Abstract
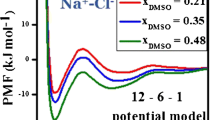
Constrained molecular dynamics simulations have been used to investigate the LiCl and NaCl ionic association in water in terms of atom-bond electronegativity equalization method fused into molecular mechanics (ABEEM/MM). The simulations make use of the seven-site fluctuating charge and flexible ABEEM-7P water model, based on which an ion-water interaction potential has been constructed. The mean force and the potential of mean force for LiCl and NaCl in water, the charge distributions, as well as the structural and dynamical properties of contact ion pair dissociation have been investigated. The results are reasonable and informative. For LiCl ion pair in water, the solvent-separated ion pair configurations are more stable than contact ion pair configurations. The calculated PMF for NaCl in water indicates that contact ion pair and solvent-separated ion pair configurations are of comparable stability.
Similar content being viewed by others
References
Simon J D, Peters K S. Picosecond studies of organic photoreactions. Acc Chem Res, 1984, 17: 277–283
Spears K G, Gray T H, Huang D. Ionic photodissociation and picosecond solvation dynamics of contact ion pairs. J Phys Chem, 1986, 90: 779–790
Spohn P D, Brill T B. Raman spectroscopy of the species in concentrated aqueous solutions of zinc nitrate, calcium nitrate, cadmium nitrate, lithium nitrate and sodium nitrate up to 450 degree C and 30 Mpa. J Phys Chem, 1989, 93: 6224–6231
Fleissner G, Hallbrucker A, Mayer E. Increasing contact ion pairing in the supercooled and glassy states of “dilute” aqueous magnesium, calcium, and strontium nitrate solution: Implications for biomolecules. J Phys Chem, 1993, 97: 4806–4814
Simonet V, Calzavara Y, Hazemann J L, Argoud R, Geaymond O, Raoux D. X-ray absorption spectroscopy studies of ionic association in aqueous solutions of zinc bromide from normal to critical conditions. J Chem Phys, 2002, 117: 2771–2781
Carnie S L, Patey G N. Fluids of polarizable hard spheres with dipoles and tetrahedral quadrupoles integral equation results with application to liquid water. Mol Phys, 1982, 47: 1129–1151
Pettitt B M, Rossky P J. Alkali halides in water: Ion-solvent correlations and ion-ion potentials of mean force at infinite dilution. J Chem Phys, 1986, 84: 5836–5844
Morita T, Ladanyi B M, Hynes J T. Polar solvent contributions to activation parameters for model ionic reactions. J Phys Chem, 1989, 93: 1386–1392
Hummer G, Soumpasis D M. An extended RISM study of simple electrolytes: pair correlations in a NaCl-SPC water model. Mol Phys, 1992, 75: 633–651
Kovalenko A, Hirata F. Potentials of mean force of simple ions in ambient aqueous solution. I. Three-dimensional reference interaction site model approach. J Chem Phys, 2000, 112: 10391–10402
Belch A C, Berkowitz M, McCammon J A. Solvation structure of a sodium chloride ion pair in water. J Am Chem Soc, 1986, 108: 1755–1761
Karim O A, McCammon J A. Dynamics of a sodium chloride ion pair in water. J Am Chem Soc, 1986, 108: 1762–1766
Jorgensen W L, Buckner J K, Houston S E, Rossky P J. Hydration and energetics for (CH3)3CCl ion pairs in aqueous solution. J Am Chem Soc, 1987, 109: 1891–1899
Buckner J K, Jorgensen W L. Energetics and hydration of the constituent ion pairs of tetramethylammonium chloride. J Am Chem Soc, 1989, 111: 2507–2516
Ciccotti G, Ferrario M, Hynes J T, Kapral R. Dynamics of ion pair interconversion in a polar solvent. J Chem Phys, 1990, 93: 7137–7147
Smith D E, Dang L X. Computer simulations of NaCl association in polarizable water. J Chem Phys, 1994, 100: 3757–3766
Rey R, Guàrdia E. Dynamical aspects of the Na+-Cl− ion pair association in water. J Phys Chem, 1992, 96: 4712–4718
Dang L X, Rice J E, Kollmann P A. The effect of water models on the interaction of the sodium-chloride ion pair in water: Molecular dynamics simulations. J Chem Phys, 1990, 93: 7528–7529
Smith D E, Haymet A D J. Structure and dynamics of water and aqueous solutions: The role of flexibility. J Chem Phys, 1992, 96: 8450–8459
Berkowitz M, Karim O A, McCammon J A, Rossky P J. Sodium chloride ion pair interaction in water: Computer simulation. Chem Phys Lett, 1984, 105: 577–580
Zhu S-B, Robinson G W. Molecular-dynamics computer simulation of simulation of an aqueous NaCl solution: Structure. J Chem Phys, 1992, 97: 4336–4348
Bader J S, Chandler D. Computer simulation study of the mean forces between ferrous and ferric ions in water. J Phys Chem, 1992, 96: 6423–6427
Chialvo A A, Cummings P T, Cochran H D, Simonson J M, Mesmer R E. Na+-Cl− ion pair association in supercritical water. J Chem Phys, 1995, 103: 9379–9387
Shinto H, Morisada S, Miyahara M, Higashitani K. A reexamination of mean force potentials for the methane pair and the constituent ion pairs of NaCl in water. J Chem Eng Jpn, 2003, 36: 57–65
Zhang Z G, Duan Z H. Lithium Chloride ionic association in dilute aqueous solution: A constrained molecular dynamics study. Chem Phys, 2004, 297: 221–233
Torrie G M, Valleau J P. Nonphysical sampling distributions in Monte Carlo free-energy estimation: Umbrella sampling. J Comput Phys, 1977, 23: 187–199
Pangali C, Rao M, Berne B J. A Monte Carlo simulation of the hydrophobic interaction. J Chem Phys, 1979, 71: 2975–2981
Yang Z Z, Wu Y, Zhao D X. Atom-bond electronegativity equalization method fused into molecular mechanics. I. A seven-site fluctuating charge and flexible body water potential function for water clusters. J Chem Phys, 2004, 120: 2541–2557
Wu Y, Yang Z Z. Atom-bond electronegativity equalization method fused into molecular mechanics. II. A seven-site fluctuating charge and flexible body water potential function for liquid water. J Phys Chem A, 2004, 108: 7563–7576
Yang Z Z, Qian P. A study of N-methylacetamide in water cluster: Based on atom-bond electronegativity equalization method fuse into molecular mechanics. J Chem Phys, 2006, 125(6): 064311–064326
Qian P, Yang Z Z. Application of ABEEM/MM model to study the properties of the water clusters (H2O)n, n=7−10. Sci Chin Ser B-Chem, 2007, 50: 190–204
Li X, Yang Z Z. Study of lithium cation in water clusters: Based on atom-bond electronegativity equalization method fused into molecular mechanics. J Phys Chem A, 2005, 109: 4102–4111
Li X, Yang Z Z. Hydration of Li+-ion in atom-bond electronegativity equalization method-7p water: A molecular dynamics simulation study. J Chem Phys, 2005, 122: 084514–084528
Yang Z Z, Li X. Ion solvation in water from molecular dynamics simulation from the ABEEM/MM force field. J Phys Chem A, 2005, 109: 3517–3520
Bultinck P, Langenaeker W, Lahorte P, Proft F D, Geerlings P, Waroquier M, Tollenaere J P. The electronegativity equalization method I: Parameterization and validation for atomic charge calculations. J Phys Chem A, 2002, 106: 7887–7894
Bultinck P, Langenaeker W, Lahorte P, Proft F D, Geerlings P, Alsenoy C V, Tollenaere J P. The electronegativity equalization method II: Application of different atomic charge schemes. J Phys Chem A, 2002, 106: 7895–7901
Chelli R, Procacci P. A transferable polarizable electrostatic force field for molecular mechanics based on the chemical potential equalization principle. J Chem Phys, 2002, 117: 9175–9181
Smirnov K S, van de Graaf B. Consistent implementation of the electronegativity equalization method in molecular mechanics and molecular dynamics. J Chem Soc Faraday Trans, 1996, 92: 2469–2474
Yang Z Z, Wang C S. Atom-bond electronegativity equalization method. 1. Calculation of the charge distribution in large molecules. J Phys Chem A, 1997, 101: 6315–6321
Wang C S, Yang Z Z. Atom-bond electronegativity equalization method. II. Lone-pair electron model. J Chem Phys, 1999, 110: 6189–6197
Yang Z Z, Wang C S. Atom-bond electronegativity equalization method and its applications based on density functional theory. J Theor Comput Chem, 2003, 2: 273–299
Yang Z Z, Cui B Q. Atomic charge calculation of metallobiomolecules in terms of the ABEEM method. J Chem Theory Comput, 2007, 3: 1561–1568
Berendsen H J C, Postma J P M, van Gunsteren W F, DiNola A, Haak J R. Ionization potentials of polyacene molecules in micellar systems or in liquid homogeneous solutions. J Chem Phys, 1984, 81: 3684–3689
Steinbach P J, Brooks B R. New spherical-cutoff methods for long-range forces in macromolecular simulation. J Comput Chem, 1994, 15: 667–683
Hynes J T, Baer M, eds. The Theory of Chemical Reaction Dynamics, Chemical Rubber, Vol. IV, Boca Raton, FL, 1985. 171
Carter E A, Cicotti G, Hynes J T, Kapral R. Constrained reaction coordinate dynamics for the simulation of rare events. Chem Phys Lett, 1989, 156: 472–477
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Grant Nos. 20633050 and 20703022)
Rights and permissions
About this article
Cite this article
Li, X., Gong, L. & Yang, Z. Molecular dynamics simulations of LiCl association and NaCl association in water by means of ABEEM/MM. Sci. China Ser. B-Chem. 51, 1221–1230 (2008). https://doi.org/10.1007/s11426-008-0129-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-008-0129-x