Abstract
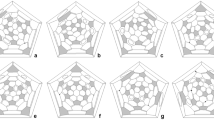
To gain insight into the structures and stability of F4F6 polyhedrons formed by squares and hexagons, a density functional theory study was performed on all isomers of F4F6 polyhedrons with sizes from 8 to 60. The calculated results demonstrate that the six squares tend to isolate from each other, i.e. these F4F6 polyhedrons obey the isolated square rule. Those isomers with fewer B44 bonds (square-square adjacencies) are more stable than those with more B44 bonds, i.e. they obey square adjacency penalty rule. Both of the two rules in F4F6 polyhedrons are in the same status as the isolated pentagon rule and pentagon adjacency penalty rule in F5F6 fullerenes and can be used to screen the lowest energy isomers of F4F6 polyhedrons as IPR and PAPR do in classic fullerenes. Structural analysis demonstrates that the pyramidalization of carbon atoms at the square-square adjacencies determines the stability of corresponding structures.
Similar content being viewed by others
References
Kroto H W, Heath J R, O’Brien S C, Curl R F, Smalley R E. C60: Buckminsterfullerene. Nature, 1985, 318: 162–163
Kroto H W. The stability of the fullerenes Cn, with n = 24, 28, 32, 36, 50, 60 and 70. Nature, 1987, 329: 529–531
Prinzbach H, Weiler A, Landen-berger P, Wahi F, Worth J, Scott L T, Gelmont M, Olevano D, Issendorff B. Gas-phase production and photoelectron spectroscopy of the smallest fullerene, C20. Nature, 2000, 407: 60
Buhl M, Hirsch A. Spherical aromaticity of fullerenes. Chem Rev, 2001, 101: 1153–1184
Xie S Y, Gao F, Lu X, Huang R B, Wang C R, Zhang X, Liu M L, Deng S L, Zheng L S. Capturing the Labile Fullerene[50] as C50Cl10. Science, 2004, 304: 699
Chen Z, King R B. Spherical aromaticity: Recent work on fullerenes, polyhedral boranes, and related structures. Chem Rev, 2005, 105: 3613–3642
Lu X, Chen Z. Curved Pi-conjugation, aromaticity, and the related chemistry of small fullerenes <C60 and single-walled carbon nanotubes. Chem Rev, 2005, 105: 3643–3696
Diaz-Tendero S, Martin F, Alcami M. Structure and electronic properties of fullerenes C52 q+: Is C52 2+ an exception to the pentagon adjacency penalty rule? Chem Phys Chem, 2005, 6: 92–100
Campbell E E B, Fowler P W, Mitchell D, Zerbetto F. Increasing cost of pentagon adjacency for larger fullerenes. Chem Phys Lett, 1996, 250: 544–548
Albertazzi E, Domene C, Fowler P W, Heine T, Seifert G, Alsenoyd C V, Zerbettoe F. Pentagon adjacency as a determinant of fullerene stability. Phys Chem Chem Phys, 1999, 1: 2913–2918
Slanina Z, Ishimura K, Kobayashi K, Nagase S. C72 isomers: The IPR-satisfying cage is disfavored by both energy and entropy. Chem Phys Lett, 2004, 384: 114–118
Díaz-Tendero S, Alcamí M, Martín F. Fullerene C50: Sphericity takes over, not strain. Chem Phys Lett, 2005, 407: 153–158
Sun M L, Slanina Z, Lee S L. Square/hexagon route towards the boron-nitrogen clusters. Chem Phys Lett, 1995, 233: 279–283
Wu H S, Xu X H, Stout D L, Jiao H. The structure and stability of B N cages: A computational study. J Mol Model, 2005, 12: 1–8
Fowler P W, Heine T, Manolopoulos D E, Mitchell D, Orlandi G, Schmidt R, Seifert G, Zerbetto F. Energetics of fullerenes with four-membered rings. J Phys Chem, 1996, 100: 6984–6991
Gao Y D, Herndon W C. Fullerenes with four-membered rings. J Am Chem Soc, 1993, 115: 8459–8460
Qian W Y, Bartberger M D, Pastor S J, Houk K N, Wilkins C L, Rubin Y. C62, a non-classical fullerene incorporating a four-membered ring. J Am Chem Soc, 2000, 122: 8333–8334
Ayuela A, Fowler P W, Mitchell D, Schmidt R, Seifert G, Zerbetto F. C62: Theoretical evidence for a nonclassical fullerene with a heptagon. J Phys Chem, 1996, 100: 15634–15636
Domene M C, Fowler P W, Mitchell D, Seifert G, Zerbetto F. Energetics of C20 and C22 fullerene and near-fullerene carbon cages. J Phys Chem A, 1997, 101: 8339–8344
Cui Y H, Chen D L, Tian W Q, Feng J K. Structures, stabilities, and electronic and optical properties of C62 fullerene isomers. J Phys Chem A, 2007, 111: 7933–7939
Haddon R C. Measure of nonplanarity in conjugated organic molecules: Which structurally characterized molecule displays the highest degree of pyramidalization? J Am Chem Soc, 1990, 112: 3385–3389
Haddon R C. Chemistry of the fullerenes: The manifestation of strain in a class of continuous aromatic molecules. Science, 1993, 261: 1545–1550
Bürgi H B, Blanc E, Schwarzenbach D, Liu S Z, Lu Y J, Kappes M M, Ibers J A. The structure of C60: Orientational disorder in the low-temperature modification of C60. Angew Chem Int Ed, 1992, 31: 640–643
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R, Montgomery J A, Vreven T, Kudin K N, Burant J C, Millam J M, Iyengar S S, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson G A, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda, Y, Kitao O, Nakai H, Klene M, Li X, Knox J E, Hratchian H P, Cross J B, Adamo C, Jaramillo J, Gomperts R, Stratmann R E, Yazyev O, Austin A J, Cammi R, Pomelli C, Ochterski J W, Ayala P Y, Morokuma K, Voth G. A, Salvador P, Dannenberg J J, Zakrzewski V G, Dapprich S, Daniels A D, Strain M C, Farkas O, Malick D K, Rabuck A D, Raghavachari K, Foresman J B, Ortiz J V, Cui Q, Baboul A G, Clifford S, Cioslowski J, Stefanov B B, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin R L, Fox D J, Keith T, Al-Laham M A, Peng C Y, Nanayakkara A, Challacombe M, Gill P M W, Johnson B, Chen W, Wong M W, Gonzalez C, Pople J A. Gaussian 03, Revision B, 05. Gaussian Inc, Pittsburgh, PA, 2003
Balaban A T. From chemical topology to 3D geometry. J Chem Inf Comput Sci, 1997, 37: 645–650
Schmalz T G, Seitz W A, Klein D J, Hite G E. Elemental carbon cages. J Am Chem Soc, 1988, 110: 1113–1127
Qian W, Chuang S C, Amador R B, Jarrosson T, Sander M, Pieniazek S, Khan S I, Rubin Y. Synthesis of stable derivatives of C62: The first nonclassical fullerene incorporating a four-membered ring. J Am Chem Soc, 2003, 125: 2066–2067
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported from Southwest University, China (Grant No. SWNUB2005002) and Key Laboratory of the Three Gorges Reservoir Region’s Eco-Environment within Ministry of Education, Chongqing University (Grant No. KLVF-2007-5)
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gan, L., Liu, J., Zou, J. et al. Carbon polyhedrons formed by squares and hexagons obeying isolated square rule. Sci. China Ser. B-Chem. 52, 1085–1089 (2009). https://doi.org/10.1007/s11426-009-0139-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-009-0139-3