Abstract
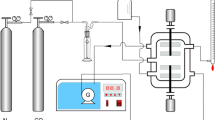
A novel alkanolamine-based ionic liquid, N-methyl-diethanolammonium tetrafluoroborate ([MDEA][BF4]), was synthesized in our laboratory. The ionic liquid-based composite solution consisting of N-methyl-diethanolamine (MDEA), [MDEA][BF4], piperazine (PZ) and H2O was investigated for CO2 capture. The optimal performance for CO2 capture was found at 45 °C, 1.50 MPa, probably due to a synergistic action of the reaction and the transport. No apparent corrosion was found on stainless and carbon steel with the above composite solution. This finding is very significant to the promotion of its engineering application.
Similar content being viewed by others
References
Haszeldine RS. Carbon capture and storage: How green can black be? Science, 2009, 325: 1647–1652
Bara JE, Camper DE, Gin DL, Noble RD. Room-temperature ionic liquids and composite materials: Platform technologies for CO2 capture. Acc Chem Res, 2009, 43: 152–159
Rochelle GT. Amine scrubbing for CO2 capture. Science, 2009, 325: 1652–1654
Speyer D, Ermatchkov V, Maurer G. Solubility of carbon dioxide in aqueous solutions of N-methyldiethanolamine and piperazine in the low gas loading region. J Chem Eng Data, 2010, 55: 283–290
Tounsi KNH, Barreau A, Le Corre E, Mougin P, Neau E. Measurement of carbon dioxide solubility in a solution of diethanolamine mixed with methanol. Ind Eng Chem Res, 2005, 44: 9239–9243
Sada E, Kumazawa H, Butt MA. Solubilities of gases in aqueous solutions of amine. J Chem Eng Data, 2002, 22: 277–278
Hirst LL. Pinkel II. Absorption of carbon dioxide by amines di- and triethanolamine and tetramine. Ind Eng Chem Res, 2002, 28: 1313–1315
Addicks J, Owren GA, Fredheim AO, Tangvik K. Solubility of carbon dioxide and methane in aqueous methyldiethanolamine solutions. J Chem Eng Data, 2002, 47: 855–860
Astarita GSDW, Bisio A. Gas treating with Chemical Solvents. New York: John Wiley & Sons, 1983
Camper D, Bara JE, Gin DL, Noble RD. Room-temperature ionic liquid-amine solutions: tunable solvents for efficient and reversible capture of CO2. Ind Eng Chem Res, 2008, 47: 8496–8498
Zhang XP, Zhao YS, Zeng SJ, Zhang SJ, Liu L, Tian X. Synthesis of a task-specific alcamine ionic liquids. CN. 200910093071.X
DiLabio GA, Pratt DA. Density functional theory based model calculations for accurate bond dissociation enthalpies. 2. Studies of X-X and X-Y (X, Y = C, N, O, S, Halogen) bonds. J Phys Chem A, 2000, 104: 1938–1943
Zhang SJ, Yuan XL, Chen YH, Zhang XP. Solubilities of CO2 inbutyl-3-methylimidazolium hexafluorophosphate and 1,1,3,3-tetramethylguanidium lactate at elevated pressures. J Chem Eng Data, 2005, 50: 1582–1585
Bates ED, Mayton RD, Ntai L, Davis JH. CO2 capture by a taskspecific ionic liquid. J Am Chem Soc, 2002, 124: 926–927
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, S., Zhang, X., Zhao, Y. et al. A novel ionic liquids-based scrubbing process for efficient CO2 capture. Sci. China Chem. 53, 1549–1553 (2010). https://doi.org/10.1007/s11426-010-4030-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-010-4030-z