Abstract
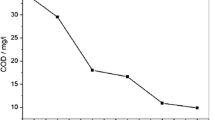
A coupled ultrasound/electrocatalysis (US/EC) process was used to enhance the decomposition efficiency of organics. The synergetic kinetics and the mechanism of 2-chlorophenol (2-CP) decomposition with coupled US/EC were studied. It was found that in a US/EC process 2-CP is attacked by active radicals (such as hydroxyl radicals) to form 2-chloro-p-benzoquinone, and the latter is oxidized to simple organic acids when the ring is opened. The enhancement factor expressed by the apparent rate constant of 2-CP decomposition with coupled US/EC is 1.324 at a current density of 20 mA · cm−2, an ultrasonic frequency of 20 kHz, an ultrasonic intensity of 0.27 W · cm−2, and a 2-CP initial concentration of 200 mg · L−1, which means that a synergetic effect exists. A model derived from Langmuir adsorption theory of solid surface and reaction kinetics equations can describe exactly the decomposition of 2-CP with coupled US/EC. The numerical values are in good agreement with the experimental data. The model parameters are associated with reaction conditions.
Similar content being viewed by others
References
Pi Y Z, Wang J L. The mechanism and pathway of the ozonation of 4-chlorophenol in aqueous solution. Sci China Ser B-Chem, 2006, 49(4): 379–384
Wang J L, Hegemann W. Microbial dehalogenation of trichlorophenol by a bacterial consortium: characterization and mechanism. Sci China Ser B-Chem, 2003, 46(2): 207–215
Comninellis Ch, Nerini A. Anodic oxidation of phenol in the presence of NaCl for wastewater treatment. J Appl Electrochem, 1995, 25(1): 23–28
Rajeshwar K, Ibanex J G, Swain G M. Electrochemistry and the environment. J Appl Electrochem, 1994, 24(11): 1077–1091
Birkin P R, Silva-Martines S. Determination of heterogeneous electron transfer kinetics in the presence of ultrasound at microelectrode employing sampled voltammetry. Anal Chem, 1997, 69(11): 2055–2062
Liu J, Xie Y, Bian S H. Experimental study on sonoelectrochemical treatment of dyestuff wastewater. Shanghai Environ Sci (in Chinese), 2001, 20(3): 151–153
Gao Y, Fu M, Huang J. Study on treatment of aniline solution by electrochemical oxidation process combined with ultrasound. Environ Prot Chem Ind (in Chinese), 2003, 23(6): 318–322
Zhou M H, Dai Q Z, Lei L C, Ma C A, Wang D H. Long life modified lead dioxide anode for organic wastewater treatment: electrochemical characteristics and degradation mechanism. Environ Sci Technol, 2005, 39(1): 363–370
Feng Y J, Li X Y, You H, Ding F. The Application of Electrochemistry Technology in the Environmental Engineering (in Chinese). Beijing: Chem Ind Press, 2002. 132
Sun Y Q, Wang J D, Chen X, Chen J M. Ultrasound assisted electrocatalysis oxidation for enhanced decomposition of 2-chlorophenol in water. J Zhejiang Univ Technol (in Chinese), 2006, 34(5): 495–520
Shang N C, Yu Y H, Ma H W, Chang C H, Liou M L. Toxicity measurements in aqueous solution during ozonation of mono-chlorophenols. J Environ Manage, 2006, 78(3): 216–222
Polcaro A M, Palmas S. Electrochemical oxidation of chlorophenol. Ind Eng Chem Res, 1997, 36(5): 1791–1798
Santos A, Yustos P, Quintanilla A, Rodriguez S, García-Ochoa F. Route of the catalytic oxidation of phenol in aqueous phase. Appl Catal B: Environ, 2002, 39(2): 97–113
Walton D J, Iniesta J, Plattes M, Mason T J, Lorimer J P, Ryley S, Phull S S, Chyla A, Heptinstall J, Thiemann T, Fuji H, Mataka S, Tanaka Y. Sonoelectrochemical effects in electro-organic systems. Ultrason Sonochem, 2003, 10: 209–216
Panizza M, Bocca C, Cerisola G. Electrochemical treatment of wastewater containing polyaromatic organic pollutants. Water Res, 2000, 34: 2601–2605
Song Q, Qu J H. Mechanism and kinetics of 2-chlorophenol degradation in drinking water by photo-electrochemical synergic effect. Sci China Ser B-Chem, 2003, 46(3): 259–270
Li M Y, Xiong L, Chen Y Y, Zhang N, Zhang Y M, Yin H. Studies on photo-electro-chemical catalytic degradation of acid scarlet 3R dye. Sci China Ser B-Chem, 2005, 48(4): 297–304
Ezerskis Z, Jusys Z. Oxidation of chlorophenols on Pt electrode in alkaline solution studied by cyclic voltammetry, galvanostatic electrolysis, and gas chromatography-mass spectrometry. Pure Appl Chem, 2001, 73: 1929–1940
Jin Y X, Yang X P, Liu J J, Zou R Y, Cai D C. Factors affecting treatment of copper ior-containing wastewater by cathodic reduction. Min Metall Eng (in Chinese), 2005, 25(6): 55–56
Liu S H, Wang S L, Sun X Y, Li W Z, Zhu D Z, Liu J Y, Zheng G H, Ni Y M, Wang W F, Wang M, Yao S D. Mechanism study on the oxidizing degradation of 2-chlorophenol aqueous solution. Sci Technol Eng (in Chinese), 2003, 3(2): 129–132
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by Science and Technology Department of Zhejiang Province (Grant No. 2005C23056)
Rights and permissions
About this article
Cite this article
Wang, J., Zhu, R., Chen, X. et al. Mechanism and kinetics of 2-chlorophenol decomposition using coupled ultrasound and electrocatalysis. Sci. China Ser. B-Chem. 51, 577–585 (2008). https://doi.org/10.1007/s11426-007-0137-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-007-0137-2