Abstract
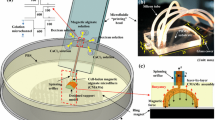
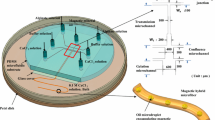
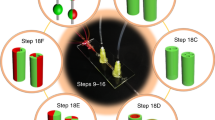
This paper proposes a novel method of magnetically guided assembly to construct multi-layer porous scaffold for three-dimensional cell culture by apply magnetic microfibers. Microfibers are composed of biocompatible and biodegradable alginate solution with homogeneous magnetic nanoparticles, which are continuously spun from a microfluidic device by precise pressure control of the syringe pump. Magnetic nanoparticles enable the control of magnetic field on microfibers. Meanwhile, magnetized device combining with a round permanent magnet are utilized to guide the distribution of spouted microfibers. The device is composed by pure iron wire arrays and wax, which stimulates powerful magnetic flux density and magnetic field gradients for the capture and assembly of microfibers. Thus, magnetic microfibers are spun on desired places of the magnetized device by motion control of the micromanipulation robot, and precise locations are adjusted by magnetic force couple with the assist of glass micropipette. Afterwards, microfibers are spatially organized by periodic magnetic force and crossed layer-by-layer to form micro-pore structure with both length and width of 650 μm. Finally, the authors construct a multilayer microfiber-based scaffold with high porosity to provide a satisfactory environment for long-term cell culture. The experimental results demonstrate the effectiveness of the proposed method.
Similar content being viewed by others
References
Badylak S F, A scaffold immune microenvironment, Science, 2016, 352(6283): 298–298.
Ratner M, Shire punts on bioscaffolds for cell-based regenerative medicine, Nature Biotechnology, 2012, 30(8): 727–728.
Zhang X J, Li Y, Chen Y E, et al., Cell-free 3D scaffold with two-stage delivery of miRNA-26a to regenerate critical-sized bone defects, Nature Communications, 2016, 7: 10376–10390.
Hollister S J, Porous scaffold design for tissue engineering, Nature Materials, 2005, 4(7): 518–524.
Griffith L G and Naughton G, Tissue engineering-current challenges and expanding opportunities, Science, 2002, 295: 1009–1014.
Matsunaga Y T, Morimoto Y, and Takeuchi S, Molding cell beads for rapid construction of macroscopic 3D tissue architecture, Advanced Materials, 2011, 23: H90–H94.
Tamayol A, Akbari M, Annabi N, et al., Fiber-based tissue engineering: Progress, challenges and opportunities, Biotechnology Advances, 2013, 31(5): 669–687.
Yue T, Nakajima M, Takeuchi M, et al., On-chip self-assembly of cell embedded microstructures to vascular-like microtubes, Lab on a Chip, 2014, 14(6): 1151–1161.
Murphy S V and Anthony A, 3D bioprinting of tissue and organs, Nature Biotechnology, 2014, 32: 773–785.
Durmus N G, Tasoglu S, and Demirci U, Bioprinting: Functional droplet networks, Nature Materials, 2013, 12(6): 478–479.
Kang H W, Sang J L, Ko I K, et al., A 3D bioprinting system to produce human-scale tissue constructs with structural integrity, Nature Biotechnology, 2016, 34(3): 312–319.
Lai K L and Xu T, Bioprinting of cartilage: Recent progress on bioprinting of cartilage, Nature, 2015, 88(20): 241–242.
Pawar A A, Saada G, Cooperstein I, et al., High-performance 3D printing of hydrogels by waterdispersible photoinitiator nanoparticles, Science Advances, 2016, 2(4): 1501381–1501387.
Kolesky D B, Truby R L, Gladman A S, et al., 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs, Advanced Materials, 2014, 26(19): 3124–3130.
Zhao Y, Yao R, Ouyang L, et al., Three-dimensional printing of hela cells for cervical tumor model in vitro, Biofabrication, 2014, 6(3): 035001.
Xu C X, Chai W X, Huang Y, et al., Scaffold-free inkjet printing of three-dimensional zigzag cellular tubes, NBiotechnology and Bioengineering, 2012, 109: 3152–3160.
Duan B, Hockaday L A, Kang K H, et al., 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels, Journal of Biomedical Materials Research Part A, 2013, 101A: 1255–1264.
Wang H P, Huang Q, Shi Q, et al., Automated assembly of vascular-like microtube with repetitive single-step contact manipulation, IEEE Transactions on Biomedical Engineering, 2015, 62(11): 2620–2628.
Wang H P, Shi Q, Guo Y N, et al., Contact assembly of cell-laden hollow microtubes through automated micromanipulator tip locating, Journal of Micromechanics and Microengineering, 2017, 27(1): 1–13.
Zhang Y, Chen B K, Liu X Y, et al., Autonomous robotic pick-and-place of microobjects, IEEE Transactions on Robotics, 2010, 26: 200–207.
Mol A, Van Lieshout M I, Dam-de Veen C G, et al., Fibrin as a cell carrier in cardiovascular tissue engineering applications, Biomaterials, 2005, 26(16): 3113–3121.
Yu Y, Wen H, Ma J, et al., Flexible fabrication of biomimetic bamboo-like hybrid microfibers, Advanced Materials, 2014, 26(16): 2494–2499.
Onoe H, Okitsu T, Itou A, et al., Metre-long cell-laden microfibers exhibit tissue morphologies and functions, Nature materials, 2013, 12: 584–590.
Yamada M, Utoh R, Ohashi K, et al., Controlled formation of heterotypic hepatic micro-organoids in anisotropic hydrogel microfibers for long-term preservation of liver-specific functions, Biomaterials, 2012, 33(33): 8304–8315.
Sun T, Hu C Z, Nakajima M, et al., On-chip fabrication and magnetic force estimation of peapodlike hybrid microfibers using a microfluidic device, Microfluidics and Nanofluidics, 2014, 18(5–6): 1177–1187.
Lee H W, Kim K C, and Lee J, Review of maglev train technologies, IEEE Transactions on Magnetics, 2006, 42(7): 1917–1925.
Goya G F, Calatayud M P, Sanz B, et al., Magnetic nanoparticles for magnetically guided therapies against neural diseases, Mrs Bulletin, 2014, 39(11): 965–969.
Tasoglu S, Diller E, Guven S, et al., Untethered micro-robotic coding of three-dimensional materials composition, Nature Communications, 2014, 5(1): 3124.
Mejías R, Pérez-Yagüe S, Gutiérrez L, et al., Dimercaptosuccinic acid-coated magnetite nanoparticles for magnetically guided in vivo delivery of interferon gamma for cancer immunotherapy, Biomaterials, 2011, 32(11): 2938–2952.
Fuchigami T, Kawamura R, Kitamoto Y, et al., A magnetically guided anti-cancer drug delivery system using porous FePt capsules, Biomaterials, 2012, 33(5): 1682–1687.
Arcese L, Fruchard M, and Ferreira A, Endovascular magnetically guided robots: Navigation modeling and optimization, IEEE Transactions on Biomedical Engineering, 2012, 59(4): 977–987.
Alsberg E, Feinstein E, Joy M P, et al., Magnetically-guided self-assembly of fibrin matrices with ordered nano-scale structure for tissue engineering, Tissue Engineering, 2006, 12(11): 3247–3256.
Hu C Z, Uchida T, Tercero C, et al., Development of biodegradable scaffolds based on magnetically guided assembly of magnetic sugar particles, Journal of Biotechnology, 2012, 159(1–2): 90–98.
He X H, Wang W, Liu Y M, et al., Microfluidic fabrication of bio-Inspired microfibers with controllable magnetic spindle-knots for 3D assembly and water collection, ACS Applied Materials & Interfaces, 2015, 7(31): 17471–17481.
Author information
Authors and Affiliations
Corresponding author
Additional information
This research was supported by the Beijing Natural Science Foundation under Grant No. 4164099, and the National Nature Science Foundation of China under Grant Nos. 61375108, 61520106011, and 61603044.
This paper was recommended for publication by Editor SUN Jian.
Rights and permissions
About this article
Cite this article
Li, X., Wang, H., Shi, Q. et al. Construction of Multilayer Porous Scaffold Based on Magnetically Guided Assembly of Microfiber. J Syst Sci Complex 31, 581–595 (2018). https://doi.org/10.1007/s11424-017-6164-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11424-017-6164-y