Abstract
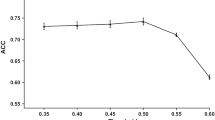
In the MHC class I molecule binding antigenic peptides processing and presentation pathway, the ubiquitin-proteasome system plays a key role in degrading the protein substrate. For the purpose of studying the specificities of proteasomal cleavage sites, partial least squares method is used to predict the proteasomal cleavage sites, and the predictive accuracy of the model is 82.8%. The specificities of the cleavage sites and the adjacent positions come from the contribution of the amino acids of the samples to the cleavage sites, showing the information of proteasome interacting with antigen protein. It demonstrates that the proteasome cleaving to target protein is selective, but not random.
Similar content being viewed by others
References
Adams J. The proteasome: Structure, function, and role in the cell. Cancer Treat Rev, 2003, 29(1): 3–9
Smith D, Benaroudj N, Goldberg A L. Proteasomes and their associated ATPases: A destructive combination. J Structural Biol, 2006, 156: 72–83
Coux O, Tanaka K, Goldberg A L. Structure and functions of the 20S and 26S proteasomes. Annu Rev Biochem, 1996, 65: 801–847
Heinemeyer W, Ramos P C, Dohmen R J. The ultimate nanoscale mincer: Assembly, structure and active sites of the 20S proteasome core. Cell Mol Life Sci, 2004, 61: 1562–1578
Kuttler C, Nussbaum A K, Dick T P, Rammensee H G, Schild H, Hadeler K P. An algorithm for the prediction of proteasomal cleavages. J Mol Biol, 2000, 298(3): 417–429
Nussbaum A K, Kuttler C, Hadeler K P, Rammensee H G, Schild H. PAProC: A prediction algorithm for proteasomal cleavages available on the www. Immunogenetics, 2001, 53(2): 87–94
Holzhutter H G, Frommel C, Kloetzel P M. A theoretical approach towards the identification of cleavage-determining amino acid motifs of the 20S proteasome. J Mol Biol, 1999, 286(4): 1251–1265
Holzhutter H G, Kloetzel P M. A kinetic model of vertebrate 20S proteasome accounting for the generation of major proteolytic fragments from oligomeric peptide substrates. Biophysical J, 2000, 79: 1196–205.
Kesmir C, Nussbaum A K, Schild H, Detours V, Brunak S. Prediction of proteasome cleavage motifs by neural networks. Protein Eng, 2002, 15(4): 287–296
Goldberg A L, Cascio P, Saric T, Rock K L. The importance of the proteasome and subsequent proteolytic steps in the generation of antigenic peptides. Mol Immunol, 2002, 39(3–4): 147–164
Blythe M J, Doytchinova I A, Flower D R. JenPep: A database of quantitative functional peptide data for immunology. Bioinformatics, 2002, 18: 434–439
Altuvia Y, Margalit H. Sequence signals for generation of antigenic peptides by the proteasome: Implications for proteasomal cleavage mechanism. J Mol Biol, 2000, 295: 879–890
Song Z, Liu T, Liu W, Zhu M H, Wang X G. The QSAR model study of interaction between peptides and MHC molecules. Acta Phys Chim Sin (in Chinese), 2007, 23(2): 198–205
Song Z, Liu T, Wang X Y, Liu W. Application of partial least square method in studying of the quantitative structure-activity relationship of T cells epitopes. Immunol J (in Chinese), 2007, 23(2): 166–171
Liu T, Song Z, Liu W, Wang X Y, Qiu X M. Prediction of CTL epitopes based on modified artificial neural network. J Dalian Univ Technol (in Chinese), 2007, 47(4), 473-478
Saxova P, Buus S, Brunak S, Kesmir C. Predicting proteasomal cleavage sites: A comparison of available methods. Int Immunol, 2003, 15(7): 781–787
Bhasin M, Raghava G P. Prediction of CTL epitopes using QM, SVM and ANN techniques. Vaccine, 2004, 22: 3195–3204
Wenzel T, Eckerskorn C, Lottspeich F, Baumeister W. Existence of a molecular ruler in proteasomes suggested by analysis of degradation products. FEBS Lett, 1994, 349: 205–209
Kisselev A F, Akopian T N, Woo K M, Goldberg A L. The sizes of peptides generated from protein by mammalian 26 and 20S proteasomes. Implications for understanding the degradative mechanism and antigen presentation. J Biol Chem, 1999, 274(6): 3363–3371
Dick L R, Moomaw C R, DeMartino G N, Slaughter C A. Degradation of oxidized insulin B chain by the multiproteinase complex macropain (proteasome). Biochemistry, 1991, 30: 2725–2734
Altuvia Y, Margalit H. A structure-based approach for prediction of MHC-binding peptides. Methods, 2004, 34(4): 454–459
Falk K, Rotzschke O, Stevanovic S, Jung G, Rammensee H G. Allele-specific motifs revealed by sequencing of self-peptides eluted from MHC molecules. Nature, 1991, 351: 290–296
Nussbaum A K, Dick T P, Keilholz W, Schirle M, Stevanovic S, Dietzk, Heinemeyer W, Groll M, Wolf D H, Huber R, Rammensee H G, Schild H. Cleavage motifs of the yeast 20S proteasome β subunits deduced from digests of enolase 1. Proc Natl Acad Sci USA, 1998, 95(21): 12504–12509
Unno M, Mizushima Y, Morimoto Y, Tomisugi K, Tanaka N, Yasuoka T. The structure of the mammalian 20S proteasome at 2.75 Å resolution. Structure, 2002, 10: 609–618
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, Z., Liu, T., Jiao, C. et al. Specificity of the proteasome cleavage to the antigen protein. Sci. China Ser. B-Chem. 52, 39–47 (2009). https://doi.org/10.1007/s11426-008-0107-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-008-0107-3