Abstract
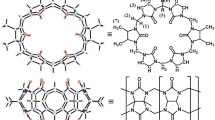
Interactions between a symmetrical tetramethyl-substituted cucurbit[6]uril (host: TMeQ[6]) and 1,ω-alkylenedipyridine (ω = 2, 4, 6, 8, 10) dicationic guests were investigated using 1H NMR spectroscopy and single crystal X-ray crystallography. In these inclusion complexes, combined cavity and portal binding in TMeQ[6] were observed, and the length of the bridged alkylene was found to play an important role not only in balancing the overall hydrophilic/hydrophobic interaction between the host and the guest, but also in defining the structure of the resulting inclusion complexes. For the guest 1,2-ethylenedipyridine (Edpy), TMeQ[6] includes a positively charged pyridine ring of Edpy to form an unsymmetrical inclusion complex; for the guest 1,4-butylenedipyridine (Bdpy), TMeQ[6] includes a positively charged pyridine ring of Bdpy, but the different competitive interactions in and between the related inclusion complexes could lead to a fast exchange between the hosts and guests. For the guests with longer bridge chains, such as 1,6-hexamethylenedipyridine (Hdpy) or 1,8-octylenedipyridine (Odpy), a stable pseudorotaxane inclusion complex is formed by combining the hydrophobic cavity and the outer portal dipole-ion interactions. However, for 1,10-decatylenedipyridine (Ddpy), the two TMeQ[6] host molecules include the two end pyridine rings of Ddpy and form a dumbbell inclusion complex.
Similar content being viewed by others
References
Freeman W A, Mock W L. Cucurbituril. J Am Chem Soc, 1981, 103(24): 7367–7368
Day A I, Arnold A P, Blanch R J. WO 2000/068232, 2000-11-16
Kim J, Jung I S, Kim S Y, Lee E, Kang J K, Sakamoto S, Yamaguchi K, Kim, K. New cucurbituril homologues: Syntheses, isolation, characterization, and X-ray crystal structures of cucurbit[n]uril (n = 5, 7, and 8). J Am Chem Soc, 2000, 122(3): 540–541
Day A I, Blanck R J, Amold A P, Lorenzo S, Lewis G R, Dance I. A cucurbituril-based gyroscane: A new supramolecular form. Angew Chem Int Ed, 2002, 41(2): 275–277
Lagona J, Mukhopadhyay P, Chakrabarti S, Isaacs L. The cucurbit[ n]uril family. Angew Chem Int Ed, 2005, 44(31): 4844–4870
Lee J W, Samal S, Selvapalam N, Kim H J, Kim K. Cucurbituril homologues and derivatives: New opportunities in supramolecular chemistry. Acc Chem Res, 2003, 36: 621–630
Gerasko O A, Samsonenko D G, Fedin V P. Supramolecular chemistry of cucurbiturils. Russian Chem Rev, 2002, 71: 741–760
Elemans J A A W, Rowan A E, Nolte R J M. Self-assembled architectures from glycoluril. Ind Eng Chem Res, 2000, 39: 3419–3428
Hubin T J, Kolchinski A G, Vance A L, Busch D H. Template control of supramolecular architecture. Adv Supramol Chem, 1999, 5: 237–357
Cintas P. Cucurbituril: Supramolecular perspectives for an old ligand. J Incl Phenom Molec Rec Chem, 1994, 17: 205–220
Jeon Y M, Kim J, Whang D, Kim K. Molecular container assembly capable of controlling binding and release of its guest molecules: Reversible encapsulation of organic molecules in sodium ion complexed cucurbituril. J Am Chem Soc, 1996, 118(40): 9790–9791
Sun X Z, Li B, Zhou Q B, Zhang H B, Cheng G Z, Zhou X H. Pseudopolyrotaxanes of cucurbit[6]uril: A novel three-dimensional network self-assembled by (H2O)3 clusters and Br-(H2O)3 anion clusters. Crys Growth Des, 2008, 8(8): 2970–2974
Zhao J, Kim H J, Oh J, Kim S Y, Lee J, Sakamoto W S, Yamaguchi K, Kim K. Cucurbit[n]uril derivatives soluble in water and organic solvents. Angew Chem Int Ed, 2001, 40: 4233–4235
Isobe H, Sato S, Nakamura E. Synthesis of disubstituted cucurbit[6]uril and its rotaxane derivative. Org Lett, 2002, 4(8): 1287–1289
Jon S Y, Selvapalam N, Oh, D H, Kang J K, Kim S Y, Jeon Y J, Lee J W, Kim K. Synthesis of cucurbit[n]uril derivatives via direct functionalization: Expanding utilization of cucurbit[n]uril. J Am Chem Soc, 2003, 125(34): 10186–10187
Lagona J, Wagner B D, Isaacs L. Molecular-recognition properties of a water-soluble cucurbit[6]uril analogue. J Org Chem, 2006, 71(3): 1181–1190
Day A I, Arnold A P, Blanch R J. A method for synthesizing partially substituted cucurbit[n]uril. Molecules, 2003, 8(1): 74–84
Lagona J, Fettinger J C, Isaacs L. Cucurbit[n]uril analogues: Synthetic and mechanistic studies. J Org Chem, 2005, 70 (25): 10381–10392
Wagner B D, Boland P G, Lagona J, Isaacs L. A cucurbit[6]uril analogue: host properties monitored by fluorescence spectroscopy. J Phys Chem B, 2005, 109 (16): 7686–7691
Sasmal S, Sinha M K, Keinan E. Facile purification of rare cucurbiturils by affinity chromatography. Org Lett, 2004, 6(8): 1225–1228
Zhao Y J, Xue S F, Zhu Q J, Tao Z, Zhang J X, Wei Z B, Long L S, Hu M L, Xiao H P, Day A I. Synthesis of a symmetrical tetrasubstituted cucurbit[6]uril and its host-guest inclusion complex with 2,2′-bipyridine. Chin Science Bull, 2004, 49(11): 1111–1116
Zheng L M, Zhu Q J, Zhu J N, Zhang Y Q, Tao Z, Xue S F, Wei Z B, Long L S. Synthesis and crystal structure of a novel self-assembled (1,4-discyclohexyl cucurbituril) sodium(I) complex. Chin J Inorg Chem (in Chinese), 2005, 21(10): 1583–1588
Sheldrick G M. SHELXL97, Program for Crystal Structure Determination. University of Göttingen, 1997
Fu H Y, Xue S F, Zhu Q J, Tao Z, Zhang J X, Day A I. Investigation of host-guest compounds of cucurbit[n=5–8]uril with some ortho aminopyridines and bispyridine. J Inc Phenom Macro Chem, 2005, 52(1), 101–107
Ko Y H, Kim K, Kang J K, Chun H, Lee J W, Sakamoto S, Yamaguchi K, Fettinger J C, Kim K. Designed self-assembly of molecular necklaces using host-stabilized charge-transfer interactions. J Am Chem Soc, 2004, 126(7): 1932–1937
Lee J W, Kim K, Choi S W, Ko Y H, Sakamoto S, Yamaguchi K, Kim K. Unprecedented host-induced intramolecular charge-transfer complex formation. Chem Commun, 2002: 2692–2693
Xu Z Q, Yao X Q, Xue S F, Zhu Q J, Tao Z, Zhang J X, Wei Z B, Long L S. Investigation of structures of the self-assembled pseudorotaxane of cucurbiturils with some alkyldiamines. Acta Chim Sinica (in Chinese), 2004, 62(19): 1927–1936
Marquez C, Hudgins R R, Nau, W M. Mechanism of host-gest complexation by cucurbituril. J Am Chem Soc, 2004, 126(18): 5806–5816
Marquez C, Nau, W M. Two mechanisms of slow host-guest complexation between cucurbit[6]uril and cyclohexylmethylamine: pHresponsive supramolecular kinetics. Angew Chem Int Ed, 2001, 40(17): 3155–3160
Sindelar V, Silvi S, Kaifer A E. Switching a molecular shuttle on and off: Simple, pH-controlled pseudorotaxanes based on cucurbit[7]uril. Chem Commun, 2006, 20: 2185–2187
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Grant Nos. 20662003 & 20767001), the International Collaborative Project of Guizhou Province (Grant No. 2007400108), the Science Technology Fund of Guizhou Province (Grant No. J-2008-2012) and the Natural Science Youth Foundation of Guizhou University (Grant No. 2007-005)
Rights and permissions
About this article
Cite this article
Xiao, X., Zhang, Y., Zhu, Q. et al. Host-guest complexes of a water soluble cucurbit[6]uril derivative with some dications of 1,ω-alkyldipyridines: 1H NMR and X-ray structures. Sci. China Ser. B-Chem. 52, 475–482 (2009). https://doi.org/10.1007/s11426-009-0089-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-009-0089-9