Abstract
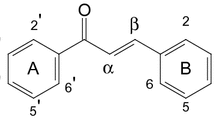
Two new caffeoyl quinic acid α-glucosides, together with three known caffeoyl quinic acids and five known flavonoid glucosides, were isolated from the leaves of Moringa oleifera Lam. The structures of the new compounds were elucidated as 4-O-(4′-O-α-d-glucopyranosyl)-caffeoyl quinic acid (1) and 4-O-(3′-O-α-d-glucopyranosyl)-caffeoyl quinic acid (2) by spectroscopic analyses.

Similar content being viewed by others
References
Sastri BN (1962) The wealth of India. Council of Scientific and Industrial Research, New Delhi
Basu BD, Kirtikar KR (1980) Indian medicinal plants, vol. 1. International Book Distributors, Dehradun, pp 676–83
Vaidyaratnam PSV (1994) Indian medicinal plants: a compendium of 500 species, vol. 4. Orient Longman, Madras, pp 59–64
Anwar F, Bhanger MI (2003) Analytical characterization of Moringa oleifera seed oil grown in temperate regions of Pakistan. J Agric Food Chem 51:6558–6563
Anwar F, Ashraf M, Bhanger MI (2005) Interprovenance variation in the composition of Moringa oleifera oilseeds from Pakistan. J Am Oil Chem Soc 82:45–51
Eilert U, Wolters B, Nahrstedt A (1981) Moringa oleifera and Moringa stenopetala. Planta Med 42:55–61
Faizi S, Siddiqui BS, Saleem R, Siddiqui S, Aftab K, Gilani AH (1994) Novel hypotensive agents, niazimin A, niazimin B, niazicin A and niazicin B from Moringa oleifera: isolation of first naturally occurring carbamates. J Chem Soc Perkin Trans 1:3035–3040
Faizi S, Siddiqui BS, Saleem R, Aftab K, Shaheen F, Gilani AH (1998) Hypotensive constituents from the pods of Moringa oleifera. Planta Med 64:225–228
Guevara AP, Vargas C, Sakurai H, Fujiwara Y, Hashimoto K, Maoka T, Kozuka M, Ito Y, Tokuda H, Nishino H (1999) An antitumor promoter from Moringa oleifera Lam. Mutat Res Fundam Mol Mech Mutagen 440:181–188
Murakami A, Kitazono Y, Jiwajinda S, Koshimizu K, Ohigashi H (1998) Niaziminin, a thiocarbamate from the leaves of Moringa oleifera, holds a strict structural requirement for inhibition of tumor promoter-induced Epstein-Barr virus activation. Planta Med 64:319–323
Cheenpracha S, Park EJ, Yoshida WY, Barit C, Wall M, Pezzuto JM, Chang LC (2010) Potential anti-inflammatory phenolic glycosides from the medicinal plant Moringa oleifera fruits. Bioorg Med Chem 18:6598–6602
Morishita H, Iwahashi H, Osaka N, Kido R (1984) Chromatographic separation and identification of naturally occurring chlorogenic acids by 1H nuclear magnetic resonance spectroscopy and mass spectrometry. J Chromatogr A 315:253–260
Jaramillo K, Dawid C, Hofmann T, Fujimoto Y, Osorio C (2011) Identification of antioxidative flavonols and anthocyanins in Sicana odorifera fruit peel. J Agric Food Chem 59:975–983
Burkard W, Victor W, Rudolf G, Karl H (1989) Malonated flavonol glycosides and 3,5-dicaffeoylquinic acid from pears. Phytochemistry 28:663–664
Liu X, Ye W, Yu B, Zhao S, Wu H, Che C (2004) Two new flavonol glycosides from Gymnema sylvestre and Euphorbia ebracteolata. Carbohydr Res 339:891–895
Wagner GK, Pesnot T (2010) Glycosyltransferases and their assays. ChemBioChem 11:1939–1949
Christiane Goedl C, Nidetzky B (2009) Sucrose phosphorylase harbouring a redesigned, glycosyltransferase-like active site exhibits retaining glucosyl transfer in the absence of a covalent intermediate. ChemBioChem 10:2333–2337
Hu Y, Walker S (2002) Remarkable structural similarities between diverse glycosyltransferases. Chem Biol 9: 1287–1296
Streb S, Egli B, Eicke S, Zeeman CC (2009) The debate on the pathway of starch synthesis: a closer look at low-starch mutants lacking plastidial phosphoglucomutase supports the chloroplast-localized pathway. Plant Physiol 151:1769–1772
Ullah N, Ahmed S, Muhammad P, Ahmed Z, Nawaz HR, Malik A (1999) Coumarinolignoid glycoside from Daphne oleoides. Phytochemistry 51:103–105
Shi SY, Zhou Q, Peng H, Zhou CX, Hu MH, Tao QF, Hao XJ, Stöckigt J, Zhao Y (2007) Four new constituents from Taraxacum mongolicum. Chin Chem Lett 18:1367–1370
Cuong NX, Nhiem NX, Thao NP, Nama NH, Dat NT, Anh HLT, Huong LM, Kiem PV, Minh CV, Wonc JH, Chung WY, Kim YH (2010) Inhibitors of osteoclastogenesis from Lawsonia inermis leaves. Bioorg Med Chem Lett 20:4782–4784
She G, Guo Z, Lv H, She D (2009) New flavonoid glycosides from Elsholtzia rugulosa Hemsl. Molecules 14:4190–4196
Myers RW, Lee RT, Lee YC, Thomas GH, Reynolds LW, Uchida Y (1980) The synthesis of 4-methylumbelliferyl α-ketoside of N-acetylneuraminic acid and its use in a fluorometric assay for neuraminidase. Anal Biochem 101:166–174
Reed L, Muench H (1938) A simple method of estimating fifty percent endpoints. Am J Hygiene 27:493–497
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kashiwada, Y., Ahmed, F.A., Kurimoto, Si. et al. New α-glucosides of caffeoyl quinic acid from the leaves of Moringa oleifera Lam.. J Nat Med 66, 217–221 (2012). https://doi.org/10.1007/s11418-011-0563-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-011-0563-5