Abstract
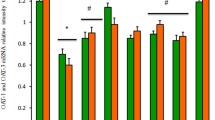
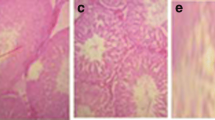
Recently, the nephroprotective property of Pueraria tuberosa DC. tuber (PT) has been reported by our group. Here, PT-embedded biscuits were prepared and tested on cisplatin-induced nephrotoxicity in Swiss albino mice. The PT powder was characterized by RAPD (random amplified polymorphic DNA) to ascertain its authenticity and PT biscuits were prepared in different concentrations (1, 2, or 4 g of PT powder). These biscuits were given as diet for a total of 10 days, but on the 7th day cisplatin injection (8 mg/kg bw, i.p.) was given. On the 10th day animals were killed to collect kidneys for assessment of antioxidant status. Blood samples were collected on both the 7th and 10th days for assessment of liver and kidney functions. In mice, PT biscuit showed significant protection against cisplatin-induced nephrotoxicity, but there was a transient rise in alanine aminotransferase and aspartate aminotransferase at the dose of 4 g PT biscuit. Therefore, it is suggested that PT biscuit might be an effective food supplement for cancer patients undergoing cisplatin-chemotherapy. However, periodical liver function monitoring is required, especially when PT is used for longer periods or at higher doses.

Similar content being viewed by others
References
Kawai Y, Nakao T, Kunimura N, Kohda Y, Gemba M (2006) Relationship of intracellular calcium and oxygen radicals to cisplatin-related renal cell injury. J Pharmacol Sci 100:65–72
Pabla N, Dong Z (2008) Cisplatin nephrotoxicity: mechanisms and renoprotective strategies. Kidney Int 73:994–1007
Sugiyama S, Hayakawa M, Kato T, Hanaki Y, Shimizu K, Ozawa T (1989) Adverse effects of anti-tumor drug, cisplatin, on rat kidney mitochondria: disturbances in glutathione peroxidase activity. Biochem Biophys Res Commun 159:1121–1127
Matsushima H, Yonemura K, Ohishi K, Hishida A (1998) The role of oxygen free radicals in cisplatin-induced acute renal failure in rats. J Lab Clin Med 131:518–526
Chirino YI, Trujillo J, Sanchez-Gonzalez DJ, Martinez-Martinez CM, Cruz C, Bobadilla NA, Pedraza-Chaverri J (2008) Selective iNOS inhibition reduces renal damage induced by cisplatin. Toxicol Lett 176:48–57
Faubel S, Lewis EC, Reznikov L, Ljubanovic D, Hoke TS, Somerset H, Oh DJ, Lu L, Klein CL, Dinarello CA, Edelstein CL (2007) Cisplatin-induced acute renal failure is associated with an increase in the cytokines interleukin (IL)-1beta, IL-18, IL-6, and neutrophil infiltration in the kidney. J Pharmacol Exp Ther 322:8–15
Lau AH (1999) Apoptosis induced by cisplatin nephrotoxic injury. Kidney Int 56:1295–1298
Nagwani S, Tripathi YB (2010) Amelioration of cisplatin induced nephrotoxicity by PTY: a herbal preparation. Food Chem Toxicol 48:2253–2258
Tripathi YB, Tripathi P, Arjmandi BH (2005) Nutraceuticals and cancer management. Frontiers Biosci 10:1607–1618
Pandey GS, Chunekar KC (1998) Vidari Kand, Nighantu BP (eds) Chaukambha Vidya Bhavan, Varanasi, vol 1:388–389
Tanwar YS, Goyal S, Ramawat KG (2008) Hypolipidemic effects of tubers of Indian Kudzu (Pueraria tuberosa). J Herbal Med Toxicol 2(1):21–25
Gupta RS, Sharma R, Sharma A (2004) Antifertility effects of Pueraria tuberosa root extract in male rats. Pharm Biol 42(8):603–609
Hsu LF, Liu MI, Kuo HD, Chen CW, Su CH, Cheng TJ (2003) Antihyperglycemic effect of Puerarin in streptozotocin-induced rats. J Nat Prod 66:788–792
Shukla S, Jonathan S, Sharma A (1996) Protective action of butanolic extract of Pueraria tuberosa DC against carbon tetrachloride induced hepatotoxicity in adult rats. Phytother Res 10(7):608–609
Pandey N, Chaurasia JK, Tiwari OP, Tripathi YB (2007) Antioxidant properties of different fractions of tubers from Pueraria tuberosa. Food Chem 105:219–222
Pandey N, Tripathi YB (2010) Antioxidant activity of tuberosin isolated from Pueraria tuberosa Linn. J Inflamm 7:47
Nagwani S, Kumar M, Singh R, Tripathi YB (2010) Hepatotoxicity of tubers of Indian Kudzu (Pueraria tuberosa) in rats. Food Chem Toxicol 48:1066–1071
Khanuja PSS, Shasany AK, Darokar MP, Kumar S (1999) Rapid isolation of DNA from dry and fresh samples of plants producing large amounts of secondary metabolites and essential oils. Plant Mol Bio Rep 17:1–7
Devaiah KM, Venkatasubramanian P (2008) Development of SCAR marker for authentication of Pueraria tuberose (Roxb. ex. Willd.) DC. Curr Sci 94(10):1306–1309
Waynforth PLHB (1980) Experimental and surgical technique in the rat. Academic Press, London
Schumann G, Bonora R, Ceriotti F, Férard G, Ferrero CA, Franck PFH, Gella FJ, Hoelzel W, Jørgensen PJ, Kanno T, Kessner A, Klauke R, Kristiansen N, Lessinger JM, Linsinger TPJ, Misaki H, Panteghini M, Pauwels J, Schiele F, Schimmel HG (2002) 725 IFCC primary reference procedures for the measurement of catalytic activity concentrations of enzymes at 37°C, part 5. Clin Chem Lab Med 40:725–733
Garber CC (1981) Jendrassik–Grof analysis for total and direct bilirubin in serum with a centrifugal analyzer. Clin Chem 27:1410–1416
Fawcett JK, Scott JE (1960) A rapid and precise method for the determination of urea. J Clin Pathol 13:156–159
Larsen K (1972) Creatinine assay by a reaction-kinetic principle. Clin Chim Acta 41:209–217
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxide in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Tripathi YB, Sharma M (1998) Comparison of antioxidant action of alcoholic extract of Rubia cordifolia with rubiadin. Indian J Biochem Biophys 35:313–316
McCord JM, Fridovich I (1969) Superoxide dismutase: an enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055
Tripathi YB, Singh VP (1996) Role of Tamra bhasma, an Ayurvedic preparation, in the management of lipid peroxidation in liver of albino rats. Indian J Exp Biol 34:66–70
Boyne AF, Ellman GL (1972) A methodology for analysis of tissue sulfhydryl components. Anal Biochem 46(2):639–653
Tripathi YB, Shukla S, Sharma M, Shukla VK (1995) Antioxidant property of Rubia cordifolia extract and its comparison with Vitamin E and Parabenzoquinone. Phytother Res 9:440–443
Lowry OH, Rosebrough AL, Earr KJ, Randall KJ (1951) Protein measurements with the folin phenol reagent. J Biol Chem 193:265–275
Kuhlmann MK, Burkhardt G, Kohler H (1997) Insights into potential cellular mechanisms of cisplatin nephrotoxicity and their clinical application. Nephrol Dial Transplant 12:2478–2480
Somani S, Husain K, Whitworth C, Trammell G, Malafa M, Rybak L (2000) Dose-dependent protection by lipoic acid against cisplatin-induced nephrotoxicity in rats: antioxidant defense system. Pharmacol Toxicol 86(5):234–241
Somani SM, Ravi R, Rybak LP (1995) Diethyldithiocarbamate protection against cisplatin nephrotoxicity: antioxidant system. Drug Chem Toxicol 18(2–3):151–170
Badary OA, Nagi MN, Al-Shabanah OA, Al-Sawaf HA, Al-Sohaibani MO, Al-Bekairi AM (1997) Thymoquinone ameliorates the nephrotoxicity induced by cisplatin in rodents and potentiates its antitumor activity. Can J Physiol Pharmacol 75(12):1356–1361
Yoshida M, Khokhar A, Kido Y, Ali-Osman F, Siddik Z (1994) Correlation of total and interstrand DNA adducts in tumor and kidney with antitumor efficacies and differential nephrotoxicities of cis-ammine/cyclohexylamine-dichloroplatinum (II) and cisplatin. Biochem Pharmacol 17:793–799
Appenroth D, Frob S, Kersten L, Splinter FK, Winnefeld K (1997) Protective effects of vitamin E and C on cisplatin nephrotoxicity in developing rats. Arch Toxicol 71:677–683
Braunlich H, Appenroth D, Fleck C (1997) Protective effects of methimazole against cisplatin-induced nephrotoxicity in rats. J Appl Toxicol 17:41–45
Davis CA, Nick HS, Agarwal A (2001) Manganese superoxide dismutase attenuates cisplatin-induced renal injury: importance of superoxide. J Am Soc Nephrol 12:2683–2690
Husain K, Morris C, Whitworth C, Trammell GL, Rybak LP, Somani SM (1998) Protection by ebselen against cisplatin-induced nephrotoxicity: antioxidant system. Mol Cell Biochem 178:127–133
Liu H, Baliga R (2000) Effect of iron chelator, hydroxyl radical scavenger and cytochrome P450 inhibitors on the cytotoxicity of cisplatin to tumor cells. Anticancer Res 20:4547–4550
Mishima K, Hidaka S, Takamura N, Shinozawa S (1999) Protection against cis-diamminedichloroplatinum-induced nephrotoxicity by 2,3-dimercaptosuccinic acid in rats. Ren Fail 21:593–602
Nishikawa M, Nagatomi H, Nishijima M, Ohira G, Chang BJ, Sato E, Inoue M (2001) Targeting superoxide dismutase to renal proximal tubule cells inhibits nephrotoxicity of cisplatin and increases the survival of cancer-bearing mice. Cancer Lett 171:133–138
Yuhas JM, Culo F (1980) Selective inhibition of the nephrotoxicity of cis-dichlorodiammineplatinum (II) by WR-2721 without altering its antitumor properties. Cancer Treat Rep 64:57–64
Sadzuka Y, Shoji T, Takino Y (1992) Effect of cisplatin on the activities of enzymes which protect against lipid peroxidation. Biochem Pharmacol 43:1873–1875
Halliwell B, Gutteridge JM (1989) Protection against oxidants in biological systems: the superoxide theory of oxygen toxicity. In: Halliwell B, Gutteridge JM (eds), Free radicals in biology and medicine, 2nd ed. Oxford: Clarendon. p 87–187
Sherlock S (1997) Assessment of liver function disease of liver and biliary system, 10th ed. Blackwell Science Ltd., London. p 17–32
Iseri S, Ercan F, Gedik N, Yuksel M, Alican I (2007) Simvastatin attenuates cisplatin-induced kidney and liver damage in rats. Toxicol 230:256–264
Acknowledgments
The authors are thankful to the Council of Scientific and Industrial Research and the Board of Research in Nuclear Sciences for financial assistance to conduct this study. The authors would also like to express their thanks to Bananas Hindu University for providing a scholarship for the associated student.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tripathi, Y.B., Nagwani, S., Mishra, P. et al. Protective effect of Pueraria tuberosa DC. embedded biscuit on cisplatin-induced nephrotoxicity in mice. J Nat Med 66, 109–118 (2012). https://doi.org/10.1007/s11418-011-0559-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-011-0559-1