinking. J Polym Sci, Part A: Polym Chem, 2005, 43: 5710–5714
Ren Q, Gong FH, Liu CL, Zhai GQ, Jiang BB, Liu C, Chen YH. Synthesis of branched polystyrene by ATRP exploiting divinylbenzene as branching comonomer. Eur Polym J, 2006, 42: 2573–2580
Gao H, Min K, Matyjaszewski K. Determination of gel point during atom transfer radical copolymerization with cross-linker. Macromolecules, 2007, 40: 7763–7770
Bouhier MH, Cormack PAG, Graham S, Sherrington DC. Synthesis of densely branched poly(methyl methacrylate)s via ATR copolymerization of methyl methacrylate and ethylene glycol dimethacrylate. J Polym Sci, Part A: Polym Chem, 2007, 45: 2375–2386
Gao H, Miasnikova A, Matyjaszewski K. Effect of cross-linker reactivity on experimental gel points during ATRcP of monomer and cross-linker. Macromolecules, 2008, 41: 7843–7849
París R, Mosquera B, Fuente JL. Atom transfer radical copolymerization of glycidyl methacrylate and allyl methacrylate, two functional monomers. Eur Polym J, 2008, 44: 2920–2926
Liu B, Kazlauciunas A, Guthrie JT, Perrier S. One-pot hyperbranched polymer synthesis mediated by reversible addition fragmentation chain transfer (RAFT) polymerization. Macromolecules, 2005, 38: 2131–2136
Liu B, Kazlauciunas A, Guthrie JT, Perrier S. Influence of reaction parameters on the synthesis of hyperbranched polymers via reversible addition fragmentation chain transfer (RAFT) polymerization. Polymer, 2005, 46: 6293–6299
Vo CD, Rosselgong J, Armes SP, Billingham NC. RAFT synthesis of branched acrylic copolymers. Macromolecules, 2007, 40: 7119–7125
Dong ZM, Liu XH, Lin Y, Li YS. Branched polystyrene with abundant pendant vnyl functional groups from asymmetric divinyl monomer. J Polym Sci, Part A: Polym Chem, 2008, 46, 6023-6034
Yang HJ, Jiang BB, Huang WY, Zhang DL, Kong LZ, Chen JH, Liu CL, Gong FH, Yu Q, Yang Y. Development of branching in atom transfer radical copolymerization of styrene with triethylene glycol dimethacrylate. Macromolecules, 2009, 42: 5976–5982
Odian G. Principles of Polymerization, New York: John Wiley & Sons. 1991. 480
Matyjaszewski K, Shipp DA, Wang JL, Grimaud T, Patten TE. Utilizing halide exchange to Improve control of atom transfer radical polymerization. Macromolecules, 1998, 31: 6836–6840
Matyjaszewski K, Wang JL, Grimaud T, Shipp DA. Controlled/“living” atom transfer radical polymerization of methyl methacrylate using various initiation systems. Macromolecules, 1998, 31: 1527–1534
Borman CD, Jackson AT, Bunn A, Cutter AL, Irvine DJ. Evidence for the low thermal stability of poly(methyl methacrylate) polymer produced by atom transfer radical polymerisation. Polymer, 2000, 41: 6015–6020
Jackson AT, Bunn A, Priestnall IM, Borman CD, Irvine DJ. Molecular spectroscopic characterisation of poly(methyl methacrylate) generated by means of atom transfer radical polymerisation (ATRP). Polymer, 2006, 47: 1044–1054
Singha NK, Rimmer S, Klumperman B. Mass spectrometry of poly (methyl methacrylate) (PMMA) prepared by atom transfer radical polymerization (ATRP). Eur Polym J, 2004, 40: 159–163
Hild G, Okasha R. Kinetic investigation of the free radical crosslinking copolymerization in the pre-gel state, 1. Styrene/m- and p-divinylbenzene systems. Macromol Chem, 1985, 186: 93–110
He XH, Liang HJ, Pan CY. Self-condensing vinyl polymerization in the presence of multifunctional initiator with unequal rate constants: Monte Carlo simulation. Polymer, 2003, 44: 6697–6706
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sun, M., Pan, C. Formation of hyperbranched polymers in atom transfer radical copolymerization of MMA and DVB. Sci. China Chem. 53, 2440–2451 (2010). https://doi.org/10.1007/s11426-010-4155-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-010-4155-0