Abstract
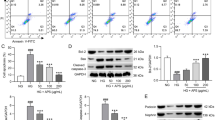
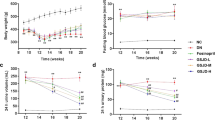
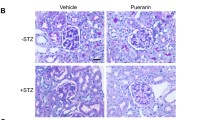
Diabetic nephropathy (DN) is one of the major microvascular complications in diabetes. Podocyte injury such as slit diaphragm effacement is regarded as a determinant in the occurrence and development of albuminuria in DN. In this study, we examined the effect of hyperoside, an active flavonoid glycoside, on proteinuria and renal damage in a streptozotocin-induced DN mouse model at the early stage. The results showed that oral administration of hyperoside (30 mg/kg/day for 4 weeks could significantly decrease urinary microalbumin excretion and glomerular hyperfiltration in DN mice, but did not affect the glucose and lipid metabolism. Periodic acid−Schiff staining and transmission electron microscopy showed that glomerular mesangial matrix expansion and podocyte process effacement in DN mice were significantly improved by hyperoside. Further investigations via immunofluorescence staining, real-time reverse transcription polymerase chain reaction and Western blot analysis showed that the decreased slit diaphragm protein nephrin and podocin mRNA expression and protein levels in DN mice were restored by hyperoside treatment. Collectively, these findings demonstrated that hyperoside could decrease albuminuria at the early stage of DN by ameliorating renal damage and podocyte injury.




Similar content being viewed by others
References
Gross JL, de Azevedo MJ, Silveiro SP et al (2005) Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care 28:164–176
Wada T, Shimizu M, Toyama T et al (2012) Clinical impact of albuminuria in diabetic nephropathy. Clin Exp Nephrol 16:96–101
Klausen K, Borch-Johnsen K, Feldt-Rasmussen B et al (2004) Very low levels of microalbuminuria are associated with increased risk of coronary heart disease and death independently of renal function, hypertension, and diabetes. Circulation 110:32–35
Coca SG, Ismail-Beigi F, Haq N et al (2012) Role of intensive glucose control in development of renal end points in type 2 diabetes mellitus: systematic review and meta-analysis intensive glucose control in type 2 diabetes. Arch Intern Med 172:761–769
Vejakama P, Thakkinstian A, Lertrattananon D et al (2012) Reno-protective effects of renin-angiotensin system blockade in type 2 diabetic patients: a systematic review and network meta-analysis. Diabetologia 55:566–578
Wolf G, Chen S, Ziyadeh FN (2005) From the periphery of the glomerular capillary wall toward the center of disease: podocyte injury comes of age in diabetic nephropathy. Diabetes 54:1626–1634
Cao Z, Cooper ME (2011) Pathogenesis of diabetic nephropathy. J Diabetes Investig 2:243–247
Tryggvason K, Wartiovaara J (2001) Molecular basis of glomerular permselectivity. Curr Opin Nephrol Hypertens 10:543–549
Liu G, Kaw B, Kurfis J et al (2003) Neph1 and nephrin interaction in the slit diaphragm is an important determinant of glomerular permeability. J Clin Investig 112:209–221
Zhang A, Huang S (2012) Progress in pathogenesis of proteinuria. Int J Nephrol 2012:314251. doi:10.1155/2012/314251
Toyoda M, Suzuki D, Umezono T et al (2004) Expression of human nephrin mRNA in diabetic nephropathy. Nephrol Dial Transplant 19:380–385
Doublier S, Salvidio G, Lupia E et al (2003) Nephrin expression is reduced in human diabetic nephropathy: evidence for a distinct role for glycated albumin and angiotensin II. Diabetes 52:1023–1030
Eto N, Wada T, Inagi R et al (2007) Podocyte protection by darbepoetin: preservation of the cytoskeleton and nephrin expression. Kidney Int 72:455–463
Li ZL, Hu J, Li YL et al (2013) The effect of hyperoside on the functional recovery of the ischemic/reperfused isolated rat heart: potential involvement of the extracellular signal-regulated kinase 1/2 signaling pathway. Free Radic Biol Med 57:132–140
Zeng KW, Wang XM, Ko H et al (2011) Hyperoside protects primary rat cortical neurons from neurotoxicity induced by amyloid beta-protein via the PI3 K/Akt/Bad/Bcl(XL)-regulated mitochondrial apoptotic pathway. Eur J Pharmacol 672:45–55
Li ZL, Liu JC, Hu J et al (2012) Protective effects of hyperoside against human umbilical vein endothelial cell damage induced by hydrogen peroxide. J Ethnopharmacol 139:388–394
Wu LL, Yang XB, Huang ZM et al (2007) In vivo and in vitro antiviral activity of hyperoside extracted from Abelmoschus manihot (L) medik. Acta Pharmacol Sin 28:404–409
Ku SK, Zhou W, Lee W, et al. (2014) Anti-Inflammatory effects of hyperoside in human endothelial cells and in mice. Inflammation 38:784–799
Yan Y, Feng Y, Li W et al (2014) Protective effects of quercetin and hyperoside on renal fibrosis in rats with unilateral ureteral obstruction. Exp Ther Med 8:727–730
Zhang Z, Sethiel MS, Shen W et al (2013) Hyperoside downregulates the receptor for advanced glycation end products (RAGE) and promotes proliferation in ECV304 cells via the c-Jun N-terminal kinases (JNK) pathway following stimulation by advanced glycation end-products in vitro. Int J Mol Sci 14:22697–22707
Ku SK, Kwak S, Kwon OJ et al (2014) Hyperoside inhibits high-glucose-induced vascular inflammation in vitro and in vivo. Inflammation 37:1389–1400
Brosius FC 3rd, Alpers CE, Bottinger EP et al (2009) Mouse models of diabetic nephropathy. J Am Soc Nephrol 20:2503–2512
An XF, Zhao Y, Yu JY et al (2010) Plasma sRAGE is independently associated with high sensitivity C-reactive protein in type 2 diabetes without coronary artery disease. Diabetes Res Clin Pract 87:e19–e22
Alsaad KO, Herzenberg AM (2007) Distinguishing diabetic nephropathy from other causes of glomerulosclerosis: an update. J Clin Pathol 60:18–26
Sugimoto H, Grahovac G, Zeisberg M et al (2007) Renal fibrosis and glomerulosclerosis in a new mouse model of diabetic nephropathy and its regression by bone morphogenic protein-7 and advanced glycation end product inhibitors. Diabetes 56:1825–1833
Jefferson JA, Shankland SJ, Pichler RH (2008) Proteinuria in diabetic kidney disease: a mechanistic viewpoint. Kidney Int 74:22–36
Patari-Sampo A, Ihalmo P, Holthofer H (2006) Molecular basis of the glomerular filtration: nephrin and the emerging protein complex at the podocyte slit diaphragm. Ann Med 38:483–492
Lenkkeri U, Mannikko M, McCready P et al (1999) Structure of the gene for congenital nephrotic syndrome of the finnish type (NPHS1) and characterization of mutations. Am J Hum Genet 64:51–61
Aaltonen P, Luimula P, Astrom E et al (2001) Changes in the expression of nephrin gene and protein in experimental diabetic nephropathy. Lab Investig 81:1185–1190
Coward RJ, Welsh GI, Koziell A et al (2007) Nephrin is critical for the action of insulin on human glomerular podocytes. Diabetes 56:1127–1135
Xue C, Guo J, Qian D et al (2011) Identification of the potential active components of Abelmoschus manihot in rat blood and kidney tissue by microdialysis combined with ultra-performance liquid chromatography/quadrupole time-of-flight mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 879:317–325
Acknowledgments
This study is supported by Shandong Province Natural Science Foundation (ZR2014CM040) and Jiangsu Province Natural Science Foundation (BK20141503).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
All the authors have no potential conflicts of interest to declare.
Additional information
J. Zhang and H. Fu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, J., Fu, H., Xu, Y. et al. Hyperoside reduces albuminuria in diabetic nephropathy at the early stage through ameliorating renal damage and podocyte injury. J Nat Med 70, 740–748 (2016). https://doi.org/10.1007/s11418-016-1007-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-016-1007-z