Abstract
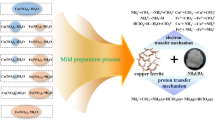
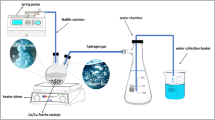
Hydrogen generation through thermal chemical water splitting technology has recently received increasingly international interest in the nuclear hydrogen production field. Besides the main known sulfur-iodine (S-I) cycle developed by the General Atomics Company and the UT3 cycle (iron, calcium, and bromine) developed at the University of Tokyo, the thermal cycle based on metal oxide two-step water splitting methods is also receiving research and development attention worldwide. In this work, copper ferrite was prepared by the co-precipitation method and oxygen-deficient copper ferrite was synthesized through first and second calcination steps for the application of hydrogen production by a two-step water splitting process. The crystal structure, properties, chemical composition and δ were investigated in detail by utilizing X-ray diffraction (XRD), thermogravimetry (TG) and differential thermal analysis (DTA), atomic absorption spectrometer (AAS), ultraviolet spectrophotometry (UV), gas chromatography (GC), and so on. The experimental two-step thermal chemical cycle reactor for hydrogen generation was designed and developed in this lab. The hydrogen generation process of water splitting through CuFe2O4−δ and the cycle performance of copper ferrite regeneration were firstly studied and discussed.
Similar content being viewed by others
References
Project Group of State Statistical Bureau. Positive abate the tight energy supply and active strive for order development. Stat Res (in Chinese), 2005(6): 3–10
Shen B W. Power in the 21century-hydrogen and Hydrogen Energy (in Chinese). Tianjin: Nankai University Press, 2001, 15–58
Li X. Li B. Hydrogen economy. Sol Energy (in Chinese), 2004, (2): 11–12
Funk J E, Reinstrom R M. System study of hydrogen generation by thermal energy. TID 20441, (EDR 3714) (2) (Suppl A), Allison Division of General Motors Corporation, 1964
Funk J E, Reinstrom R M. Energy requirements in the production of hydrogen from water. Ind Eng Chem-Proc Des Dev, 1966, 5(3): 336–342
Funk J E. Thermochemical hydrogen production: Past and present. Int J Hydrogen Energy, 2001, 26(3): 185–190
Nakamura T. Hydrogen production from water utilizing solar heat at high temperatures. Sol Energy, 1977, 19(5): 467–475
Kaneko H. Decomposition of Zn-ferrite for O2 generation by concentrated solar radiation. Sol Energy, 2004, 76(1): 317–322
Tamaura Y, Kojima N. Stoichiometric studies of H2 generation reaction for H2O/Zn/Fe3O4 system. Int J Hydrogen Energy, 2001, 26(9): 917–922
Tamaura Y, Water splitting with the Mn(II)-Ferrite-CaO-H2O system. Energy, 1998, 23(10): 879–886
Hosokawa Y, Kojima N, Yokota O. Multi-step water splitting with Mn-ferrite/sodium carbonate system. In: Proceedings of 34th Intersociety Energy Conversion Engineering Conference. Vancouver: British Columbia (CA), 1999, 2670–2673
Nalbandian L, Zaspalis V T, Evdou A, Agrafiotis C, Konstandopoulos A G. Redox materials for hydrogen production from the water decomposition reaction. Chem Eng Trans, 2004, (4): 43–48
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the specialized research fund for the Doctoral Program of Higher Education, Ministry of Education of China (Grant No. 20070003033)
Rights and permissions
About this article
Cite this article
Yu, B., Zhang, P., Zhang, L. et al. Studies on the preparation of active oxygen-deficient copper ferrite and its application for hydrogen production through thermal chemical water splitting. Sci. China Ser. B-Chem. 51, 878–886 (2008). https://doi.org/10.1007/s11426-008-0059-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-008-0059-7