Abstract
Purpose
Interpretation of drug levels in decomposed and buried corpses is difficult. We aimed to determine postmortem distribution of drugs in putrefied-decomposed visceral tissues and buried bones, and the effect of decomposition and burial on distribution of drugs in organs and bone tissues by administering drug groups selected from various drug classes to domestic pigs.
Methods
Pigs were divided into groups (n = 5) and dosed with a variety of drugs. Peripheral blood, organ and bone samples in different anatomical locations were collected from pigs killed. Organ samples were collected at postmortem 4th, 24th, 48th, 72nd and 96th hour. Then the corpses were buried below soil ground. Bone samples from buried corpses were collected at 5th and 10th months of the burial time by exhumation. All samples were analysed using liquid chromatography-tandem mass spectrometry after making sample preparation using appropriate methods.
Results
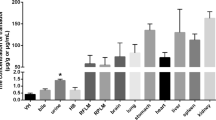
Only 10 of the 14 drugs were detected in the initial peripheral blood. For all bone types analyzed, highest drug levels were detected at thorax region and lowest drug levels were detected at the lower extremity. Given the fact that most of the soft tissue is located in the thorax region, it is possible that during the decomposition process, drugs partitioned from the liquefied tissue into bones. That the drug levels in bones decreased was observed when exhumation was done at 10th month of the burial time.
Conclusions
Drug distribution in organ and bone of decomposed and burial corpses vary due to unknown mechanisms and completely unexplained conditions such as postmortem redistribution.




Similar content being viewed by others
References
Levine B (2003) Principles of forensic toxicology, 2nd edn. AACC Press, Washington
Dinis-Oliveira RJ, Carvalho F, Duarte JA et al (2010) Collection of biological samples in forensic toxicology. Toxicol Mech Methods 20:363–414
Skopp G (2004) Preanalytic aspects in postmortem toxicology. Forensic Sci Int 142:75–100. https://doi.org/10.1016/j.forsciint.2004.02.012
Dent BB, Forbes SL, Stuart BH (2004) Review of human decomposition processes in soil. Environ Geol 45:576–585. https://doi.org/10.1007/s00254-003-0913-z
Käferstein H, Sticht G, Madea B (2013) Chlorprothixene in bodies after exhumation. Forensic Sci Int 229:e30–34.https://doi.org/10.1016/j.forsciint.2013.03.051
Wyman JF, Dean DE, Yinger R et al (2011) The temporal fate of drugs in decomposing porcine tissue. J Forensic Sci 56:694–699. https://doi.org/10.1111/j.1556-4029.2011.01725.x
Watterson JH, Desrosiers NA, Betit CC et al (2010) Relative distribution of drugs in decomposed skeletal tissue. J Anal Toxicol 34:510–515. https://doi.org/10.1093/jat/34.8.510
Drummer OH (2008) Drugs in bone and bone marrow. In: Jenkins AJ (ed) Drug testing in alternate biological specimens. Humana Press, Totowa, pp 131–137
Stepensky D, Kleinberg L, Hoffman A (2003) Bone as an effect compartment. Clin Pharmacokinet 42:863–881. https://doi.org/10.2165/00003088-200342100-00001
McIntyre IM, King C, Boratto M, Drummer OH (2000) Postmortem drug analyses in bone and bone marrow. Ther Drug Monit 22:79–83. https://doi.org/10.1097/00007691-200002000-00017
Watterson JH, Donohue JP (2011) Relative distribution of ketamine and norketamine in skeletal tissues following various periods of decomposition. J Anal Toxicol 35:452–458. https://doi.org/10.1093/anatox/35.7.452
Wiebe TR, Watterson JH (2014) Analysis of tramadol and O-desmethyltramadol in decomposed skeletal tissues following acute and repeated tramadol exposure by gas chromatography mass spectrometry. Forensic Sci Int 242:261–265. https://doi.org/10.1016/j.forsciint.2014.07.010
Fraser CD, Cornthwaite HM, Watterson JH (2015) Analysis of dextromethorphan and dextrorphan in decomposed skeletal tissues by microwave assisted extraction, microplate solid-phase extraction and gas chromatography-mass spectrometry (MAE-MPSPE-GCMS). Drug Test Anal 7:708–713. https://doi.org/10.1002/dta.1754
Imfeld AB, Watterson JH (2016) Influence of dose-death interval on colchicine and metabolite distribution in decomposed skeletal tissues. Int J Legal Med 130:371–379. https://doi.org/10.1007/s00414-015-1196-0
Mella M, Schweitzer B, Mallet CR et al (2017) Detection of cocaine and metabolites in bone following decomposition using 2D LC–MS-MS. J Anal Toxicol. https://doi.org/10.1093/jat/bkx106
McIntyre IM, Mallett P (2012) Sertraline concentrations and postmortem redistribution. Forensic Sci Int 223:349–352. https://doi.org/10.1016/j.forsciint.2012.10.020
Drummer OH (2004) Postmortem toxicology of drugs of abuse. Forensic Sci Int 142:101–113. https://doi.org/10.1016/j.forsciint.2004.02.013
Lafreniere NM, Watterson JH (2009) Detection of acute fentanyl exposure in fresh and decomposed skeletal tissues. Forensic Sci Int 185:100–106. https://doi.org/10.1016/j.forsciint.2008.12.019
Watterson JH, VandenBoer TC (2008) Effects of tissue type and the dose-death interval on the detection of acute ketamine exposure in bone and marrow with solid-phase extraction and ELISA with liquid chromatography-tandem mass spectrometry confirmation. J Anal Toxicol 32:631–638. https://doi.org/10.1093/jat/32.8.631
Desrosiers NA, Watterson JH, Dean D, Wyman JF (2012) Detection of amitriptyline, citalopram, and metabolites in porcine bones following extended outdoor decomposition. J Forensic Sci 57:544–549. https://doi.org/10.1111/j.1556-4029.2011.01994.x
Scientific Working Group for Forensic Toxicology (2013) Scientific Working Group for Forensic Toxicology (SWGTOX) standard practices for method validation in forensic toxicology. J Anal Toxicol 37:452–474. https://doi.org/10.1093/jat/bkt054
Flanagan RJ, Amin A, Seinen W (2003) Effect of postmortem changes on peripheral and central whole blood and tissue clozapine and norclozapine concentrations in the domestic pig (Sus scrofa). Forensic Sci Int 132:9–17. https://doi.org/10.1016/S0379-0738(02)00414-0
Nilsson GH, Kugelberg FC, Kronstrand R, Ahlner J (2010) Stability tests of zopiclone in whole blood. Forensic Sci Int 200:130–135. https://doi.org/10.1016/j.forsciint.2010.04.001
Shiota H, Nakashima M, Terazono H et al (2004) Postmortem changes in tissue concentrations of triazolam and diazepam in rats. Leg Med 6:224–232. https://doi.org/10.1016/j.legalmed.2004.05.006
Cattaneo C, Gigli F, Lodi F, Grandi M (2003) The detection of morphine and codeine in human teeth: an aid in the identification and study of human skeletal remains. J Forensic Odontostomatol 21:1–5
McGrath KK, Jenkins AJ (2009) Detection of drugs of forensic importance in postmortem bone. Am J Forensic Med Pathol 30:40–44. https://doi.org/10.1097/PAF.0b013e31818738c9
Watterson JH, Desrosiers NA (2011) Examination of the effect of dose-death interval on detection of meperidine exposure in decomposed skeletal tissues using microwave-assisted extraction. Forensic Sci Int 207:40–45. https://doi.org/10.1016/j.forsciint.2010.08.021
Watterson JH, Cornthwaite HM (2013) Discrimination between patterns of drug exposure by toxicological analysis of decomposed skeletal tissues. Part II: amitriptyline and citalopram. J Anal Toxicol 37:565–572. https://doi.org/10.1093/jat/bkt078
Delabarde T, Keyser C, Tracqui A et al (2013) The potential of forensic analysis on human bones found in riverine environment. Forensic Sci Int 228:e1–5. https://doi.org/10.1016/j.forsciint.2013.03.019
Vardakou I, Athanaselis S, Pistos C et al (2014) The clavicle bone as an alternative matrix in forensic toxicological analysis. J Forensic Leg Med 22:7–9. https://doi.org/10.1016/j.jflm.2013.11.012
Cornthwaite HM, Watterson JH (2014) The influence of body position and microclimate on ketamine and metabolite distribution in decomposed skeletal remains. J Anal Toxicol 38:548–554. https://doi.org/10.1093/jat/bku053
Moffaf A, Osselton M, Widdop B (2011) Clarke’s analysis of drugs and poisons, 4th edn. Pharmaceutical Press, London
Baselt RC (2009) Disposition of toxic drugs and chemicals in man, 8th edn. Biomedical Publications, Foster City
Molina DK (2010) Handbook of forensic toxicology for medical examiners. CRC Press, Boca Raton, Florida
Funding
This work was supported by Research Fund of the Cukurova University (Project Number: TF2013YL14).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that there is not conflict of interest for the article: “Determination of Drug Distributions in Decomposed and Buried Postmortem Tissues and Bones of Pigs Administered Drugs”.
Ethical approval
The experiment was conducted at the Experimental Research and Application Center of Medical Sciences at Cukurova University, Adana, Turkey. Çukurova University Animal Experiments Local Ethics Committee (AELEC) in Adana, Turkey approved the study. All applicable international, national, and/or institutional guidelines were followed for the use of the animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Goren, I.E., Gokce Daglioglu, N., Daglioglu, Y.K. et al. Determination of drug distributions in decomposed and buried postmortem tissues and bones of pigs administered of drugs. Forensic Toxicol 38, 129–140 (2020). https://doi.org/10.1007/s11419-019-00497-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11419-019-00497-8