Abstract
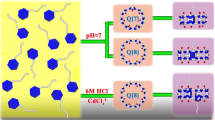
The slightly water-soluble anticancer drug camptothecin (CPT) and its inclusion complexes with cucurbit[n = 7, 8]uril (Q[n] (n = 7, 8)) were investigated. The formation of 1:2 complexes with Q[n] (n = 7, 8) in aqueous solution was confirmed by fluorescence spectroscopy and the apparent stability constants were determined to be higher than 3.01 × 1012 L2/mol2. The solid inclusion complexes of CPT and Q[n] (n = 7, 8) were also prepared by the co-evaporation method and characterized by Fourier transformation-infrared spectroscopy, differential scanning calorimetry and powder X-ray diffraction. Aqueous solubility and dissolution studies indicate that the complexes exhibited significantly increased dissolution rates compared with the pure drug and physical mixtures. The potential of Q[7] or Q[8] for stabilizing lactone modality of CPT was investigated by the High Performance Liquid Chromatography (HPLC) method. The results reveal more than 63% CPT lactone form (active form) in CPT-Q[7] or Q[8] complexes compared to only 36% CPT lactone form in the absence of Q[7] or Q[8] after being incubated in the phosphate buffer solution (pH 7.4 at 37°C) for 5 h.
Similar content being viewed by others
References
Wall ME, Wani MC, Cook CE. Plant antitumor agents. I. The isolation and structure of camptothecin, a novel alkaloidal leukemia and tumor inhibitor from camptotheca acuminata1. J Am Chem Soc, 1966, 88:3888–3890
Hertzberg RP, Caranfa MJ, Holden KG, Jakas DR, Gallagher G, Mattern MR, Mong SM, Bartus JO’L, Johnson RK, Kingsbury WD. Modification of the hydroxy lactone ring of camptothecin: Inhibition of mammalian topoisomerase and biological activity. J Med Chem, 1989, 32:715–720
Zhou CH, Zhang FF, Gan LL, Zhang YY, Geng RX. Review on supermolecules as chemical drugs. Sci China Series B-Chem, 2009, 52(4):415–458
Uekama K, Hirayama F, Irie T. Cyclodextrin drug carrier systems. Chem Rev, 1998, 98:2045–2076
Zhabg JJ, Shao Y, Wei L, Li Y, Sheng X, Liu F, Lu GY. Design of artificial nucleases and studies of their interaction with DNA. Sci China Series B-Chem, 2009, 52(4):402–414
Kim J, Jung IS, Kim SY, Lee E, Kang J, Sakamoto KS, Yamaguchi K, Kim K. New cucurbituril homologues: Syntheses, isolation, characterization, and X-ray crystal structures of cucurbit[n]uril (n = 5, 7, and 8). J Am Chem Soc, 2000, 122:540–541
Lagona J, Fettinger JC, Isaacs L. Cucurbit[n]uril analogues. Org Lett, 2003, 5:3745–3747
Lee JW, Samal S, Selvapalam N, Kim HJ, Kim K. Cucurbituril homologues and derivatives: New opportunities in supramolecular chemistry. Acc Chem Res, 2003, 36:621–630
Kim K, Selvapaam N, Ko YH, Park KM, Kim D, Kim J. Functionalized cucurbiturils and their applications. Chem Soc Rev, 2007, 36:267–279
Elemans JA, Rowan AW, Nolte AE. Self-assembled architectures from glycoluril. Ind Eng Chem Res, 2000, 39:3419–3428
Cintas P. Cucurbituril: Supramolecular perspectives for an old ligand. J Incl Phenom Molec Reco Chem, 1994, 17:205–220
Jon SY, Selvapalam N, Oh DH, Kang JK, Kim SY, Jeon YJ, Lee JW, Kim K. Facile synthesis of cucurbit[n]uril derivatives via direct functionalization: Expanding utilization of cucurbit[n]uril. J Am Chem Soc, 2003, 125:10186–10187
Wheate NJ, Day AI, Blanch RJ, Arnold AP, Cullinance C, Collins JG. Multi-nuclear platinum complexes encapsulated in cucurbit[n]uril as an approach to reduce toxicity in cancer treatment. Chem Commun, 2004, 1424–1425
Jeon YJ, Kim SY, Ko YH, Sakamoto S, Yamagchi K, Kim K. Novel molecular drug carrier: Encapsulation of oxaliplatin in cucurbit[7]uril and its effects on stability and reactivity of the drug. Org Biomol Chem, 2005, 3:2122–2125
Huang Y, Xue SF, Tao Z. Inclusion interaction between cucurbit[7]uril with Adenine and its derivatives. Supramol Chem, 2008, 20:279–287
Liu LX, Zhu SY. A study on the supramolecular structure of inclusion complex of β-cyclodextrin with prazosin hydrochloride. Carbohyd Polym, 2007, 68:472–476
Khan KA. The concept of dissolution efficiency. J Pharm Pharmacol, 1975, 27:48–49
Cong H, Tao LL, Yu YH, Yang F, Du Y, Xue SF, Tao Z. Molecular recognition of aminoacid by cucurbiturils. Acta Chim Sin, 2006, 64:989–996
Giordano F, Novak C, Moyano JR. Thermal analysis of cyclodextrins and their inclusion compounds. Therm Acta, 2001, 380:123–151
Ruiz MA, Reyes I, Parera A, Gallardo V. Determination of the stability of omeprazole by means of different scanning calorimetry. J Therm Anal, 1998, 51:29–35
Pinto LMA, Fraceto LF, Santana MHA, Pertinhez TA, Junior SO, Paula ED. Physico-chemical characterization of benzocaine-β-cyclodextrin inclusion complexes. J Pharm Biom Anal, 2005, 39: 956–963
Ning L, Zhang YH, Wu YN, Xiong XL, Zhang YH. Inclusion complex of trimethoprim with β-cyclodextrin (short communication). J Pharm Biom Anal, 2005, 39:824–829
Zingone G, Rubessa F. Preformulation study of the inclusion complex warfarin-[beta]-cyclodextrin. Int J Pharm, 2005, 291:3–10
Yap KL, Liu X, Thenmozhiyal JC, Ho PC. Characterization of the 13-cis-retinoic acid/cyclodextrin inclusion complexes by phase solubility, photostability, physicochemical and computational analysis. Eur J Pharm Sci, 2005, 25:49–56
Rajendrakumar K, Madhusudan S, Pralhad T. Cyclodextrin complexes of valdecoxib: Properties and anti-inflammatory activity in rat. Eur J Pharm Biopharm, 2005, 60:39–46
Marques HC, Hadgraft J, Kellaway I. Studies of cyclodextrin inclusion complexes. I. The salbutamol-cyclodextrin complex as studied by phase solubility and DSC. Int J Pharm, 1990, 63:259–266
Xu CY, Huang MZ, Xue CX. Synthesis and spectral characterization of 10-hydroxycamptothecin. Spectrosc Spect Anal, 2005, 25:1772–1774
Figuerias A, Carvalho RA, Ribeiro L, Torres-Labandeira JJ, Veiga FJB. Solid-state characterization and dissolution profiles of the inclusion complexes of omeprazole with native and chemically modified β-cyclodextrin. Eur J Pharm Biopharm, 2007, 67:531–539
Lerchen HG, Baumgarten J, Bruch KVD, Lehmann TE, Sperzel M, Kempka G, Fiebig HH. Design and optimization of 20-o-linked camptothecin glycoconjugates as anticancer agents. J Med Chem, 2001, 44:4186–4195
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dong, N., Dong, M., Zhao, A. et al. Preparation and characterization of inclusion complexes of antitumor camptothecin with cucurbit[n = 7, 8]urils. Sci. China Chem. 53, 2304–2310 (2010). https://doi.org/10.1007/s11426-010-4067-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-010-4067-z