Abstract
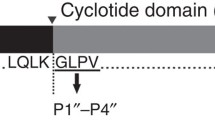
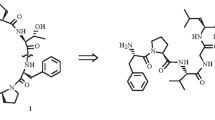
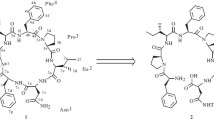
The crude enzyme (PH-1) isolated from Pseudostellaria heterophylla by our group has catalyzed enzymatic cyclization of linear peptide NH2-Gly1-Gly2-Leu-Pro-Pro-Pro-Ile-Phe-COOH (4) into cyclopeptide heterophyllin B (HB) from plant for the first time. To ensure this reaction, some analytical methods including TLC, HPLC, MS, NMR, and 3C labeling were used to prove that the reaction of substrate 4 sharing residue of NH-Phe-Gly-CO was successful.
Similar content being viewed by others
References
Zhou, J., Tan, N. H., Application of a new TLC chemical method for detection of cyclopeptides in plants, Chinese Science Bulletin, 2000, 45(20): 1825–1831.
Kohli, R. M., Walsh, C. T., Burkart, M. D., Biomimetics synthesis and optimization of cyclic peptide antibiotics, Nature, 2002, 418(6898): 658–661.
Kohli, R. M., Burke, M. D., Tao, J. H., Walsh, C. T., Chemoenzymatic route to macrocyclic hybrid peptide/polyketide-like molecules, J. Am. Chem. Soc., 2003, 125(24): 7160–7161.
Jackson, D. Y., Burnier, J. P., Wells, J. A., Enzymatic cyclization of linear peptides esters using subtiligase, J. Am. Chem. Soc., 1995, 117(2): 819–820.
Chinese Academy of Medical Sciences, Zhong Yao Zhi Vol. 2, Beijing: The People’s Health Publishing House, 1995, 266-268.
Tan, N. H., Zhou, J., Chen, C. X., Zhao, S. X., Cyclopeptides from the root of Pseudostellaria heterophylla, Phytochemistry, 1993, 32(5): 1327–1330.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jia, A., Li, X., Tan, N. et al. Enzymatic cyclization of linear peptide to plant cyclopeptide heterophyllin B. SCI CHINA SER B 49, 63–66 (2006). https://doi.org/10.1007/s11426-005-0204-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11426-005-0204-5