Abstract
Purpose
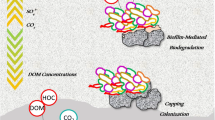
Release of polycyclic aromatic hydrocarbons (PAH) can impact surface water and overlaying sediments. Contaminant control strategies include in situ containment technologies like capping, which consists of placing layers of inert or reactive media on top of the impacted sediment. Cap placement alters the sediment environment and can influence natural processes such as biodegradation of PAH. This study examined the combined influence of capping media (sand, clay, and granular activated carbon (GAC)) and reducing conditions on the PAH biodegradation potential of benthic microbial communities.
Materials and methods
Microcosms containing naphthalene (a model PAH), benthic microbial communities enriched from PAH-impacted sediments, and capping materials were prepared to mimic sediment environments under different redox conditions (nitrate, iron, and sulfate). Microcosms were monitored for 100 days to assess changes in microbial community composition using 16S rRNA gene surveys and naphthalene biodegradation rates.
Results and discussion
Multivariate analyses showed that microbial community shifts were significant with varying capping materials and redox conditions (PERMANOVA p < 0.05). Microorganisms linked to PAH biodegradation were enriched, and the increased abundance of these genera was positively correlated (PERMANOVA p < 0.05), indicating the formation of consortia with naphthalene biodegradation potential. Enrichment of microorganisms associated with PAH biodegradation was most pronounced in microcosms where naphthalene transformation rate ratios were the highest.
Conclusion
Capping media promote the development of consortia with naphthalene biodegradation potential and affect the extent to which in situ redox conditions impact biodegradation. These findings provide insights on the relationships between capping materials and indigenous microbial communities and suggest that strategic capping material selection and placement can enrich desired communities at contaminated sites under a wide range of electron-accepting conditions.




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abbate C, Ambrosoli R, Minati JL et al (2013) Metabolic and molecular methods to evaluate the organoclay effects on a bacterial community. Environ Pollut 179:39–44. https://doi.org/10.1016/j.envpol.2013.04.012
Abbate C, Arena M, Baglieri A, Gennari M (2009) Effects of organoclays on soil eubacterial community assessed by molecular approaches. J Hazard Mater 168:466–472. https://doi.org/10.1016/j.jhazmat.2009.02.050
Abdel-Shafy HI, Mansour MSM (2016) A review on polycyclic aromatic hydrocarbons: source, environmental impact, effect on human health and remediation. Egypt J Pet 25:107–123. https://doi.org/10.1016/j.ejpe.2015.03.011
Anderson RT, Lovley DR (1999) Naphthalene and benzene degradation under Fe(III)-reducing conditions in petroleum-contaminated aquifers. Bioremediation J 3:121–135. https://doi.org/10.1080/10889869991219271
Battin TJ, Besemer K, Bengtsson MM et al (2016) The ecology and biogeochemistry of stream biofilms. Nat Rev Microbiol 14:251–263. https://doi.org/10.1038/nrmicro.2016.15
Biswas B, Juhasz AL, Mahmudur Rahman M, Naidu R (2020) Modified clays alter diversity and respiration profile of microorganisms in long-term hydrocarbon and metal co-contaminated soil. Microb Biotechnol 13:522–534. https://doi.org/10.1111/1751-7915.13510
Bokulich NA, Rideout JR, Kopylova E et al (2015) A standardized, extensible framework for optimizing classification improves marker-gene taxonomic assignments. PeerJ PrePrints
Bonaglia S, Broman E, Brindefalk B et al (2020) Activated carbon stimulates microbial diversity and PAH biodegradation under anaerobic conditions in oil-polluted sediments. Chemosphere 248:126023. https://doi.org/10.1016/j.chemosphere.2020.126023
Braun F, Hamelin J, Bonnafous A et al (2015) Similar PAH fate in anaerobic digesters inoculated with three microbial communities accumulating either volatile fatty acids or methane. PLoS ONE 10:e0125552. https://doi.org/10.1371/journal.pone.0125552
Callahan BJ, Sankaran K, Fukuyama JA et al (2016) Bioconductor workflow for microbiome data analysis: from raw reads to community analyses. F1000Research 5:1492. https://doi.org/10.12688/f1000research.8986.2
Chang B-V, Chang IT, Yuan SY (2008) Anaerobic degradation of phenanthrene and pyrene in mangrove sediment. Bull Environ Contam Toxicol 80:145–149. https://doi.org/10.1007/s00128-007-9333-1
Chang W, Um Y, Holoman TRP (2006) Polycyclic aromatic hydrocarbon (PAH) degradation coupled to methanogenesis. Biotechnol Lett 28:425–430. https://doi.org/10.1007/s10529-005-6073-3
Coates JD, Anderson RT, Woodward JC et al (1996) Anaerobic hydrocarbon degradation in petroleum-contaminated harbor sediments under sulfate-reducing and artificially imposed iron-reducing conditions. Environ Sci Technol 30:2784–2789. https://doi.org/10.1021/es9600441
Cornelissen G, Elmquist Kruså M, Breedveld GD et al (2011) Remediation of contaminated marine sediment using thin-layer capping with activated carbon—a field experiment in Trondheim Harbor, Norway. Environ Sci Technol 45:6110–6116. https://doi.org/10.1021/es2011397
Cornelissen G, van Noort PCM, Govers HAJ (1997) Desorption kinetics of chlorobenzenes, polycyclic aromatic hydrocarbons, and polychlorinated biphenyls: sediment extraction with Tenax® and effects of contact time and solute hydrophobicity. Environ Toxicol Chem 16:1351–1357. https://doi.org/10.1002/etc.5620160703
Cornelissen G, van Noort PCM, Govers HAJ (1998) Mechanism of slow desorption of organic compounds from sediments: a study using model sorbents. Environ Sci Technol 32:3124–3131. https://doi.org/10.1021/es970976k
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. https://doi.org/10.1093/bioinformatics/btq461
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Edgar RC, Haas BJ, Clemente JC et al (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Edwards SJ, Kjellerup BV (2013) Applications of biofilms in bioremediation and biotransformation of persistent organic pollutants, pharmaceuticals/personal care products, and heavy metals. Appl Microbiol Biotechnol 97:9909–9921. https://doi.org/10.1007/s00253-013-5216-z
Folwell BD, McGenity TJ, Price A et al (2016) Exploring the capacity for anaerobic biodegradation of polycyclic aromatic hydrocarbons and naphthenic acids by microbes from oil-sands-process-affected waters. Int Biodeterior Biodegrad 108:214–221. https://doi.org/10.1016/j.ibiod.2014.12.016
Freidman BL, Gras SL, Snape I et al (2017) A bio-reactive barrier sequence for petroleum hydrocarbon capture and degradation in low nutrient environments. Int Biodeterior Biodegrad 116:26–37. https://doi.org/10.1016/j.ibiod.2016.09.025
Hale SE, Meynet P, Davenport RJ et al (2010) Changes in polycyclic aromatic hydrocarbon availability in River Tyne sediment following bioremediation treatments or activated carbon amendment. Water Res 44:4529–4536. https://doi.org/10.1016/j.watres.2010.06.027
Haritash AK, Kaushik CP (2009) Biodegradation aspects of polycyclic aromatic hydrocarbons (PAHs): a review. J Hazard Mater 169:1–15. https://doi.org/10.1016/j.jhazmat.2009.03.137
Himmelheber DW, Pennell KD, Hughes JB (2011) Evaluation of a laboratory-scale bioreactive in situ sediment cap for the treatment of organic contaminants. Water Res 45:5365–5374. https://doi.org/10.1016/j.watres.2011.06.022
Himmelheber DW, Thomas SH, Löffler FE et al (2009) Microbial colonization of an in situ sediment cap and correlation to stratified redox zones. Environ Sci Technol 43:66–74. https://doi.org/10.1021/es801834e
Hyun S, Jafvert CT, Lee LS, Rao PSC (2006) Laboratory studies to characterize the efficacy of sand capping a coal tar-contaminated sediment. Chemosphere 63:1621–1631. https://doi.org/10.1016/j.chemosphere.2005.10.025
Jiao S, Chen W, Wang E et al (2016) Microbial succession in response to pollutants in batch-enrichment culture. Sci Rep 6:21791. https://doi.org/10.1038/srep21791
Johnsen AR, Wick LY, Harms H (2005) Principles of microbial PAH-degradation in soil. Environ Pollut 133:71–84. https://doi.org/10.1016/j.envpol.2004.04.015
Kan AT, Gongmin Fu, Tomson MB (1994) Adsorption/desorption hysteresis in organic pollutant and soil/sediment interaction. Environ Sci Technol 28:859–867. https://doi.org/10.1021/es00054a017
Kim YS, Nyberg LM, Jenkinson B, Jafvert CT (2013) PAH concentration gradients and fluxes through sand cap test cells installed in situ over river sediments containing coal tar. Environ Sci Process Impacts 15:1601. https://doi.org/10.1039/c3em00142c
Kümmel S, Herbst F-A, Bahr A et al (2015) Anaerobic naphthalene degradation by sulfate-reducing Desulfobacteraceae from various anoxic aquifers. FEMS Microbiol Ecol 91. https://doi.org/10.1093/femsec/fiv006
Kwon MJ, Finneran KT (2006) Microbially mediated biodegradation of hexahydro-1,3,5-trinitro-1,3,5- triazine by extracellular electron shuttling compounds. Appl Environ Microbiol 72:5933–5941. https://doi.org/10.1128/AEM.00660-06
Leglize P, Alain S, Jacques B, Corinne L (2008) Adsorption of phenanthrene on activated carbon increases mineralization rate by specific bacteria. J Hazard Mater 151:339–347. https://doi.org/10.1016/j.jhazmat.2007.05.089
Lei L, Khodadoust AP, Suidan MT, Tabak HH (2005) Biodegradation of sediment-bound PAHs in field-contaminated sediment. Water Res 39:349–361. https://doi.org/10.1016/j.watres.2004.09.021
Li C-H, Zhou H-W, Wong Y-S, Tam NF-Y (2009) Vertical distribution and anaerobic biodegradation of polycyclic aromatic hydrocarbons in mangrove sediments in Hong Kong, South China. Sci Total Environ 407:5772–5779. https://doi.org/10.1016/j.scitotenv.2009.07.034
Lu Z, Deng Y, Van Nostrand JD et al (2012) Microbial gene functions enriched in the Deepwater Horizon deep-sea oil plume. ISME J 6:451–460. https://doi.org/10.1038/ismej.2011.91
Ma C, Wang Y, Zhuang L et al (2011) Anaerobic degradation of phenanthrene by a newly isolated humus-reducing bacterium, Pseudomonas aeruginosa strain PAH-1. J Soils Sediments 11:923–929. https://doi.org/10.1007/s11368-011-0368-x
Mangwani N, Kumari S, Das S (2016) Bacterial biofilms and quorum sensing: fidelity in bioremediation technology. Biotechnol Genet Eng Rev 32:43–73. https://doi.org/10.1080/02648725.2016.1196554
Martin F, Torelli S, Le Paslier D et al (2012) Betaproteobacteria dominance and diversity shifts in the bacterial community of a PAH-contaminated soil exposed to phenanthrene. Environ Pollut 162:345–353. https://doi.org/10.1016/j.envpol.2011.11.032
McMurdie PJ, Holmes S (2013) phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8:e61217. https://doi.org/10.1371/journal.pone.0061217
Meynet P, Hale SE, Davenport RJ et al (2012) Effect of activated carbon amendment on bacterial community structure and functions in a PAH impacted urban soil. Environ Sci Technol 46:5057–5066. https://doi.org/10.1021/es2043905
Mittal M, Rockne KJ (2008) Indole production by Pseudomonas stutzeri strain NAP-3 during anaerobic naphthalene biodegradation in the presence of dimethyl formamide. J Environ Sci Health Part A 43:1027–1034. https://doi.org/10.1080/10934520802059896
Montgomery JH (2000) Groundwater chemicals desk reference, 3rd edn. CRC Lewis Publishers, Boca Raton
Müller JB, Ramos DT, Larose C et al (2017) Combined iron and sulfate reduction biostimulation as a novel approach to enhance BTEX and PAH source-zone biodegradation in biodiesel blend-contaminated groundwater. J Hazard Mater 326:229–236. https://doi.org/10.1016/j.jhazmat.2016.12.005
Navarrete-Euan H, Rodríguez-Escamilla Z, Pérez-Rueda E et al (2021) Comparing sediment microbiomes in contaminated and pristine wetlands along the coast of Yucatan. Microorganisms 9:877. https://doi.org/10.3390/microorganisms9040877
Oksanen J, Blanchet FG, Kindt R et al (2015) vegan: community ecology package.
Pagnozzi G, Carroll S, Reible DD, Millerick K (2020) Biological natural attenuation and contaminant oxidation in sediment caps: recent advances and future opportunities. Curr Pollut Rep 6:281–294. https://doi.org/10.1007/s40726-020-00153-5
Pagnozzi G, Carroll S, Reible DD, Millerick K (2021a) Powdered activated carbon (PAC) amendment enhances naphthalene biodegradation under strictly sulfate-reducing conditions. Environ Pollut 268:115641. https://doi.org/10.1016/j.envpol.2020.115641
Pagnozzi G, Reible DD, Millerick K (2021b) The effects of adsorptive materials on microbial community composition and PAH degradation at the sediment cap–water interface. Int J Sediment Res 36:555–565. https://doi.org/10.1016/j.ijsrc.2020.10.006
Pérez-Cobas AE, Gomez-Valero L, Buchrieser C (2020) Metagenomic approaches in microbial ecology: an update on whole-genome and marker gene sequencing analyses. Microb Genomics 6. https://doi.org/10.1099/mgen.0.000409
Rabus R, Boll M, Heider J et al (2016) Anaerobic microbial degradation of hydrocarbons: from enzymatic reactions to the environment. Microb Physiol 26:5–28. https://doi.org/10.1159/000443997
Ramos-Padrón E, Bordenave S, Lin S et al (2011) Carbon and sulfur cycling by microbial communities in a gypsum-treated oil sands tailings pond. Environ Sci Technol 45:439–446. https://doi.org/10.1021/es1028487
Reible DD (ed) (2014) Processes, assessment and remediation of contaminated sediments. Springer, New York, NY
Rockne KJ, Chee-Sanford JC, Sanford RA et al (2000) Anaerobic naphthalene degradation by microbial pure cultures under nitrate-reducing conditions. Appl Environ Microbiol 66:1595–1601. https://doi.org/10.1128/AEM.66.4.1595-1601.2000
Samuelsson GS, Hedman JE, Elmquist Kruså M et al (2015) Capping in situ with activated carbon in Trondheim harbor (Norway) reduces bioaccumulation of PCBs and PAHs in marine sediment fauna. Mar Environ Res 109:103–112. https://doi.org/10.1016/j.marenvres.2015.06.003
Smith AM, Kirisits MJ, Reible DD (2012) Assessment of potential anaerobic biotransformation of organic pollutants in sediment caps. New Biotechnol 30:80–87. https://doi.org/10.1016/j.nbt.2012.06.003
Stagars MH, Mishra S, Treude T et al (2017) Microbial community response to simulated petroleum seepage in Caspian Sea sediments. Front Microbiol 8:764. https://doi.org/10.3389/fmicb.2017.00764
Thomas C, Reible D (2015) Modeling compound loss from polydimethylsiloxane passive samplers. Chromatography 2:611–624. https://doi.org/10.3390/chromatography2040611
Van der Zee FP, Cervantes FJ (2009) Impact and application of electron shuttles on the redox (bio)transformation of contaminants: a review. Biotechnol Adv 27:256–277. https://doi.org/10.1016/j.biotechadv.2009.01.004
Vogt C, Gödeke S, Treutler H-C et al (2007) Benzene oxidation under sulfate-reducing conditions in columns simulating in situ conditions. Biodegradation 18:625–636. https://doi.org/10.1007/s10532-006-9095-1
Wang XQ, Thibodeaux LJ, Valsaraj KT, Reible DD (1991) Efficiency of capping contaminated bed sediments in situ. 1. Laboratory-scale experiments on diffusion-adsorption in the capping layer. Environ Sci Technol 25:1578–1584. https://doi.org/10.1021/es00021a008
Weissenfels WD, Klewer H-J, Langhoff J (1992) Adsorption of polycyclic aromatic hydrocarbons (PAHs) by soil particles: influence on biodegradability and biotoxicity. Appl Microbiol Biotechnol 36. https://doi.org/10.1007/BF00183251
Winderl C, Anneser B, Griebler C et al (2008) Depth-resolved quantification of anaerobic toluene degraders and aquifer microbial community patterns in distinct redox zones of a tar oil contaminant plume. Appl Environ Microbiol 74:792–801. https://doi.org/10.1128/AEM.01951-07
Yan S, Wu G (2017) Reorganization of gene network for degradation of polycyclic aromatic hydrocarbons (PAHs) in Pseudomonas aeruginosa PAO1 under several conditions. J Appl Genet 58:545–563. https://doi.org/10.1007/s13353-017-0402-9
Ye Q, Liang C, Chen X et al (2019) Molecular characterization of methanogenic microbial communities for degrading various types of polycyclic aromatic hydrocarbon. J Environ Sci 86:97–106. https://doi.org/10.1016/j.jes.2019.04.027
Zhang J, Kobert K, Flouri T, Stamatakis A (2014) PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30:614–620. https://doi.org/10.1093/bioinformatics/btt593
Zhang T, Bain TS, Nevin KP et al (2012) Anaerobic benzene oxidation by Geobacter species. Appl Environ Microbiol 78:8304–8310. https://doi.org/10.1128/AEM.02469-12
Zhang X, Young LY (1997) Carboxylation as an initial reaction in the anaerobic metabolism of naphthalene and phenanthrene by sulfidogenic consortia. Appl Environ Microbiol 63:4759–4764. https://doi.org/10.1128/aem.63.12.4759-4764.1997
Funding
This work was financially supported by the Electric Power Research Institute (EPRI) and the Department of Civil, Construction & Environmental Engineering at Texas Tech University (Lubbock, Texas).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Victor Magar
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pagnozzi, G., Reible, D. & Millerick, K. Shifting biogeochemical conditions and capping media impact composition and behavior of PAH-enriched microbial communities. J Soils Sediments 23, 3262–3274 (2023). https://doi.org/10.1007/s11368-023-03587-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-023-03587-8