Abstract
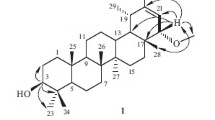
A new oligostilbenoid derivative, diptoindonesin F (1), along with five known oligostilbenoids, (−)-ampelopsin A (2), (−)-α-viniferin (3), ampelopsin E (4), (−)-vaticanol B (5), and (−)-hemsleyanol D (6), were isolated from the methanol extract of the tree bark of Shorea gibbosa. The structure of the new compound was determined based on the analysis of spectroscopic data, including UV, IR, NMR 1-D and 2-D, and mass spectra. Cytotoxic properties of the isolated oligostilbenoids were evaluated against murine leukemia P-388 cells with the result that compounds 2 and 4 showed the highest cytotoxicity.


Similar content being viewed by others
References
Sotheeswaran S, Pasupathy V (1993) Distribution of resveratrol oligomers in plants. Phytochemistry 32:1083–1092
Seo EK, Douglas KA (2000) Bioactive constituents of the family Dipterocarpaceae. In: Atta-ur-Rahman (ed) Studies in natural products chemistry. vol 23 (Bioactive Natural Products (Part D)), Elsevier, pp 531–561
Cichewicz RH, Kouzi SA (2002) Resveratrol oligomers: structure, chemistry, and biological activity. In: Atta-ur-Rahman (ed) Studies in natural products chemistry. vol 26 (Bioactive Natural Products, (Part G)), Elsevier, pp 507–579
Aminah NS, Achmad SA, Aimi N, Ghisalberti EL, Hakim EH, Kitajima M, Syah YM, Takayama H (2002) Diptoindonesin A, a new C-glucoside of ε-viniferin from Shorea seminis (Dipterocarpaceae). Fitoterapia 73:501–507
Syah YM, Aminah NS, Hakim EH, Kitajima M, Takayama H, Achmad SA (2003) Two Oligostilbenoids cis- and trans-diptoindonesian B from Dryobalanops oblongifolia. Phytochemistry 63:913–917
Sahidin, Hakim EH, Juliawaty LD, Syah YM, Din LB, Ghisalberti EL, Latip J, Said IM, Achmad SA (2005) Cytotoxic properties of oligostilbenoids from the tree bark of Hopea dryobalanoides. Z Naturforsch 60c:723–727
Muhtadi, Hakim EH, Juliawaty LD, Syah YM, Achmad SA, Latif J, Ghisalberti EL (2006) Cytotoxic resveratrol oligomers from the tree bark of Dipterocarpus hasseltii (Dipterocarpaceae). Fitoterapia 77:550–555
Oshima Y, Ueno Y, Hikino H, Yang LL, Yen KY (1990) Ampelopsins A, B and C, new oligostilbenes of Ampelopsis brevipedunculata var. hancei. Tetrahedron 46:5121–5126
Pryce RJ, Langcake P (1977) (−)-α-Viniferin: an antifungal resveratrol trimer from grapevines. Phytochemistry 16:1452–1454
Oshima Y, Ueno Y, (1993) Ampelopsins D, E, H, and cis-ampelopsin E, oligostilbenes from Ampelopsis brevipedunculata var. hancei roots. Phytochemistry 33:179–82
Tanaka T, Ito T, Nakaya K, Iinuma M, Riswan S (2000) Oligostilbenoids in the stem bark of Vatica rassak. Phytochemistry 54:63–69
Tanaka T, Ito T, Nakaya K-I, Iinuma M, Takahashi Y, Naganawa H, Riswan S (2001) Six new heterocyclic stilbene oligomers from stem bark of Shorea hemsleyana. Heterocycles 55:729–740
Kawabata J, Fukushi E, Hara M, Mizutani J (1992) Detection of connectivity between equivalent carbons in a C2 molecule using isotopomeric asymmetry: Identification of hopeaphenol in Carex pumila. Magn Reson Chem 30:6–10
Yan KX, Terashima K, Takaya Y, Niwa M (2001) A novel oligostilbene named (+)-viniferol A from the stem bark of Vitis vinifera ‘Kyohou’. Tetrahedron 57:2711–2715
Ito T, Tanaka T, Ido Y, Nakaya KI, Iinuma M, Riswan S (2000) Four new stilbenoid C-glucosides isolated from the stem bark of Shorea hemsleyana. Chem Pharm Bull 48:1959–1963
Acknowledgments
We thank Dr. H. Karasawa and Dr. A. Kusai from JEOL, Japan, for mass spectra measurements. We also thank the Herbarium Bogoriense, Bogor, Indonesia, for identification of the plant specimen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saroyobudiono, H., Juliawaty, L.D., Syah, Y.M. et al. Oligostilbenoids from Shorea gibbosa and their cytotoxic properties against P-388 cells. J Nat Med 62, 195–198 (2008). https://doi.org/10.1007/s11418-007-0205-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-007-0205-0