Abstract
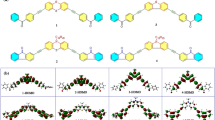
Four photopolymerization initiators with D-π-D (D, donor; π, conjugation system) structure have been synthesized by solvent-free reaction and characterized by 1H NMR spectroscopy, IR and elemental analysis. The one-photon and two-photon excited fluorescence have been investigated in different solvents. Experimental results of the one-photon and two-photon absorption cross sections show different trends in OPA and TPA ability with different substitution groups in donor units.
Similar content being viewed by others
References
Abbotto A, Beverina L, Bradamante S, Bradamante S, Bozio R, Ferrante C, Pagani G A, Signorini R. Push-pull organic chromophores for frequency up-converted lasing. Adv Mater, 2000, 12: 1963–1967
Diaspro A, Robello M. Two photon excitation of fluorescence for three dimensional optical imaging of biological structures. J Photochem Photobiol B Biol, 2000, 55: 1–8
Dyer D J, Cumpston B H, McCord-Maughon D, Thaynmanavan S, Barlow S, Perry J W, Marder S R. Turning on fluorescence by two-photon excitation and polymerization: Toward a 3-D optical memory device. Nonlinear Optics, Quantum Optics, 2004, 31: 175–184
Li X, Zhao Y X, Wang T, Shi M Q, Wu F P. Coumarin derivatives with enhanced two photon absorption cross-sections. Dyes Pigm, 2007, 74: 108–112
Qian Y, Lu Z F, Lu C G, Chen Z M, Cui Y P. Synthesis and two-photon absorption properties of 2,5-bis[4-(2-arylvinyl)phenyl]-1,3,4-oxadiazoles. Dyes Pigm, 2007, 75: 641–646
Wang Z M, Wang X M, Zhao J F, Jiang W L, Yang P, Fang X Y. Cooperative enhancement of two-photon absorption based on electron coupling in triphenylamine-branching chromophore. Dyes Pigm, 2008, 79: 145–152
Terenziani F, le Droumaguet C, Katan C, Mongin O, Blanchard-Desce M. Effect of branching on two-photon absorption in triphenyl-benzene derivatives. ChemPhysChem, 2007, 8: 723–734
Signorini R, Ferrante C, Pedron D, Zerbetto M, Cecchetto E, Slaviero M, Fortunati I, Collini E, Bozio R, Abbotto A, Beverina L, Pagani G A. Effective two-photon absorption cross section of heteroaromatic quadrupolar dyes: dependence on measurement technique and laser pulse characteristics. J Phys Chem A, 2008, 112: 4224–4234
Tian Y Q, Chen C Y, Cheng Y J, Young A C, Tucker N M, Jen A K Y. Hydrophobic chromophores in aqueous micellar solution showing large two-photon absorption cross sections. Adv Funct Mater, 2007, 17: 1691–1697
Fan H H, Chen X, Wang H Z, Tian Y P. Structure-property relationships in a new series of two-photon absorbing D-π-D divinyl-biphenyl derivatives. Chem Phys Lett, 2007, 436: 171–174
Feng Y L, Yan Y L, Wang S, Zhu W H, Qian S X, Tian H. Photochromic thiophene oligomers based on bisthienylethene: syntheses, photochromic and two-photon properties. J Mater Chem, 2006, 16: 3685–3692
Kato S I, Matsumoto T, Shigeiwa M, Gorohmaru H, Maeda S, Ishii I T, Mataka S. Novel 2,1,3-benzothiadiazole-based red-fluorescent dyes with enhanced two-photon absorption cross-sections. Chem Eur J, 2006, 12: 2303–2317
Wang C K, Macak P, Luo Y. Effects of π centers and symmetry on two photon absorption cross sections of organic chromophores. J Chem Phys, 2001, 114: 9813–9820
Bauer C, Schnabel B, Kley E B, Scherf U, Giessen H, Mahrt R F. Two-photon pumped lasing from a two-dimensional photonic band-gap structure with polymeric gain material. Adv Mater, 2002, 14: 673–676
He G S, Tan L S, Zheng Q D, Prasad P N. Multiphoton absorbing materials: Molecular designs, characterizations and applications. Chem Rev, 2008, 108: 1245–1330
Ehrlich J, Wu X, Lee I Y S, Hu Z Y, Röckel H, Marder S R, Perry J. Two-photon absorbing for biological imaging. Opt Lett, 1997, 22: 1843–1845
Kawatta S, Kawatta Y. Three-dimensional optical data storage using photochromic materials. Chem Rev, 2000, 100: 1777–1788
Ogawa K, Hasegawa H, Inaba Y, Kobuke Y, Inouye H, Kanemitsu Y, Kohno E, Hirano T, Ogura S I, Okura I. Water-soluble bis-(imida-zolylporphyrin) self-assemblies with large two-Photon absorption cross sections as potential agents for photodynamic therapy. J Med Chem, 2006, 49: 2276–2283
Bhaskar A, Ramakrishna G, Lu Z K, Twieg R, Hales J M, Hagan D J, Stryland E V, Goodson T III. Investigation of two-photon absorption properties in branched alkene and alkyne chromohhores. J Am Chem Soc, 2006, 128: 11840–11849
Woo H Y, Liu B, Kohler B, Korystov D, Mikhailovsky A, Bazan G. C. Solvent effects on the two-photon absorption of distyrylbenzene chromophores. J Am Chem Soc, 2005, 127: 14721–14729
Balema V P, Wiench J W, Pruski M, Pecharsky V K. Mechanically induced solid-State generation of Phosphorus ylides and the solvent-free wittig reaction. J Am Chem Soc, 2002, 124: 6244–6245
Ren Y, Fang Q, Yu W T, Lei H, Tian Y P, Jiang M H, Yang Q C, Thomas C K M. Synthesis, structures and two-photon pumped up-conversion lasing properties of two new organic salts. J Mater Chem, 2000, 10: 2025–2030
Wang C K, Zhao K, Su Y, Ren Y, Zhao X, Luo Y. Solvent effects on the electronic structure of a newly synthesized two-photon polymerization initiator. J Chem Phys, 2003, 119: 1208–1213
Author information
Authors and Affiliations
Additional information
Supported by the National Natural Science Foundation of China (Grant Nos. 50532030, 50703001 & 20771001), the National Natural Science Foundation of Anhui Province (Grant No. 070414188), Doctoral Program Foundation of the Ministry of Education of China, Education Committee of Anhui Province (Grant No. 2006KJ032A & KJ2009A52), Team for Scientific Innovation Foundation of Anhui Province (Grant No. 2006KJ007TD), Young Teacher Foundation of Institution of High Education of Anhui Province (Grant No. 2007jq1019), Ministry of Education and Person with Ability Foundation of Anhui University
Rights and permissions
About this article
Cite this article
Gan, X., Zhou, H., Shi, P. et al. A new series of two-photon polymerization initiators: Synthesis and nonlinear optical properties. Sci. China Ser. B-Chem. 52, 2180–2185 (2009). https://doi.org/10.1007/s11426-009-0276-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-009-0276-8